Particles and atomic structure — WJEC GCSE Study Guide
Exam Board: WJEC | Level: GCSE
Master the fundamental building blocks of Chemistry. This topic covers the particulate model, atomic structure, and isotopes — essential knowledge that underpins the entire specification and guarantees you exam marks.

## Overview
Welcome to the foundation of Chemistry. The study of **Particles and Atomic Structure** is arguably the most important topic in the entire GCSE specification. Why? Because everything else you will study — from chemical bonding and quantitative chemistry to organic reactions and electrolysis — relies on a solid understanding of what atoms are and how they behave.
This topic explores the particulate model of matter, distinguishing between physical and chemical changes, before zooming in to the subatomic level. You will learn about the nuclear model of the atom, the properties of protons, neutrons, and electrons, and how to deduce the electronic structure of the first 20 elements. We will also cover isotopes and their role in calculating relative atomic mass.
Examiners frequently test this topic across all papers. You can expect a mix of short recall questions (e.g., stating the relative charge of a neutron), calculation questions (e.g., determining the number of subatomic particles), and longer extended-response questions asking you to compare models of the atom or explain why isotopes share chemical properties. Let's secure those marks.

## Key Concepts
### Concept 1: The Particulate Model of Matter
The particulate model is our way of describing how matter behaves in different states. Everything is made of particles, and their arrangement and energy determine whether a substance is a solid, liquid, or gas.
In a **solid**, particles are packed closely together in a regular lattice arrangement. They cannot move from their positions but vibrate around fixed points. Because they are so close, solids cannot be compressed. In a **liquid**, particles are still mostly touching but are arranged randomly. They have more energy than in a solid, allowing them to move over and past one another, which is why liquids can flow and take the shape of their container. In a **gas**, particles are spread far apart and move rapidly and randomly in all directions. Gases can be easily compressed because of the large empty spaces between particles.
**Examiner Tip**: When describing changes of state (e.g., melting or boiling), always refer to the *energy* of the particles and the *forces of attraction* between them. For a solid to melt, energy must be supplied to overcome the forces of attraction holding the particles in their regular arrangement.
### Concept 2: Physical vs. Chemical Changes
Examiners often ask candidates to distinguish between physical and chemical changes. The key difference lies in whether a new substance is formed.
A **physical change** involves a change in state or shape, but no new substances are created. The particles themselves remain unchanged, they simply rearrange. Physical changes are usually easy to reverse. Examples include melting ice, dissolving salt in water, or crushing a can.
A **chemical change** (or chemical reaction) results in the formation of one or more new substances. The bonds between atoms are broken and new bonds are formed. Chemical changes are typically difficult or impossible to reverse. Signs that a chemical change has occurred include a colour change, a temperature change (exothermic or endothermic), or the production of a gas (effervescence). Examples include burning magnesium, rusting iron, or neutralising an acid.
