The Earth and its atmosphere — WJEC GCSE Study Guide
Exam Board: WJEC | Level: GCSE
Master the evolution of the Earth's atmosphere, the greenhouse effect, and the impact of human activity on our climate. This topic is heavily tested on long-answer questions where precise scientific terminology and logical explanations earn top marks.
## Overview
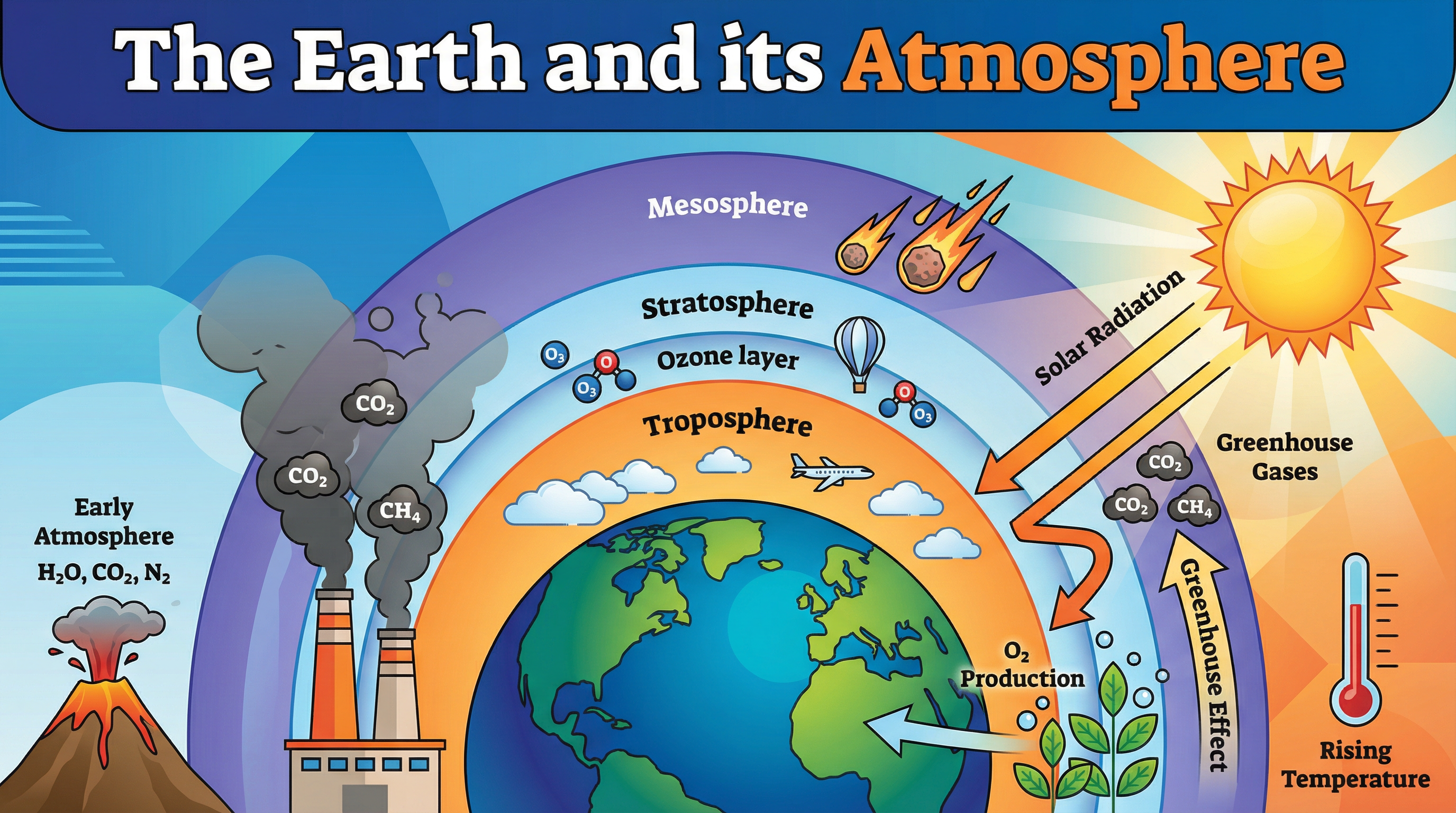
The Earth's atmosphere is a dynamic system that has evolved dramatically over 4.6 billion years. Understanding this topic is crucial for GCSE Chemistry because it links fundamental chemical principles—such as combustion, reversible reactions, and equilibrium—to real-world environmental challenges like climate change and air pollution.
