Pure substances and mixtures — WJEC GCSE Study Guide
Exam Board: WJEC | Level: GCSE
Master the scientific definition of purity and the physical techniques used to separate mixtures. This topic is essential for understanding how chemists analyse, identify, and purify substances in the laboratory and industry.

## Overview
Welcome to the foundational topic of Pure Substances and Mixtures. This topic is absolutely crucial because it underpins much of the analytical chemistry you will study at GCSE. We encounter the word 'pure' constantly in everyday life—like 'pure' orange juice or 'pure' honey—but in chemistry, this term has a strict, specific meaning that examiners will test you on rigorously.
In this section, you will learn how to distinguish between elements, compounds, and mixtures, and how to define a pure substance scientifically. More importantly, you will discover how chemists separate complex mixtures into their constituent parts using physical processes like filtration, crystallisation, and distillation.

Finally, we will explore chromatography, a powerful analytical technique used to separate and identify substances. You will learn how to calculate Rf values and interpret chromatograms. This topic connects heavily to atomic structure and bonding, and forms the basis for more advanced analytical techniques you might encounter later.
## Key Concepts
### Concept 1: The Scientific Definition of 'Pure'
In everyday language, 'pure' means something has had nothing added to it, like pure milk. However, in chemistry, a **pure substance** is defined strictly as a single element or a single compound, not mixed with any other substance.
For example, pure water consists only of H₂O molecules. If you dissolve even a tiny amount of salt in it, it is no longer chemically pure—it is a mixture.
**Why does this matter?** Pure substances have highly specific, characteristic physical properties. Most notably, they melt and boil at specific, fixed temperatures. Water melts at exactly 0°C and boils at exactly 100°C (at standard pressure).
If a substance is impure (a mixture), it will melt and boil over a *range* of temperatures. Furthermore, impurities generally lower the melting point and raise the boiling point of a substance. Examiners frequently ask you to interpret melting point data to determine purity.
**Example**: A student tests an unknown solid. It begins to melt at 134°C and finishes melting at 141°C. Because it melts over a range of temperatures rather than at a sharp, specific point, the student can conclude the substance is impure (a mixture).
### Concept 2: Formulations
A **formulation** is a complex mixture that has been designed as a useful product. In a formulation, every chemical has a specific purpose, and the components are mixed in carefully measured, exact quantities to ensure the product has the required properties.
Examples of formulations include:
- **Medicines**: The active drug is mixed with colourants, sweeteners, and smooth coatings to make it easy to swallow and digest.
- **Paints**: Contain pigments (for colour), binders (to attach to the surface), and solvents (to thin the paint so it spreads).
- **Other examples**: Cleaning agents, fuels, alloys, fertilisers, and cosmetics.
### Concept 3: Separation Techniques
Mixtures contain substances that are not chemically bonded together. Because they are not bonded, they can be separated using physical processes that do not involve chemical reactions.
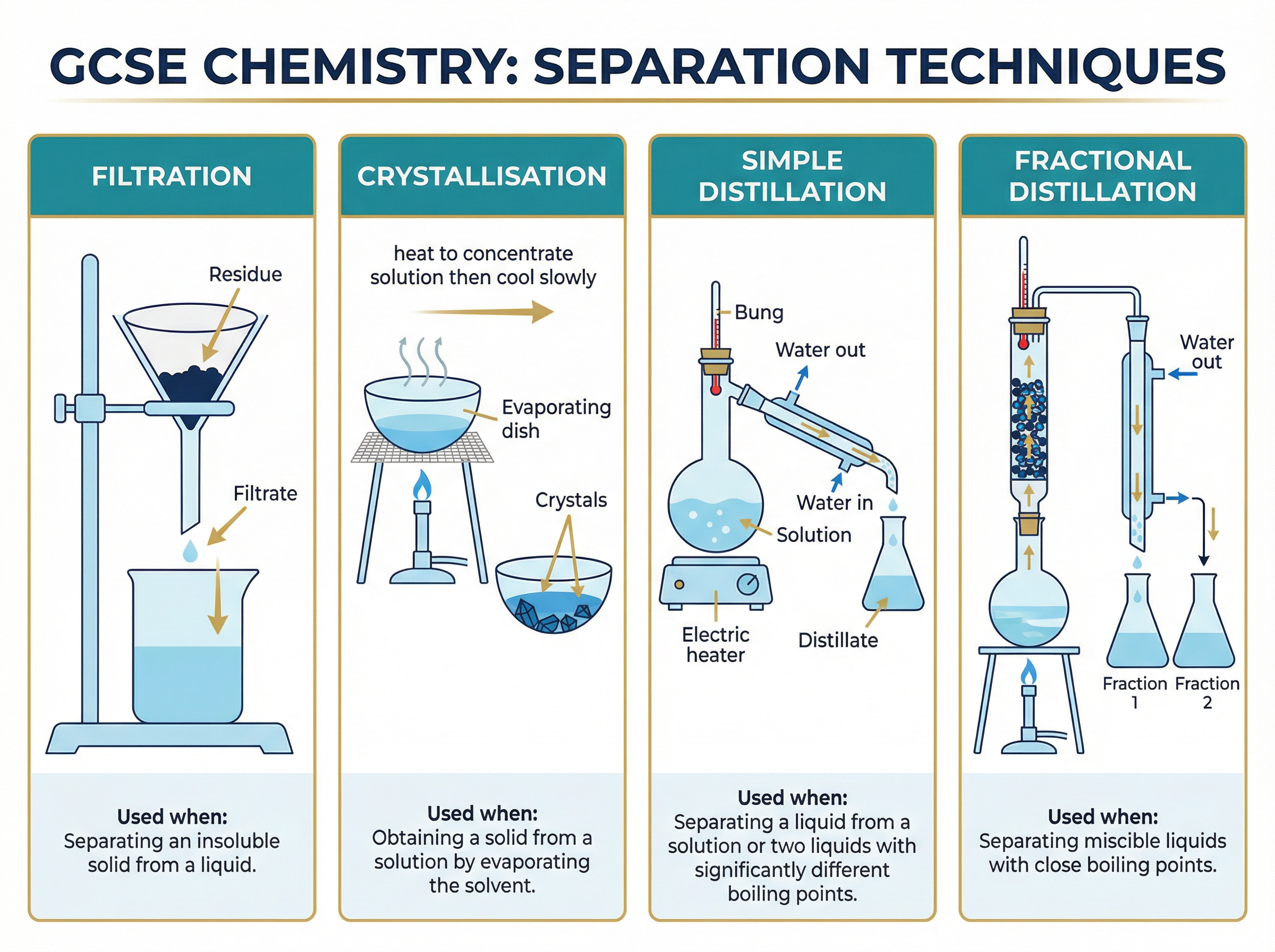
1. **Filtration**: Used to separate an insoluble solid from a liquid. The mixture is poured through filter paper. The liquid (filtrate) passes through the tiny pores, while the solid (residue) is trapped.
2. **Crystallisation**: Used to obtain a soluble solid from a solution. The solution is heated gently to evaporate some solvent, making it more concentrated. It is then left to cool. Because solids are less soluble at lower temperatures, crystals form, which can be filtered and dried.
3. **Simple Distillation**: Used to separate a liquid from a solution (e.g., pure water from seawater). The solution is heated. The solvent boils, turns to gas, and travels into a condenser where it cools and condenses back into a liquid (the distillate). The dissolved solute remains in the flask.
4. **Fractional Distillation**: Used to separate a mixture of miscible liquids with different (but often similar) boiling points. The mixture is heated in a flask fitted with a fractionating column (often filled with glass beads). The column is hotter at the bottom and cooler at the top. The liquid with the lowest boiling point evaporates first, reaches the top of the column, condenses, and is collected.
### Concept 4: Chromatography
Paper chromatography is an analytical technique used to separate and identify mixtures, particularly coloured compounds like inks or dyes.
It relies on two phases:
- **The Stationary Phase**: The part that does not move. In paper chromatography, this is the absorbent chromatography paper.
- **The Mobile Phase**: The part that moves. This is the solvent (like water or ethanol) that moves up the paper by capillary action.
**How it works**: A spot of the mixture is placed on a pencil baseline drawn on the paper. The paper is placed in a solvent, ensuring the solvent level is *below* the pencil line (so the spots don't just wash away into the beaker). As the mobile phase moves up the stationary phase, it carries the substances in the mixture with it.
Substances separate because they have different affinities for the two phases. A substance that is highly soluble in the mobile phase but weakly attracted to the stationary phase will travel further up the paper. A pure substance will produce a single spot, while a mixture will separate into multiple spots.