### Concept 3: The Nuclear Model of the Atom
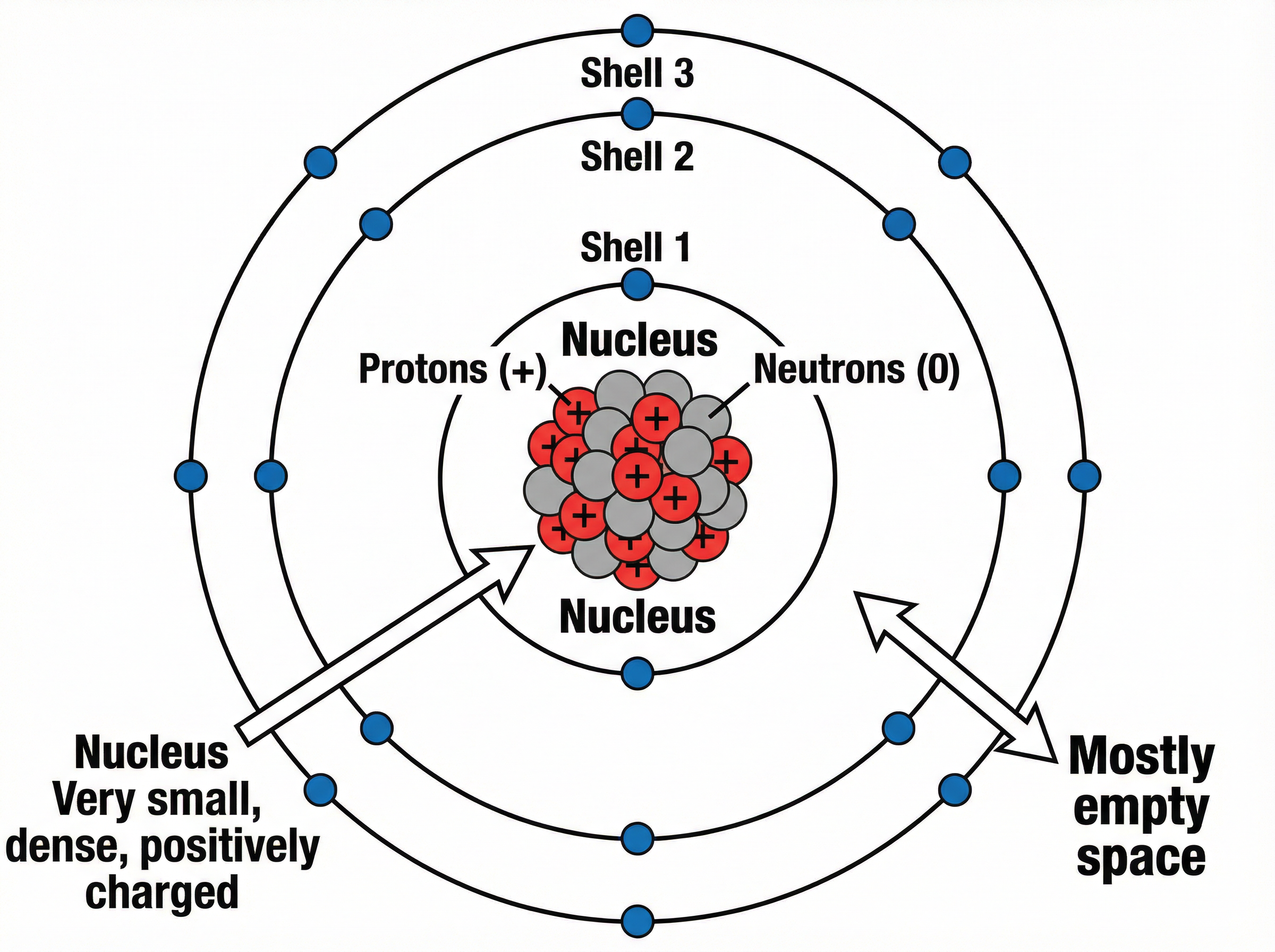
The modern accepted model of the atom is the nuclear model. It describes the atom as having a tiny, dense, positively charged central core called the **nucleus**, surrounded by negatively charged **electrons** orbiting in specific energy levels (shells).
The nucleus contains two types of subatomic particles: **protons** (which are positively charged) and **neutrons** (which have no charge; they are neutral). Because the nucleus contains only positive and neutral particles, its overall charge is positive.
However, the atom as a whole is mostly empty space. To put it in perspective, the radius of an atom is approximately $1 \times 10^{-10}$ metres. The radius of the nucleus is about $1 \times 10^{-14}$ metres. This means the nucleus is about 10,000 times smaller than the atom itself.
**Why is an atom electrically neutral?**
This is a classic 2-mark exam question. An atom has no overall electrical charge because it contains an equal number of positive protons and negative electrons. The opposite charges exactly cancel each other out.