Examiners frequently test this area using data analysis questions, asking you to interpret graphs of CO2 levels or temperature changes. You will also encounter 6-mark extended response questions requiring you to explain the greenhouse effect or evaluate the evidence for climate change. This topic synoptically links to quantitative chemistry (calculating gas volumes) and organic chemistry (combustion of fuels). Mastering the specific terminology—such as 'short-wave radiation' and 'carbon footprint'—is essential for accessing the highest marking bands.
## Key Concepts
### Concept 1: Evolution of the Early Atmosphere
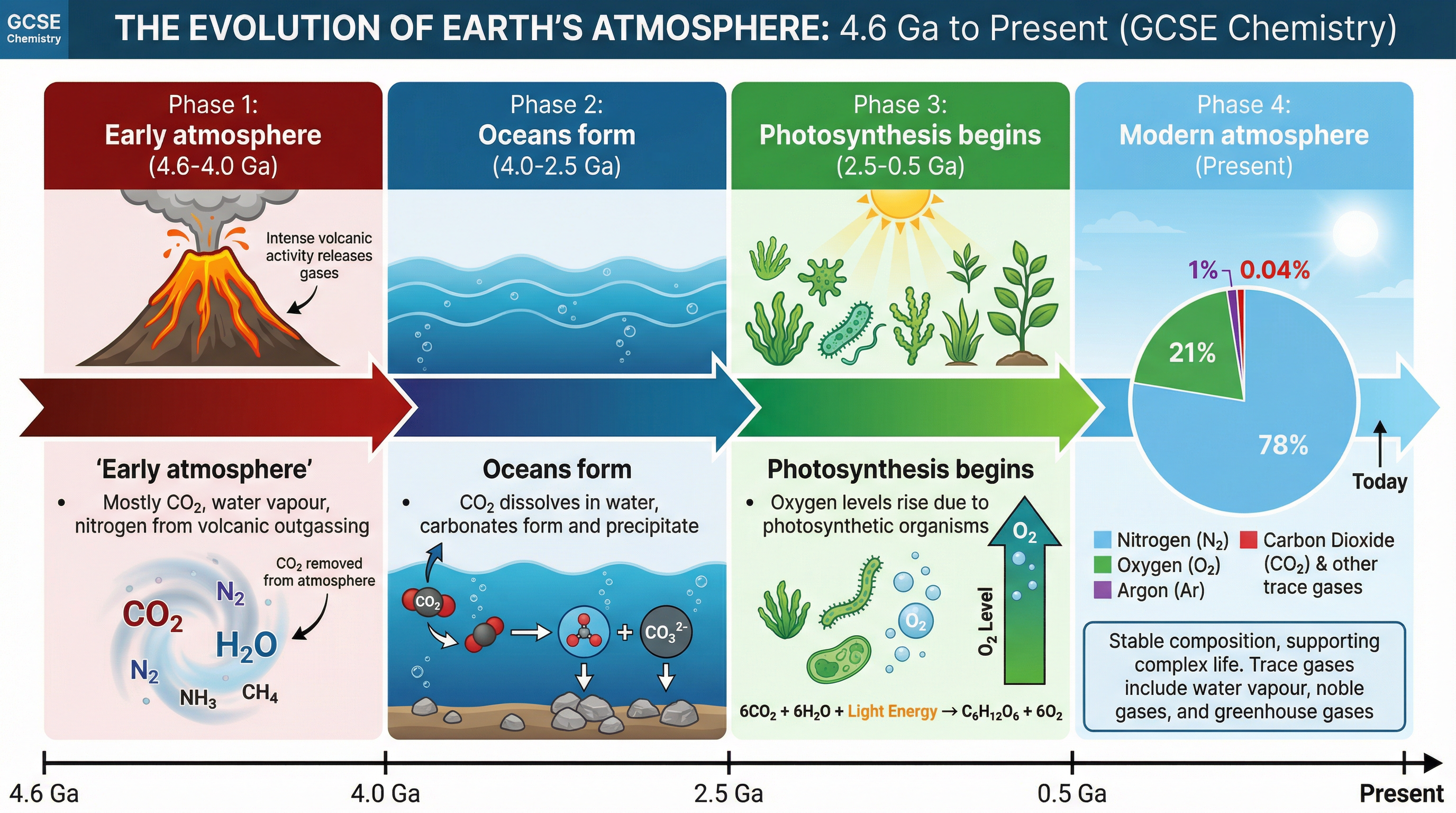
For the first billion years of Earth's history, intense volcanic activity released gases that formed the early atmosphere. This atmosphere was primarily carbon dioxide (CO2), with water vapour (H2O), nitrogen (N2), and trace amounts of methane (CH4) and ammonia (NH3). There was virtually no oxygen. This is similar to the modern atmospheres of Mars and Venus.