## Mathematical/Scientific Relationships
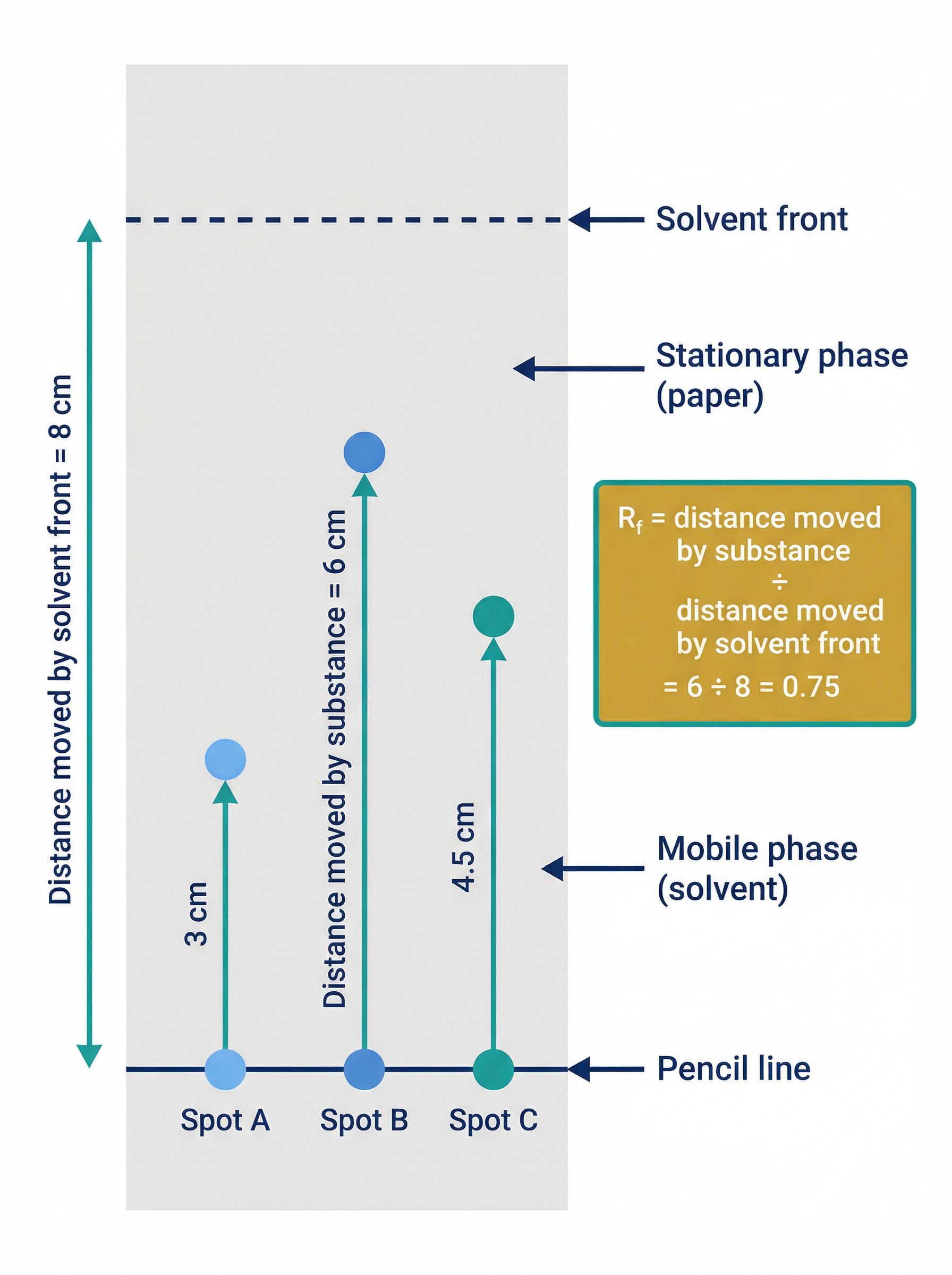
### Calculating the Retention Factor (Rf)
In chromatography, we quantify how far a substance has moved using the Retention Factor (Rf) value. This allows chemists to identify unknown substances by comparing their Rf values with a database of known values (provided the same solvent and temperature are used).
**Formula**:
Rf = (Distance moved by substance) ÷ (Distance moved by solvent front)
- **Distance moved by substance**: Measured from the pencil baseline to the centre of the spot.
- **Distance moved by solvent front**: Measured from the pencil baseline to the maximum height the solvent reached.
*Note: Rf values have no units and are always less than 1 (because the substance can never travel further than the solvent).*
## Practical Applications
**Required Practical: Chromatography**
Students must investigate how paper chromatography can be used to separate and tell the difference between coloured substances.
- **Apparatus**: Chromatography paper, capillary tube, beaker, solvent, pencil, ruler.
- **Method**: Draw a pencil line 2cm from the bottom of the paper. Use a capillary tube to place small spots of known and unknown dyes on the line. Suspend the paper in a beaker containing 1cm of solvent. Wait for the solvent to travel near the top. Remove, mark the solvent front, dry, and calculate Rf values.
- **Common Errors**: Drawing the baseline in ink (the ink will dissolve and interfere with the results). Having the solvent level above the baseline (the spots will wash off into the beaker).