Rate of chemical change and dynamic equilibrium — WJEC GCSE Study Guide
Exam Board: WJEC | Level: GCSE
Master the speed of chemical reactions and the delicate balance of dynamic equilibrium. This topic is essential for understanding how industrial processes are optimised to save time and money, and it's a guaranteed high-mark area in your GCSE Chemistry exams.
## Overview

Welcome to Rate of Chemical Change and Dynamic Equilibrium. This topic is a cornerstone of GCSE Chemistry because it connects theoretical particle models with real-world industrial applications. Understanding *how fast* a reaction happens, and *how far* it goes before stopping, is critical for chemical engineers trying to maximise yield and minimise costs.
In your exams, this topic frequently appears as extended response questions (6-markers) requiring you to explain observations using collision theory. It also features heavily in data analysis questions where you must interpret graphs and calculate rates from tangents. You will need to apply your knowledge of the particle model from earlier topics, making this a highly synoptic area of the specification.
Listen to the full audio guide for this topic below:

## Key Concepts
### Concept 1: Measuring the Rate of Reaction
The rate of a chemical reaction tells us how quickly reactants are converted into products. Examiners expect you to know three main practical methods for measuring this:
1. **Gas Collection**: If a reaction produces a gas, you can collect it using a gas syringe or an inverted measuring cylinder over water. By recording the volume of gas produced at regular time intervals, you can plot a graph of volume against time. The gradient of this graph gives the rate of reaction.
2. **Loss of Mass**: If a heavy gas (like carbon dioxide) is produced and escapes, the total mass of the reaction flask will decrease. Placing the flask on a balance and recording the mass at regular intervals allows you to plot a mass-loss graph. This is often the most accurate method as balances are highly precise.