As the Earth cooled, the water vapour condensed to form the oceans. This was a pivotal moment because carbon dioxide dissolved in the water, forming carbonate precipitates that eventually became sedimentary rocks (like limestone). This process significantly reduced the amount of CO2 in the atmosphere.
### Concept 2: The Rise of Oxygen
Around 2.7 billion years ago, algae and simple plants evolved and began to photosynthesise. Photosynthesis uses light energy to convert carbon dioxide and water into glucose and oxygen.
Over the next billion years, plants evolved and the percentage of oxygen gradually increased to a level that enabled animals to evolve. Simultaneously, the percentage of carbon dioxide continued to decrease as it was locked up in sedimentary rocks and fossil fuels (coal, crude oil, and natural gas) formed from the remains of dead plants and marine organisms.
### Concept 3: The Present-Day Atmosphere
For approximately 200 million years, the proportions of different gases in the atmosphere have been much the same as they are today:
- **Nitrogen (N2):** ~78%
- **Oxygen (O2):** ~21%
- **Argon (Ar):** ~1%
- **Carbon Dioxide (CO2):** ~0.04%
- Plus small proportions of other noble gases and varying amounts of water vapour.
### Concept 4: The Greenhouse Effect
Greenhouse gases (such as water vapour, carbon dioxide, and methane) maintain temperatures on Earth high enough to support life. The mechanism is a common exam question:
1. **Short-wave radiation** (ultraviolet and visible light) from the Sun passes through the atmosphere and is absorbed by the Earth's surface.
2. The Earth's surface cools by emitting **long-wave infrared radiation** (thermal energy).
3. Greenhouse gases absorb this long-wave radiation.
4. The gases re-emit the radiation in all directions, including back towards the Earth, trapping heat in the atmosphere.
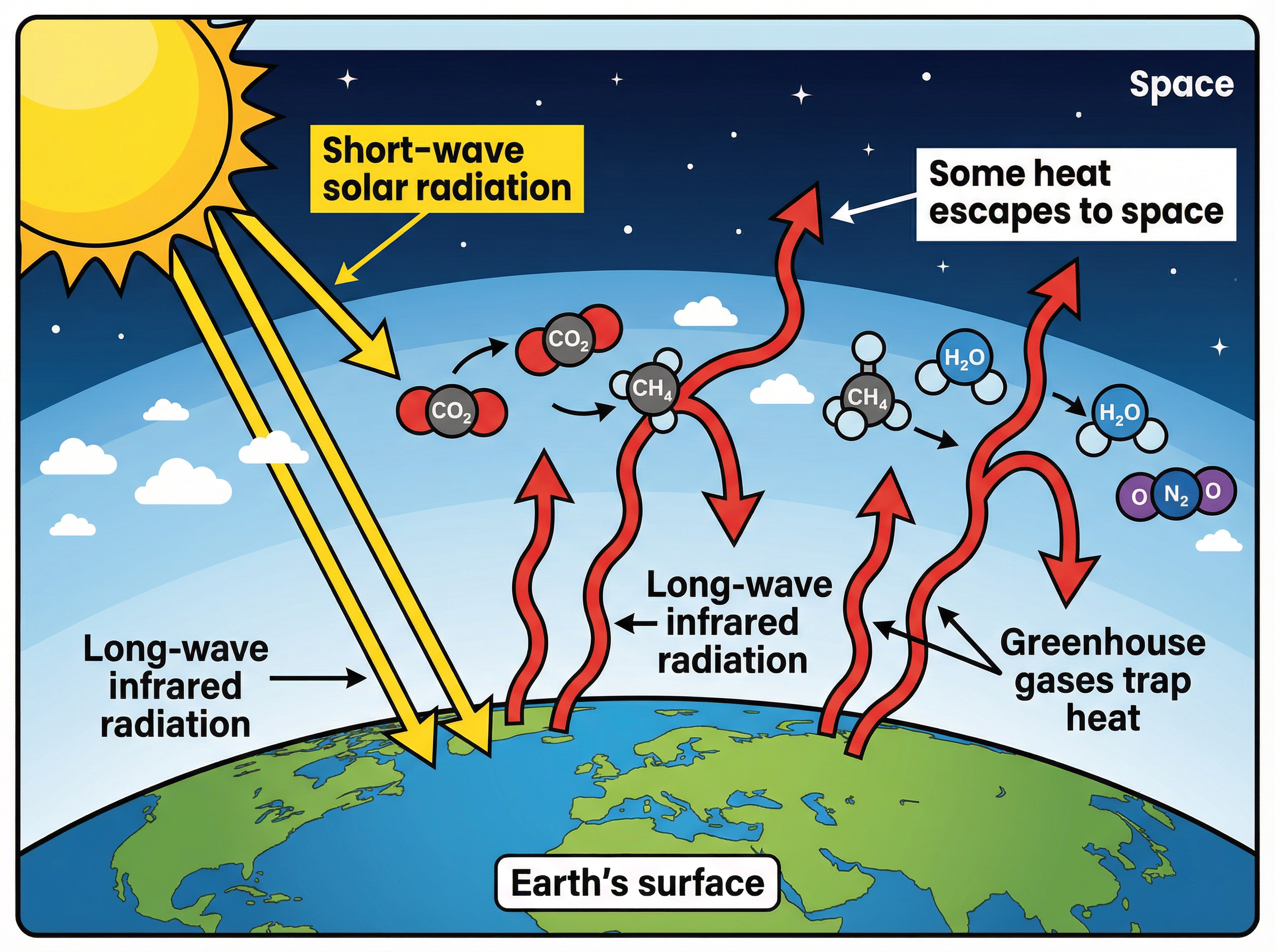
