Production, use and disposal of important chemicals and materials — WJEC GCSE Study Guide
Exam Board: WJEC | Level: GCSE
Master the essential industrial processes that shape our modern world, from the Haber process and fertiliser production to materials science and corrosion. This topic connects chemical principles with real-world applications, teaching you how to evaluate the environmental impact and sustainability of the materials we rely on daily.

## Overview
Topic 11: Production, Use and Disposal of Important Chemicals and Materials is where abstract chemistry meets the real world. This crucial section explores how we manufacture the substances that sustain modern society, particularly ammonia and nitrogenous fertilisers via the Haber Process. It is a highly synoptic topic, drawing heavily on your understanding of rates of reaction, reversible reactions, Le Chatelier's Principle, and quantitative chemistry.
Examiners frequently use this topic to test your ability to evaluate trade-offs in industrial processes—specifically the delicate balance between reaction rate, equilibrium yield, and economic viability. You will also be assessed on your understanding of materials science, comparing the properties of metals, alloys, ceramics, polymers, and composites to justify their uses. Finally, the topic covers environmental chemistry, requiring you to understand the mechanisms of corrosion, the impact of agricultural fertilisers, and how to interpret Life Cycle Assessments (LCAs). Expect a mix of calculation questions (atom economy and percentage yield), short factual recall, and extended 6-mark evaluation questions.
## Key Concepts
### Concept 1: The Haber Process and Compromise Conditions
The Haber Process is the industrial method used to manufacture ammonia ($NH_3$), which is essential for producing nitrogen-based fertilisers. The raw materials are nitrogen (extracted from the air via fractional distillation) and hydrogen (typically obtained from natural gas via steam reforming).
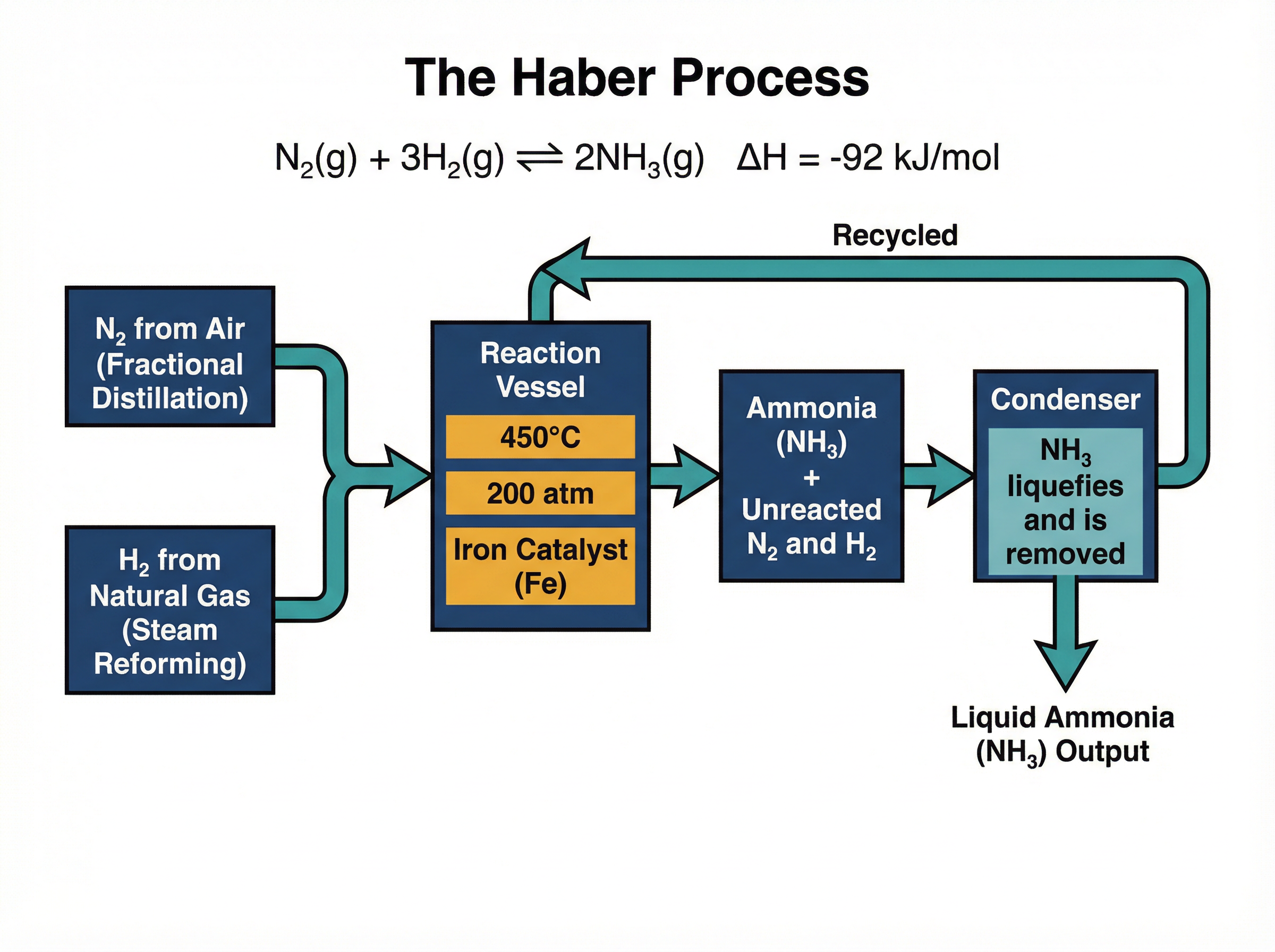
The balanced equation is:
$N_2(g) + 3H_2(g) \rightleftharpoons 2NH_3(g)$ $\Delta H = -92 \text{ kJ/mol}$
This reaction is reversible and exothermic in the forward direction. In industry, the conditions used are **$450^{\circ}C$**, **$200 \text{ atmospheres}$** of pressure, and an **iron catalyst**. These are known as 'compromise conditions' because they balance the competing demands of rate, yield, and cost.