3. **Precipitation (Disappearing Cross)**: For reactions that produce a solid precipitate (making the solution cloudy), you can time how long it takes for a mark (like a cross on a piece of paper) placed under the flask to disappear. This is a common required practical involving sodium thiosulfate and hydrochloric acid.
**Example**: In the reaction between marble chips (calcium carbonate) and hydrochloric acid, measuring the volume of carbon dioxide produced every 10 seconds allows you to track the decreasing rate as the reactants are used up.
### Concept 2: Collision Theory and Activation Energy
For a chemical reaction to occur, reactant particles must collide with each other. However, not all collisions lead to a reaction. A collision is only successful if the particles collide with sufficient energy. This minimum amount of energy required to start a reaction is called the **activation energy**.
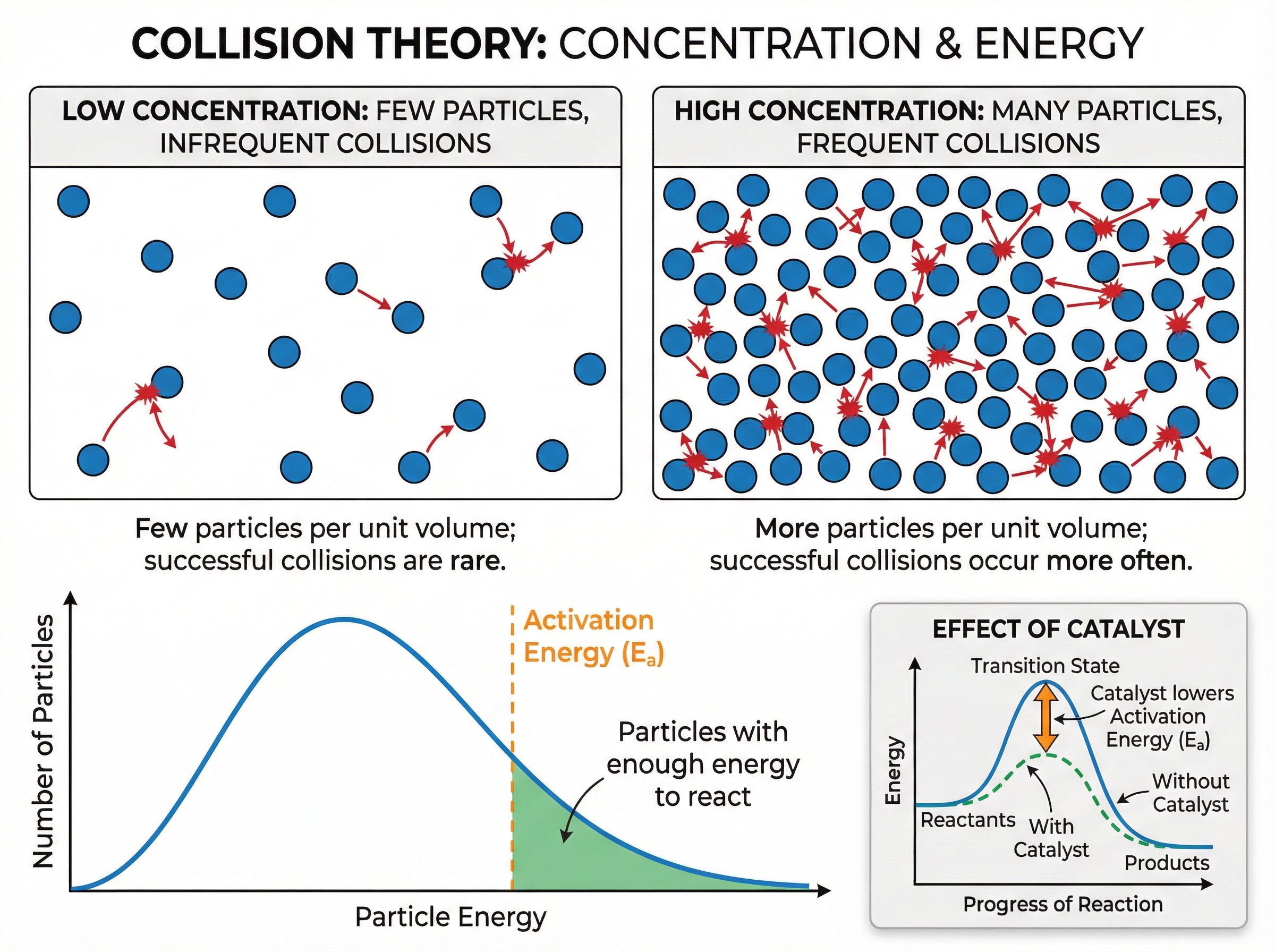
The rate of a reaction depends on two crucial factors:
1. The **frequency of collisions** (how often particles collide per unit of time).
2. The **energy of the collisions** (what proportion of particles have energy greater than or equal to the activation energy).
When explaining rate changes in exams, you *must* refer to these two points to secure full marks.
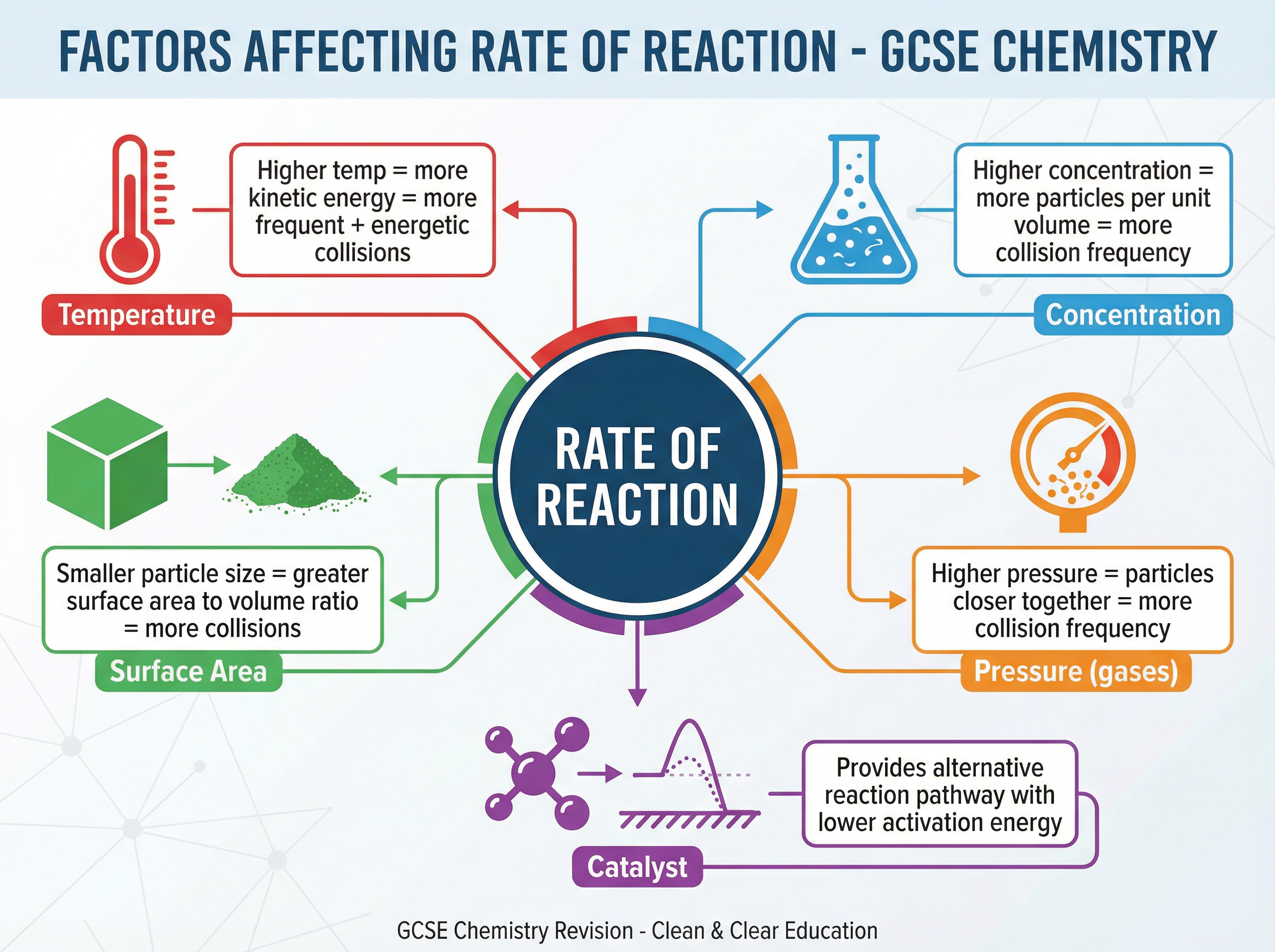
### Concept 3: Factors Affecting Rate of Reaction
Five main factors affect the rate of a chemical reaction:
* **Temperature**: Increasing temperature increases the kinetic energy of particles. They move faster, leading to more frequent collisions. Crucially, a much higher proportion of particles now have energy greater than the activation energy. This dual effect makes temperature a very powerful factor.
* **Concentration (of solutions)**: Increasing concentration means there are more reactant particles in the same volume. This increases the frequency of collisions.