### Concept 4: Subatomic Particles
You must memorise the relative charges and relative masses of the three subatomic particles.
| Particle | Relative Mass | Relative Charge | Location |
|----------|---------------|-----------------|----------|
| Proton | 1 | +1 | Nucleus |
| Neutron | 1 | 0 | Nucleus |
| Electron | Very small (approx. 1/2000) | -1 | Shells |
**Examiner Tip**: Never write that the mass of an electron is zero. It is "very small" or "negligible". Also, never state that electrons are inside the nucleus.
### Concept 5: Atomic Number and Mass Number
Every element on the Periodic Table is defined by its **atomic number** (also called the proton number). The atomic number tells you the number of protons in the nucleus. For example, every atom with 6 protons is carbon.
The **mass number** is the total number of protons and neutrons combined.
You must be able to calculate the number of each subatomic particle from these two numbers:
- **Number of protons** = Atomic number
- **Number of electrons** = Atomic number (in a neutral atom)
- **Number of neutrons** = Mass number - Atomic number
**Example**: Calculate the subatomic particles in Sodium ($^{23}_{11}\text{Na}$).
- Protons = 11 (from the atomic number)
- Electrons = 11 (because atoms are neutral)
- Neutrons = 23 - 11 = 12
### Concept 6: Electronic Structure
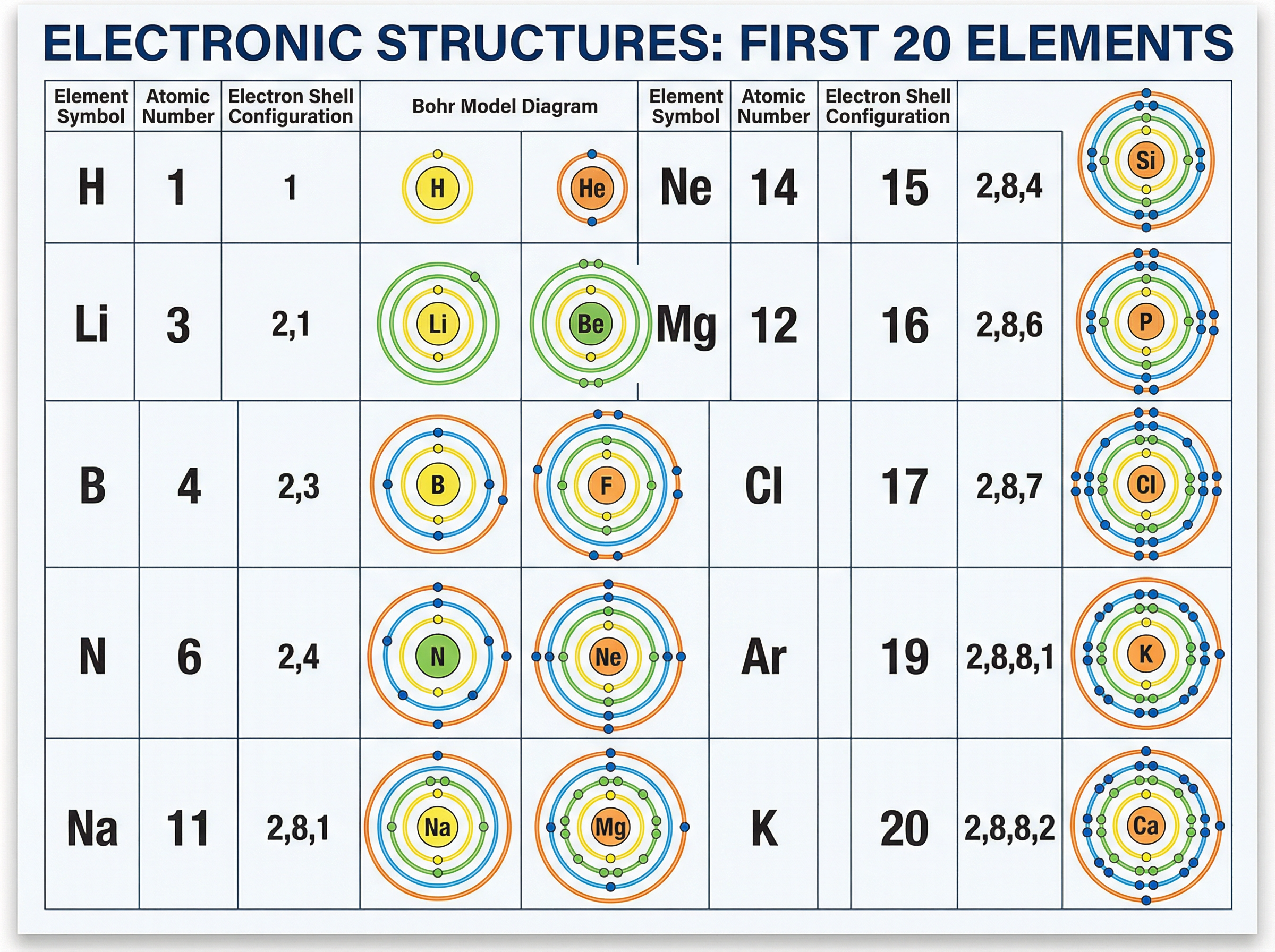
Electrons do not orbit randomly; they occupy specific energy levels called shells. The shells are filled from the lowest energy level (closest to the nucleus) outwards.
There is a strict rule for the maximum number of electrons each shell can hold for the first 20 elements:
- 1st shell: maximum 2 electrons
- 2nd shell: maximum 8 electrons
- 3rd shell: maximum 8 electrons
For example, Magnesium has an atomic number of 12, meaning it has 12 electrons. We fill the shells: 2 in the first, 8 in the second, leaving 2 for the third. Its electronic structure is written as **2,8,2**.
