Reactivity series and extraction of metals — WJEC GCSE Study Guide
Exam Board: WJEC | Level: GCSE
Master the reactivity series and the extraction of metals! This topic connects the theoretical ranking of metals to the real-world industrial processes used to extract them, forming a core part of your chemistry exams.
## Overview
Welcome to the Reactivity Series and Extraction of Metals! This topic is a cornerstone of GCSE Chemistry because it bridges the gap between theoretical principles and massive industrial applications. By understanding how metals are ranked by their reactivity, you can predict how they will behave in chemical reactions and, crucially, determine the method required to extract them from their ores.
Examiners love this topic because it synoptically links displacement reactions, redox chemistry (oxidation and reduction), and electrolysis. You can expect a mix of short factual recall questions (like identifying oxidation in an equation) and longer, 6-mark extended response questions requiring you to evaluate extraction methods or explain the processes in a blast furnace or electrolysis cell. Let's break it down so you can secure those top marks.
## Key Concepts
### Concept 1: The Reactivity Series and Displacement
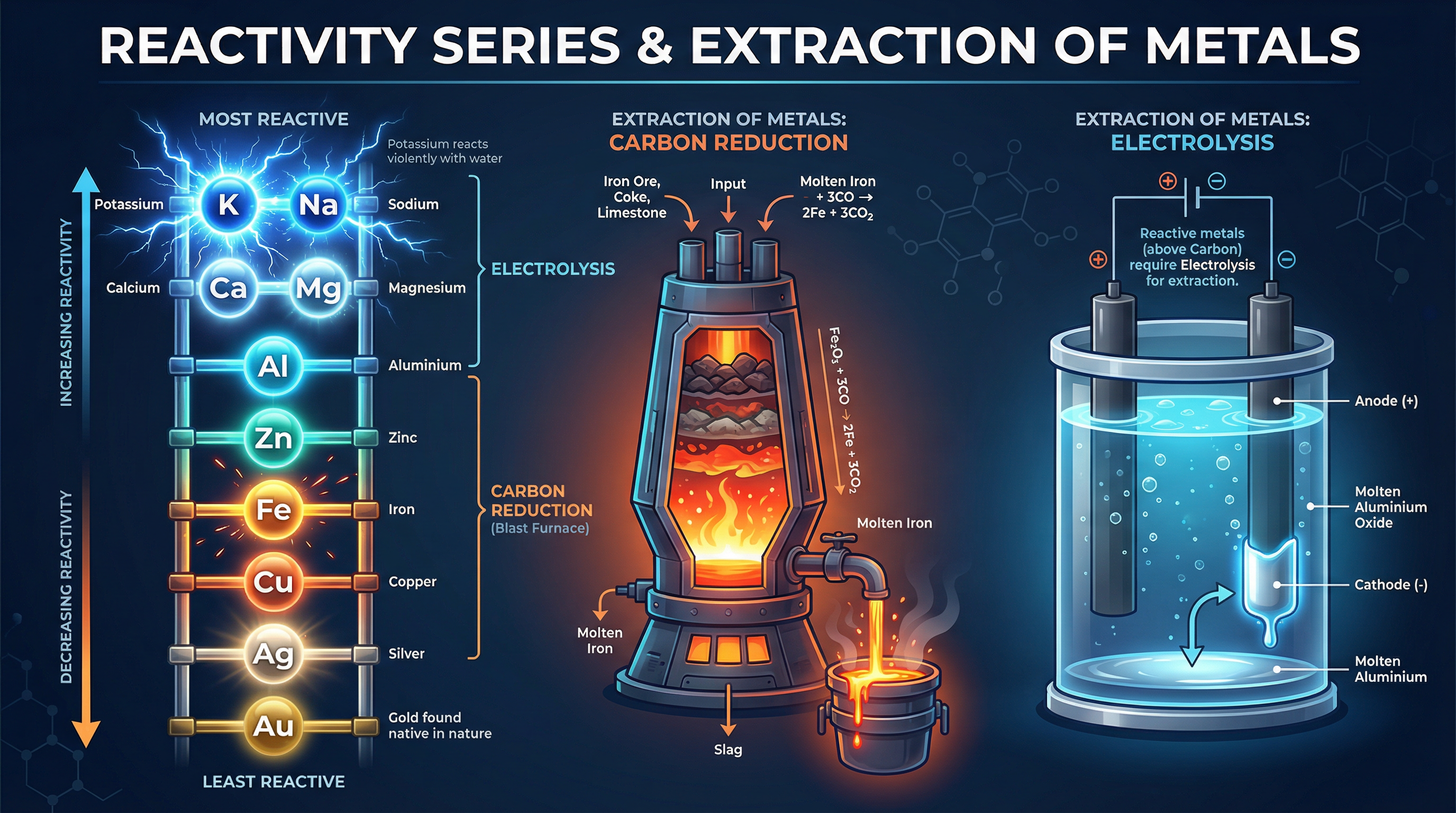
The reactivity series is a ranking of metals based on how easily they lose electrons to form positive ions. The easier a metal loses electrons, the more reactive it is.