* **Pressure (of gases)**: Increasing pressure pushes gas particles closer together, meaning more particles in a given volume. This increases the frequency of collisions.
* **Surface Area (of solids)**: Breaking a solid lump into smaller pieces or powder increases its surface area to volume ratio. More particles are exposed on the surface and available to react, increasing the frequency of collisions.
* **Catalysts**: A catalyst speeds up a reaction without being used up itself. It works by providing an alternative reaction pathway that has a lower activation energy. Therefore, a greater proportion of collisions are successful.
### Concept 4: Reversible Reactions and Dynamic Equilibrium
Some chemical reactions are reversible, meaning the products can react together to reform the original reactants. This is represented by a double-headed arrow (⇌).
If a reversible reaction occurs in a **closed system** (where no reactants or products can escape), it will eventually reach a state of **dynamic equilibrium**.
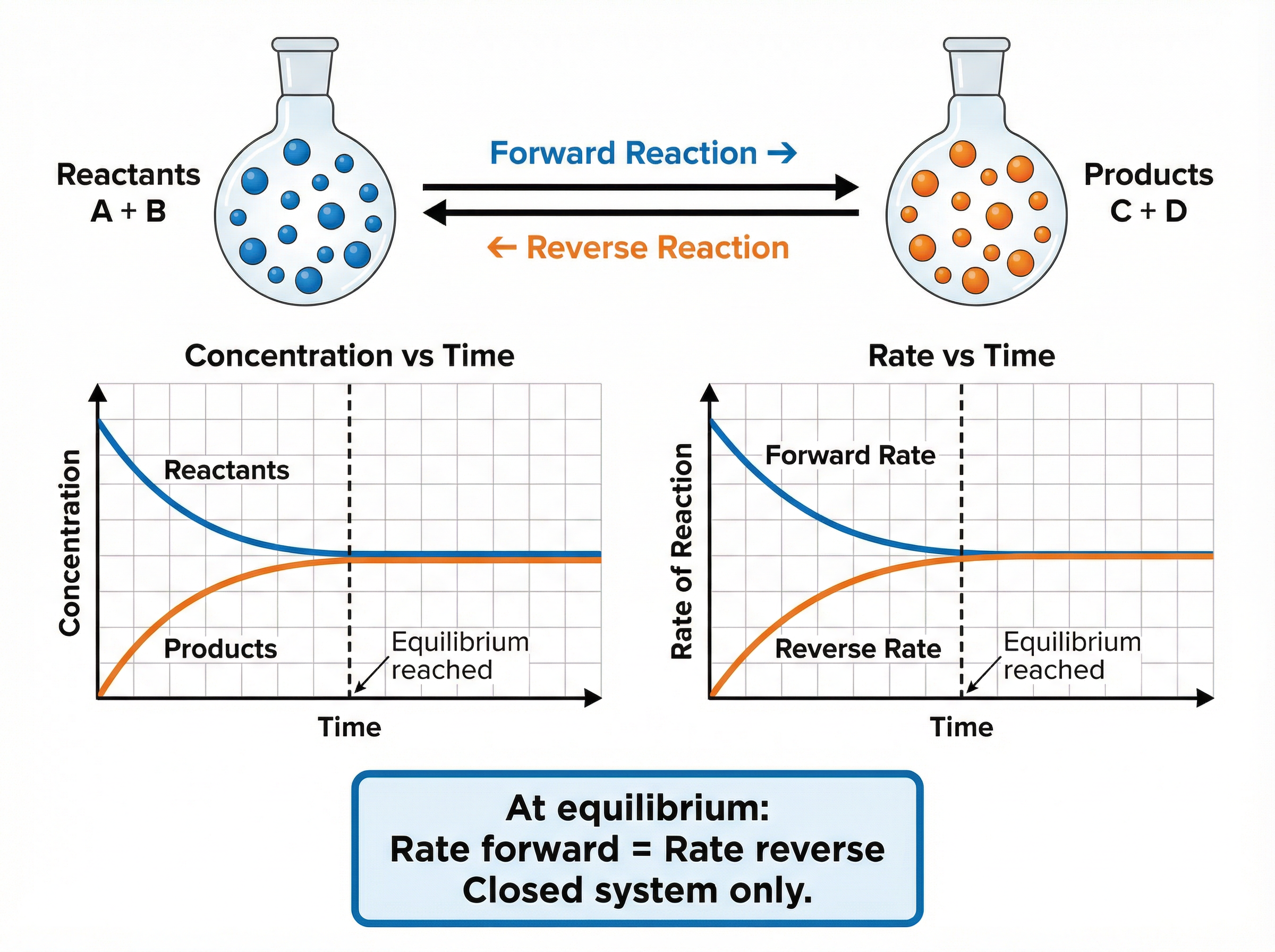
At dynamic equilibrium:
* The forward and reverse reactions are occurring at the **exact same rate**.
* The concentrations of the reactants and products remain **constant** (though not necessarily equal).
It is vital to remember that the reactions have *not* stopped; they are simply balanced.
### Concept 5: Le Chatelier's Principle
When a change in conditions (temperature, pressure, or concentration) is introduced to a system at equilibrium, the system will respond to counteract that change. This is known as Le Chatelier's Principle.
* **Changing Concentration**: If you increase the concentration of a reactant, the system tries to decrease it by making more product (equilibrium shifts to the right). If you decrease a product, the system makes more to replace it.
* **Changing Temperature**: If you increase the temperature, the system tries to cool down by favouring the endothermic (heat-absorbing) direction. If you decrease the temperature, it favours the exothermic (heat-releasing) direction.