**The Periodic Table Connection**:
- The number of electrons in the outer shell tells you the **Group** the element is in. (Magnesium has 2 outer electrons $\rightarrow$ Group 2).
- The number of occupied shells tells you the **Period** the element is in. (Magnesium has 3 shells $\rightarrow$ Period 3).
### Concept 7: Isotopes and Relative Atomic Mass
**Isotopes** are atoms of the same element that have the same number of protons but a different number of neutrons. This means they have the same atomic number but different mass numbers.
Because isotopes have the same number of protons, they also have the same number of electrons. The electronic structure dictates an element's chemical reactions. Therefore, **isotopes have identical chemical properties**. However, because they have different masses, their physical properties (like density or boiling point) may differ slightly.
The **relative atomic mass ($A_r$)** of an element is an average value that takes into account the abundance of the isotopes of the element. This is why the $A_r$ of chlorine is 35.5 — it is a weighted average of its two main isotopes, Chlorine-35 (75% abundance) and Chlorine-37 (25% abundance).
## Mathematical/Scientific Relationships
**Calculating Neutrons:**
$\text{Number of Neutrons} = \text{Mass Number} - \text{Atomic Number}$
*(Must memorise)*
**Calculating Relative Atomic Mass ($A_r$):**
$A_r = \frac{(\text{mass of isotope 1} \times \text{abundance 1}) + (\text{mass of isotope 2} \times \text{abundance 2})}{100}$
*(Must memorise)*
**Standard Form for Atomic Radius:**
Radius of an atom $\approx 1 \times 10^{-10}$ m.
Radius of a nucleus $\approx 1 \times 10^{-14}$ m.
*(Must memorise)*
## Practical Applications
Understanding atomic structure is crucial for modern technology. For example, our knowledge of isotopes allows us to use Carbon-14 for radiocarbon dating of historical artefacts, and specific isotopes of iodine and technetium in medical imaging and cancer treatments. Furthermore, the principles of electronic structure underpin the entire semiconductor industry, which powers the computer or phone you are using right now.