**Temperature Compromise:** Because the forward reaction is exothermic, Le Chatelier's Principle states that a lower temperature would shift the equilibrium to the right, producing a higher yield of ammonia. However, at low temperatures, the rate of reaction is too slow to be economically viable. Therefore, a compromise temperature of $450^{\circ}C$ is used to achieve a reasonable rate of reaction while maintaining an acceptable yield.
**Pressure Compromise:** There are 4 moles of reactant gas ($1 N_2 + 3 H_2$) and only 2 moles of product gas ($2 NH_3$). Increasing the pressure shifts the equilibrium to the side with fewer moles of gas, increasing the yield of ammonia. Higher pressure also increases the rate of reaction. However, generating and containing extremely high pressures (e.g., $1000 \text{ atm}$) requires enormously expensive equipment and poses significant safety risks. Thus, $200 \text{ atm}$ is chosen as the economic and safe compromise.
**The Role of the Catalyst:** The iron catalyst speeds up both the forward and reverse reactions equally. It **does not** change the position of equilibrium or the final yield. It simply allows the system to reach equilibrium much faster, saving time and energy.
### Concept 2: Quantitative Chemistry in Industry
Industrial chemists must evaluate the efficiency of their processes. You need to master two key calculations: Percentage Yield and Atom Economy.
**Percentage Yield** compares the amount of product you actually obtained to the maximum theoretical amount you could have obtained. Yields are rarely 100% because the reaction might be reversible (not going to completion), product may be lost during separation/purification, or unexpected side reactions might occur.
**Atom Economy** measures the proportion of reactant atoms that end up in the desired useful product. A process with high atom economy produces less waste, making it more sustainable and often more profitable. Examiners frequently ask you to evaluate why a specific industrial route is chosen; high atom economy is a key justification.
### Concept 3: Corrosion and its Prevention
Corrosion is the destruction of materials by chemical reactions with substances in the environment. Rusting is a specific term for the corrosion of iron and its alloys (like steel). For iron to rust, **both water and oxygen** must be present.
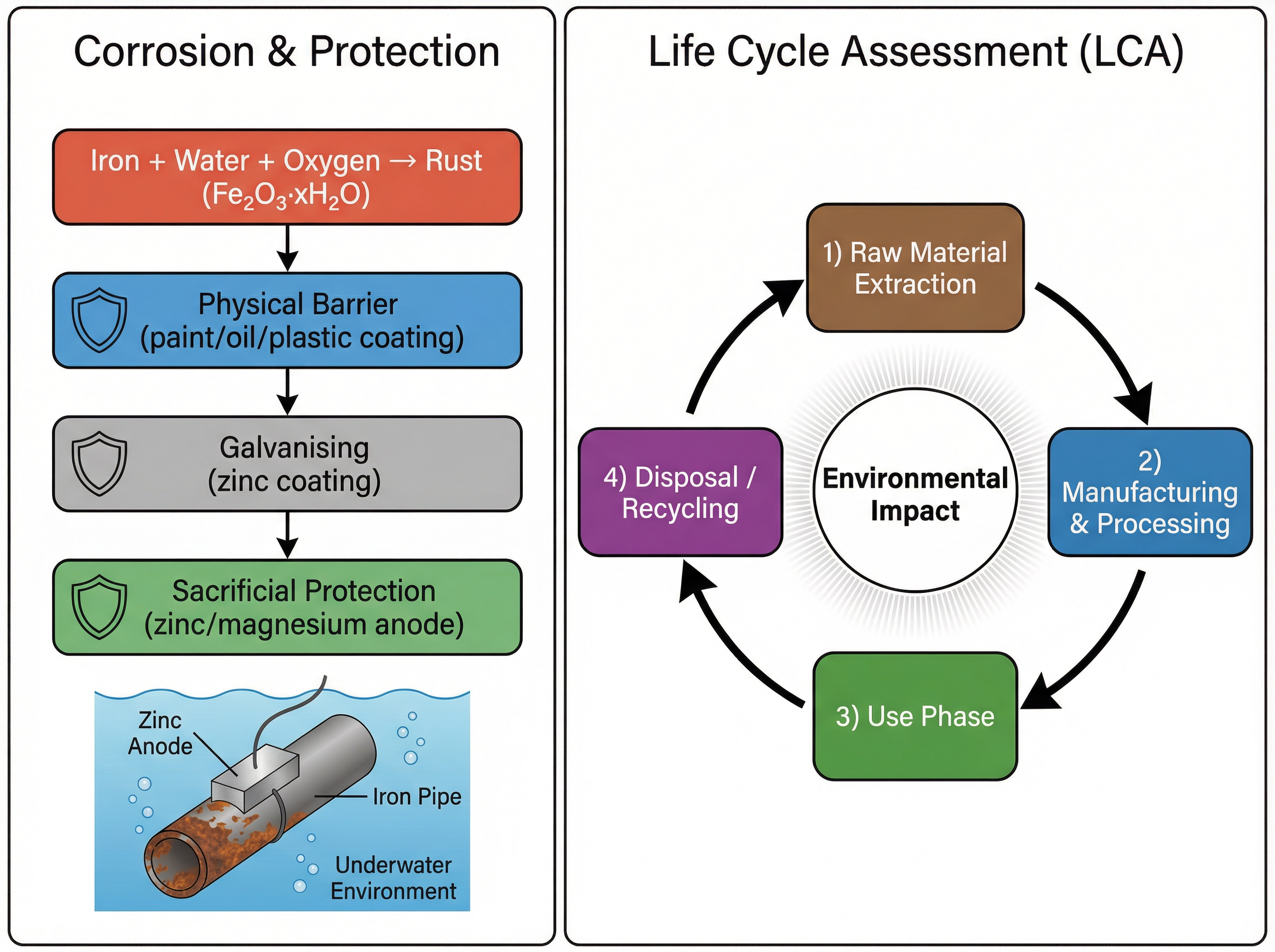
There are three main strategies to prevent corrosion:
1. **Physical Barriers:** Coating the metal with paint, oil, grease, or plastic prevents oxygen and water from reaching the surface. This only works as long as the coating is intact.
2. **Galvanising:** Coating iron with a layer of zinc. Zinc provides a physical barrier, but even if scratched, it continues to protect the iron because it acts as a sacrificial metal.
3. **Sacrificial Protection:** Attaching blocks of a more reactive metal (like zinc or magnesium) to the iron structure (e.g., ship hulls or underground pipes). The more reactive metal oxidises (corrodes) preferentially, sacrificing itself to protect the iron.