* **Changing Pressure**: If you increase the pressure, the system tries to reduce it by shifting to the side of the balanced equation with the **fewest moles of gas**.
## Mathematical/Scientific Relationships
**Rate of Reaction Formula:**
Rate of reaction = Amount of reactant used / Time
OR
Rate of reaction = Amount of product formed / Time
*Must memorise*
**Calculating Rate from a Graph:**
To find the rate at a specific time on a curved graph, you must draw a tangent to the curve at that time point and calculate its gradient.
Gradient = Change in y / Change in x
## Practical Applications
**Required Practical: Investigating the effect of concentration on rate of reaction**
*Method 1: Measuring volume of gas*
1. Support a gas syringe with a stand, boss and clamp.
2. Add 50 cm³ of dilute hydrochloric acid to a conical flask.
3. Add a 3 cm piece of magnesium ribbon to the flask and immediately connect the gas syringe and start a stopwatch.
4. Record the volume of gas produced every 10 seconds until the reaction stops.
5. Repeat with different concentrations of hydrochloric acid.
*Method 2: Disappearing Cross*
1. Measure 10 cm³ of sodium thiosulfate solution into a conical flask.
2. Place the flask on a printed black cross.
3. Add 10 cm³ of dilute hydrochloric acid, swirl the flask and start a stopwatch.
4. Look down through the flask and stop the stopwatch when the cross can no longer be seen.
5. Repeat with different concentrations of sodium thiosulfate.
**Industrial Application: The Haber Process**
The Haber process produces ammonia (NH₃) from nitrogen and hydrogen. It is a reversible, exothermic reaction. Engineers must carefully choose the temperature and pressure to maximise the yield of ammonia while maintaining a fast enough rate of reaction and keeping costs down. They use a moderately high temperature (450°C), a high pressure (200 atm), and an iron catalyst.