We establish this order experimentally using **displacement reactions**. A more reactive metal will displace a less reactive metal from a solution of its salt. For example, if you place zinc in copper(II) sulfate solution, the zinc displaces the copper because zinc is higher in the reactivity series. The blue colour of the solution fades as zinc sulfate forms, and a brown solid (copper) is deposited.
**Example**: $\text{Zn(s)} + \text{CuSO}_4\text{(aq)} \rightarrow \text{ZnSO}_4\text{(aq)} + \text{Cu(s)}$
### Concept 2: Oxidation and Reduction (Redox)
In chemistry, oxidation and reduction always occur together in what we call redox reactions. You must be able to define these in two ways:
1. **In terms of oxygen**: Oxidation is the gain of oxygen; reduction is the loss of oxygen.
2. **In terms of electrons**: Oxidation Is Loss of electrons; Reduction Is Gain of electrons (remember OIL RIG!).
In the displacement reaction above, zinc atoms lose two electrons to become $\text{Zn}^{2+}$ ions (oxidation). Copper(II) ions, $\text{Cu}^{2+}$, gain two electrons to become copper atoms (reduction).
### Concept 3: Extraction of Metals
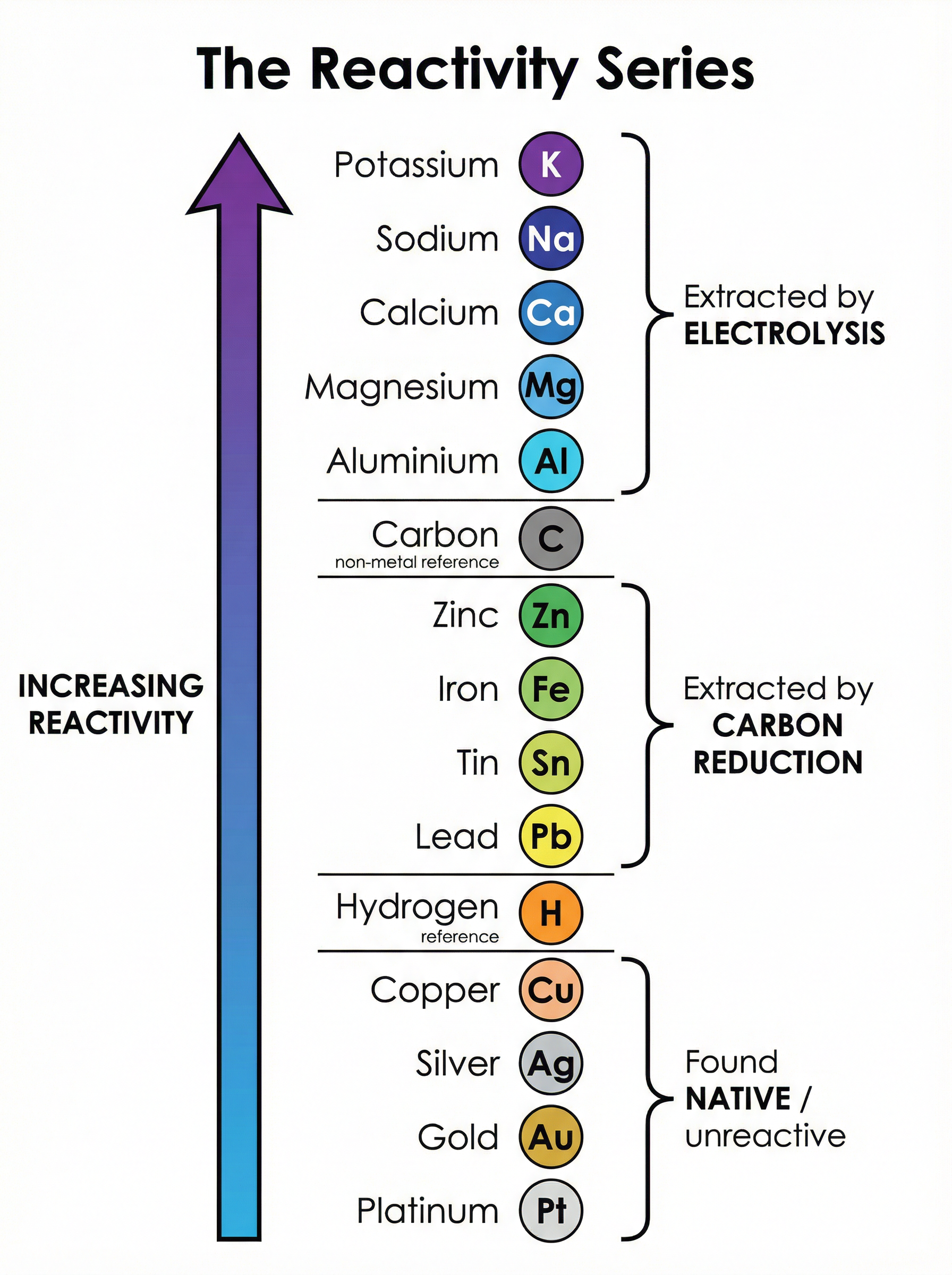
Most metals are found in the Earth's crust combined with other elements, often as metal oxides (ores). The method chosen to extract a metal depends entirely on its position in the reactivity series.
* **Metals less reactive than carbon** (e.g., zinc, iron, lead) are extracted by heating with carbon (carbon reduction). Carbon displaces the metal from its oxide.
* **Metals more reactive than carbon** (e.g., aluminium, magnesium) cannot be reduced by carbon. They must be extracted using electrolysis, which requires massive amounts of electricity and is therefore very expensive.
* **Unreactive metals** (e.g., gold, platinum) are found native (as the uncombined element).
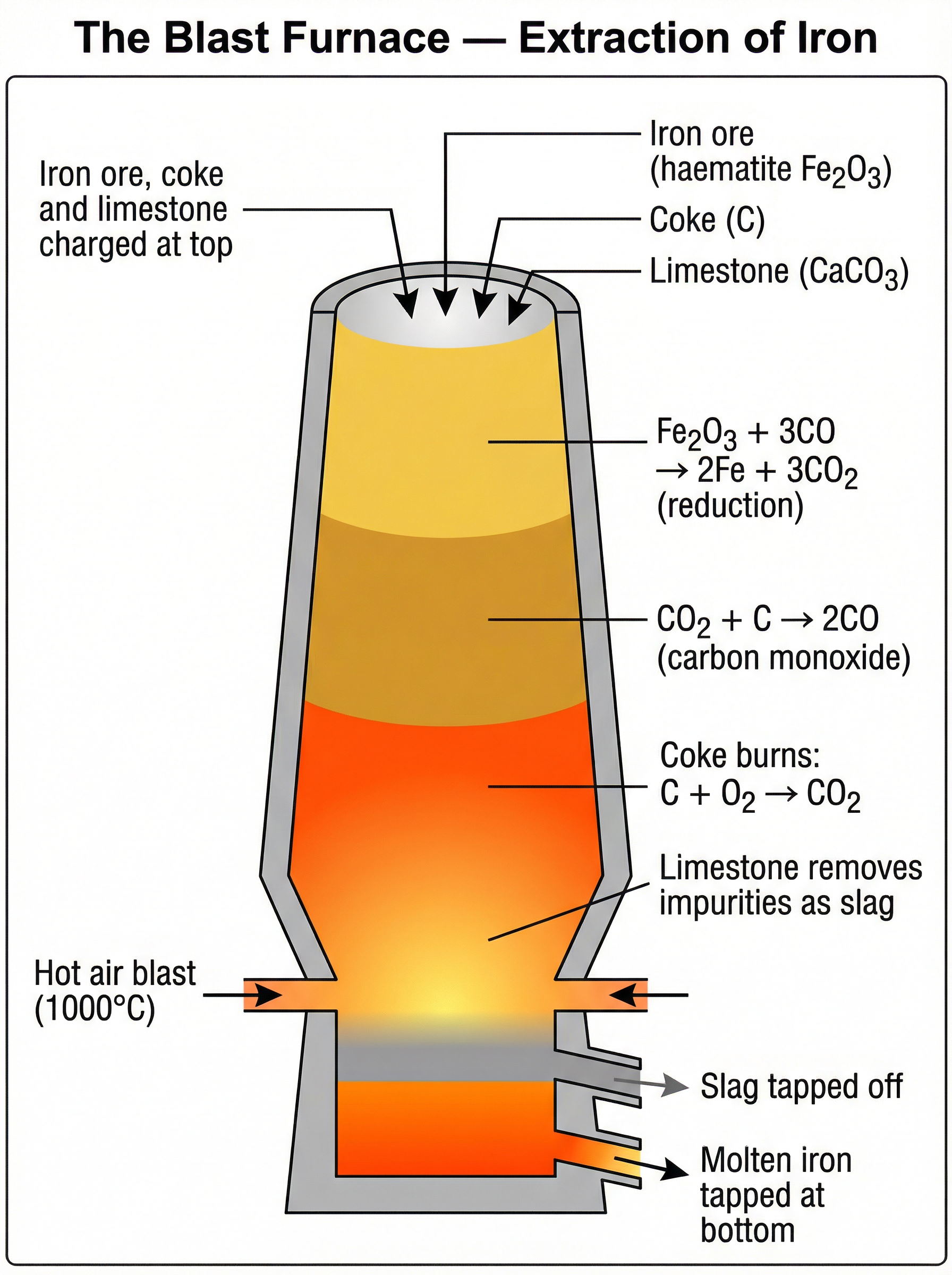
### Concept 4: Electrolysis of Molten and Aqueous Compounds
Electrolysis is the breaking down of an ionic compound using electricity. The compound must be molten or dissolved in water (aqueous) so that the ions are free to move.
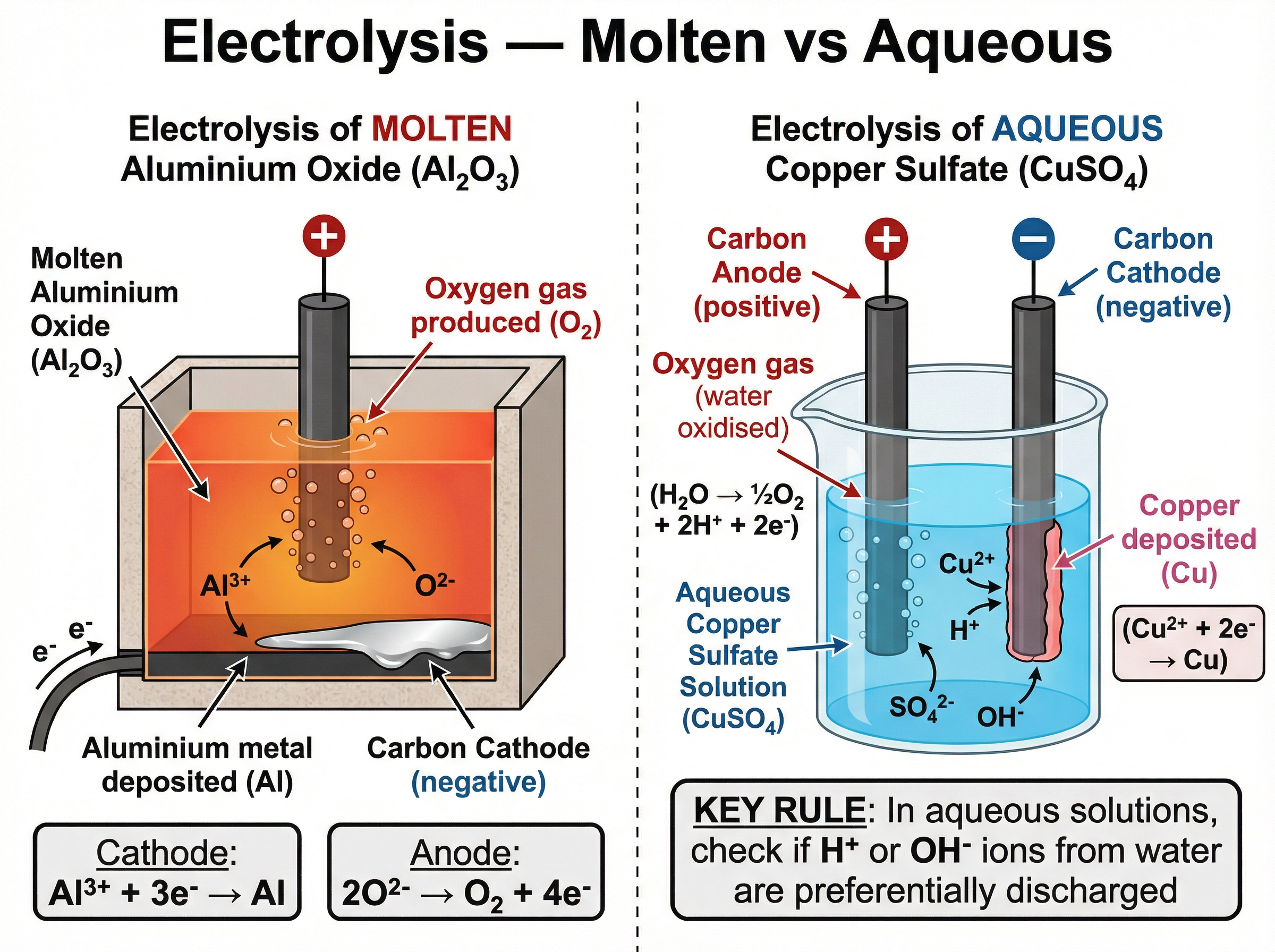