### Concept 4: Materials Science
Chemists engineer materials to suit specific purposes based on their properties.

- **Glass:** Typically soda-lime glass (made by heating sand, sodium carbonate, and limestone). It is transparent, hard, brittle, and chemically inert.
- **Ceramics:** Made by shaping wet clay and heating it in a furnace. They are hard, brittle, have high melting points, and are excellent insulators.
- **Metals and Alloys:** Metals have giant metallic lattices with delocalised electrons, making them malleable and good conductors. Alloys are mixtures of a metal with other elements (e.g., steel is iron + carbon) to disrupt the regular lattice, making them harder and stronger than pure metals.
- **Polymers:** Long-chain molecules. Thermosoftening polymers consist of individual, tangled polymer chains with weak intermolecular forces, so they melt when heated. Thermosetting polymers have strong covalent cross-links between chains, so they do not melt when heated.
- **Composites:** Consist of two materials: a matrix (binder) surrounding and binding together fragments or fibres of the other material (reinforcement). Examples include fibreglass (glass fibres in a polymer matrix) and reinforced concrete (steel bars in a concrete matrix).
### Concept 5: Life Cycle Assessments (LCAs) and Recycling
An LCA is carried out to assess the environmental impact of products in four stages:
1. Extracting and processing raw materials
2. Manufacturing and packaging
3. Use and operation during its lifetime
4. Disposal at the end of its useful life
While energy, water, and raw material usage can be easily quantified, evaluating pollutant effects is subjective. This subjectivity is a key limitation of LCAs, as different assessors may weight environmental impacts differently. Recycling metals, glass, and plastics reduces the need to extract finite raw materials, saves energy compared to primary production, and reduces waste sent to landfill.
## Mathematical/Scientific Relationships
**Percentage Yield Formula:**
$\text{Percentage Yield} = \frac{\text{Mass of product actually made}}{\text{Maximum theoretical mass of product}} \times 100$
*Use this to calculate the efficiency of product collection.*
**Atom Economy Formula:**
$\text{Atom Economy} = \frac{\text{Relative formula mass of desired product}}{\text{Sum of relative formula masses of all reactants}} \times 100$
*Use this to calculate the inherent efficiency of the reaction pathway.*
## Practical Applications
**Fertilisers (NPK):** Formulated fertilisers contain compounds of Nitrogen, Phosphorus, and Potassium to improve agricultural productivity. Ammonia is reacted with nitric acid to produce ammonium nitrate, a highly effective nitrogenous fertiliser.
**Podcast Audio Guide:**
Listen to the comprehensive audio guide covering the core concepts, common pitfalls, and exam techniques for this topic.