**Crucial Distinction:** The greenhouse effect is a natural and necessary process. However, human activities (like burning fossil fuels and deforestation) are increasing the levels of CO2 and CH4, leading to an *enhanced* greenhouse effect, which causes global warming.
### Concept 5: Atmospheric Pollutants
The combustion of fuels is a major source of atmospheric pollutants. Most fuels contain carbon and/or hydrogen, and some contain sulfur.
- **Carbon Monoxide (CO):** Formed by incomplete combustion. It is a toxic, colourless, and odourless gas that binds irreversibly to haemoglobin in red blood cells, preventing oxygen transport.
- **Sulfur Dioxide (SO2):** Formed when sulfur impurities in fossil fuels burn. It causes respiratory problems and dissolves in rain to form acid rain, which damages buildings and aquatic ecosystems.
- **Nitrogen Oxides (NOx):** Formed when nitrogen and oxygen react at the high temperatures inside car engines. They cause respiratory issues and contribute to acid rain and smog.
- **Particulates (Soot):** Solid carbon particles from incomplete combustion. They cause global dimming (reducing sunlight reaching the Earth) and respiratory health problems.

## Mathematical/Scientific Relationships
**Photosynthesis Equation:**
- Word: Carbon dioxide + Water → Glucose + Oxygen
- Symbol: 6CO2 + 6H2O → C6H12O6 + 6O2
*(Must memorise)*
**Complete Combustion of a Hydrocarbon (e.g., Methane):**
- CH4 + 2O2 → CO2 + 2H2O
*(Must memorise how to balance these)*
**Incomplete Combustion:**
- Produces Carbon Monoxide (CO) or Carbon (C, soot) alongside water.
- Example: 2CH4 + 3O2 → 2CO + 4H2O
## Practical Applications
Listen to the comprehensive audio guide for this topic to reinforce your learning:

Understanding atmospheric chemistry is vital for developing technologies to mitigate climate change, such as Carbon Capture and Storage (CCS) and designing catalytic converters for cars to reduce NOx emissions.