**Molten Electrolysis (e.g., Aluminium Oxide)**:
Aluminium is extracted from purified bauxite (aluminium oxide, $\text{Al}_2\text{O}_3$) dissolved in molten cryolite (to lower the melting point). At the cathode (negative electrode), $\text{Al}^{3+}$ ions gain electrons to form liquid aluminium. At the anode (positive electrode), $\text{O}^{2-}$ ions lose electrons to form oxygen gas, which then reacts with the carbon anodes, meaning they must be continually replaced.
**Aqueous Electrolysis (e.g., Sodium Chloride)**:
In aqueous solutions, water partially ionises into $\text{H}^+$ and $\text{OH}^-$ ions. At the cathode, the less reactive element is discharged. If the metal is more reactive than hydrogen (like sodium), hydrogen gas is produced. At the anode, if halide ions ($\text{Cl}^-$, $\text{Br}^-$, $\text{I}^-$) are present, the halogen is produced; otherwise, oxygen is produced from the $\text{OH}^-$ ions.
## Podcast Revision
Listen to this 10-minute audio summary covering the core concepts and exam tips:

## Mathematical/Scientific Relationships
* **Half Equations**: Show the transfer of electrons. Ensure charges balance on both sides.
* *Reduction (Cathode)*: $\text{Al}^{3+} + 3\text{e}^- \rightarrow \text{Al}$
* *Oxidation (Anode)*: $2\text{O}^{2-} \rightarrow \text{O}_2 + 4\text{e}^-$
* **Ionic Equations**: Show only the species that change state or charge. Spectator ions are omitted.
* *Full equation*: $\text{Mg(s)} + \text{CuCl}_2\text{(aq)} \rightarrow \text{MgCl}_2\text{(aq)} + \text{Cu(s)}$
* *Ionic equation*: $\text{Mg(s)} + \text{Cu}^{2+}\text{(aq)} \rightarrow \text{Mg}^{2+}\text{(aq)} + \text{Cu(s)}$
## Practical Applications
* **Phytoextraction and Bioleaching**: Traditional mining involves digging up large amounts of rock, which has significant environmental impacts. Alternative methods are being developed to extract metals from low-grade ores. Phytoextraction uses plants to absorb metal compounds; the plants are then harvested and burned to produce an ash rich in metal compounds. Bioleaching uses bacteria to produce leachate solutions that contain metal compounds. Both methods are more sustainable but much slower than traditional mining.