The Periodic Table and properties of elements — WJEC GCSE Study Guide
Exam Board: WJEC | Level: GCSE
Master the Periodic Table and the properties of elements with this comprehensive guide. Discover the fundamental trends in reactivity for Groups 1 and 7, learn the essential flame test colours, and understand why transition metals are crucial catalysts, all while applying examiner-approved techniques.
## Overview
This topic is the beating heart of GCSE Chemistry. The Periodic Table is not just a list of elements; it is a master key that unlocks the behaviour of the entire material world. By understanding how elements are arranged according to their atomic number and electronic structure, you can predict how they will react, what compounds they will form, and what physical properties they will exhibit. Examiners love this topic because it tests your ability to link microscopic structure (electron shells) to macroscopic observations (fizzing, colour changes, and flame colours).
In your exam, you will frequently encounter questions requiring you to explain reactivity trends in Group 1 and Group 7, deduce the products of displacement reactions, and recall specific qualitative tests like flame colours. This topic also connects heavily to atomic structure, ionic bonding, and rates of reaction. Mastering these concepts will provide a solid foundation for securing top marks.
Listen to the accompanying podcast for a deep dive into these concepts:

## Key Concepts
### Concept 1: Group 1 - The Alkali Metals
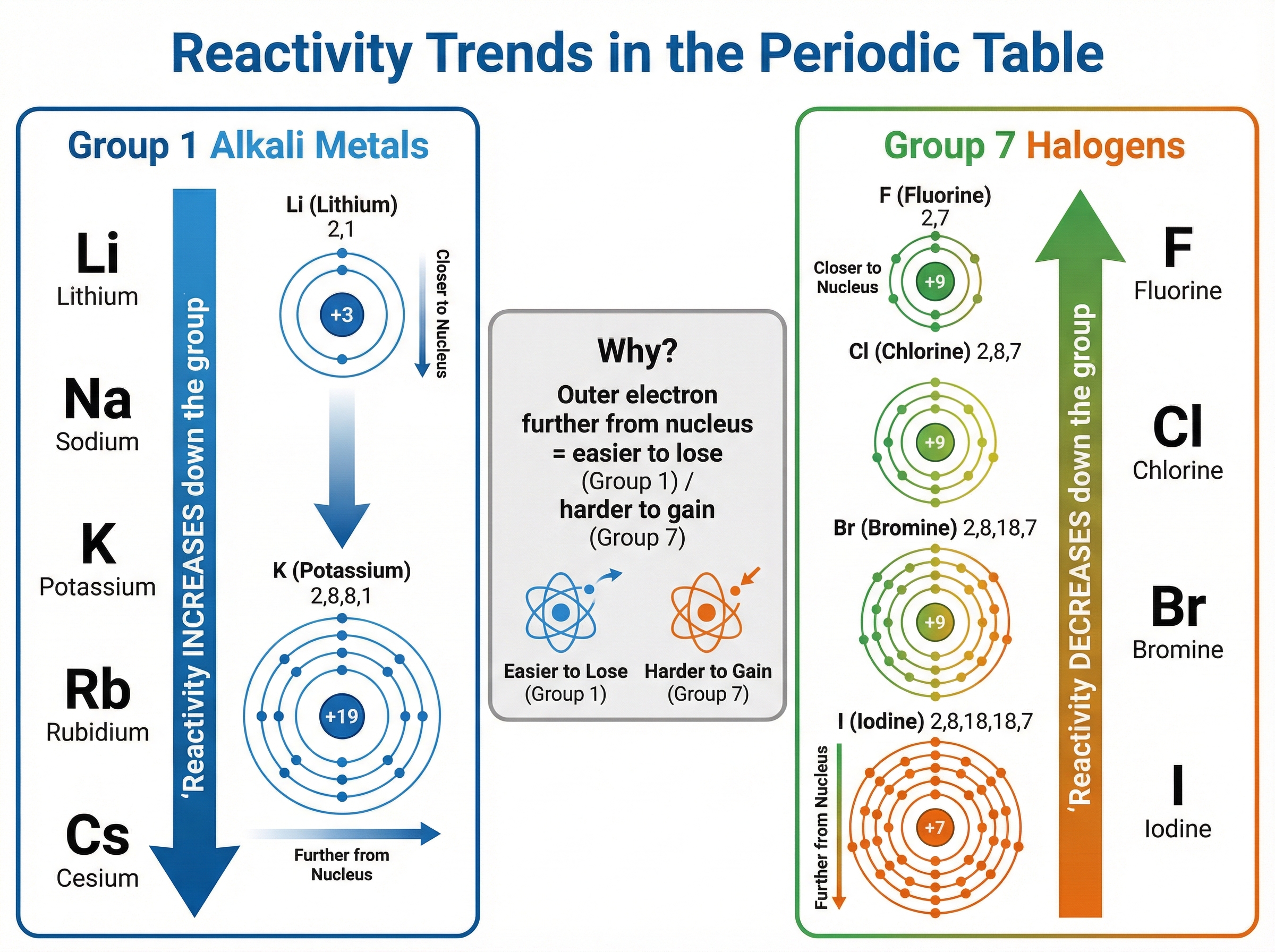
The alkali metals (Lithium, Sodium, Potassium, Rubidium, Caesium, and Francium) are highly reactive metals located in the first column of the Periodic Table. Their defining characteristic is that they all possess exactly **one electron in their outermost shell**. In chemical reactions, they lose this single electron to form stable 1+ ions with a full outer shell.
The most critical trend to understand is that **reactivity increases as you go down Group 1**. But why? As you move from Lithium down to Caesium, the atoms gain more electron shells. This means the outermost electron is located further away from the positive pull of the nucleus. Furthermore, the inner shells of electrons 'shield' the outer electron from the nuclear charge. Because the electrostatic attraction between the nucleus and the outer electron becomes weaker, the outer electron is lost much more easily, resulting in a more vigorous reaction.
**Example**: When potassium reacts with water, the reaction is so exothermic that it ignites the hydrogen gas produced, burning with a characteristic lilac flame. The equation is: 2K(s) + 2H₂O(l) → 2KOH(aq) + H₂(g).
### Concept 2: Group 7 - The Halogens
The halogens (Fluorine, Chlorine, Bromine, Iodine, and Astatine) are non-metals in Group 7. They all have **seven electrons in their outermost shell**, meaning they need to gain just one more electron to achieve a stable, full outer shell, forming 1- ions (halides).
In stark contrast to Group 1, **reactivity decreases as you go down Group 7**. As the atoms get larger with more electron shells, the outermost shell is further from the nucleus and experiences greater shielding. This makes it harder for the nucleus to attract and capture an incoming electron. Therefore, fluorine is the most reactive halogen, while iodine is much less reactive.
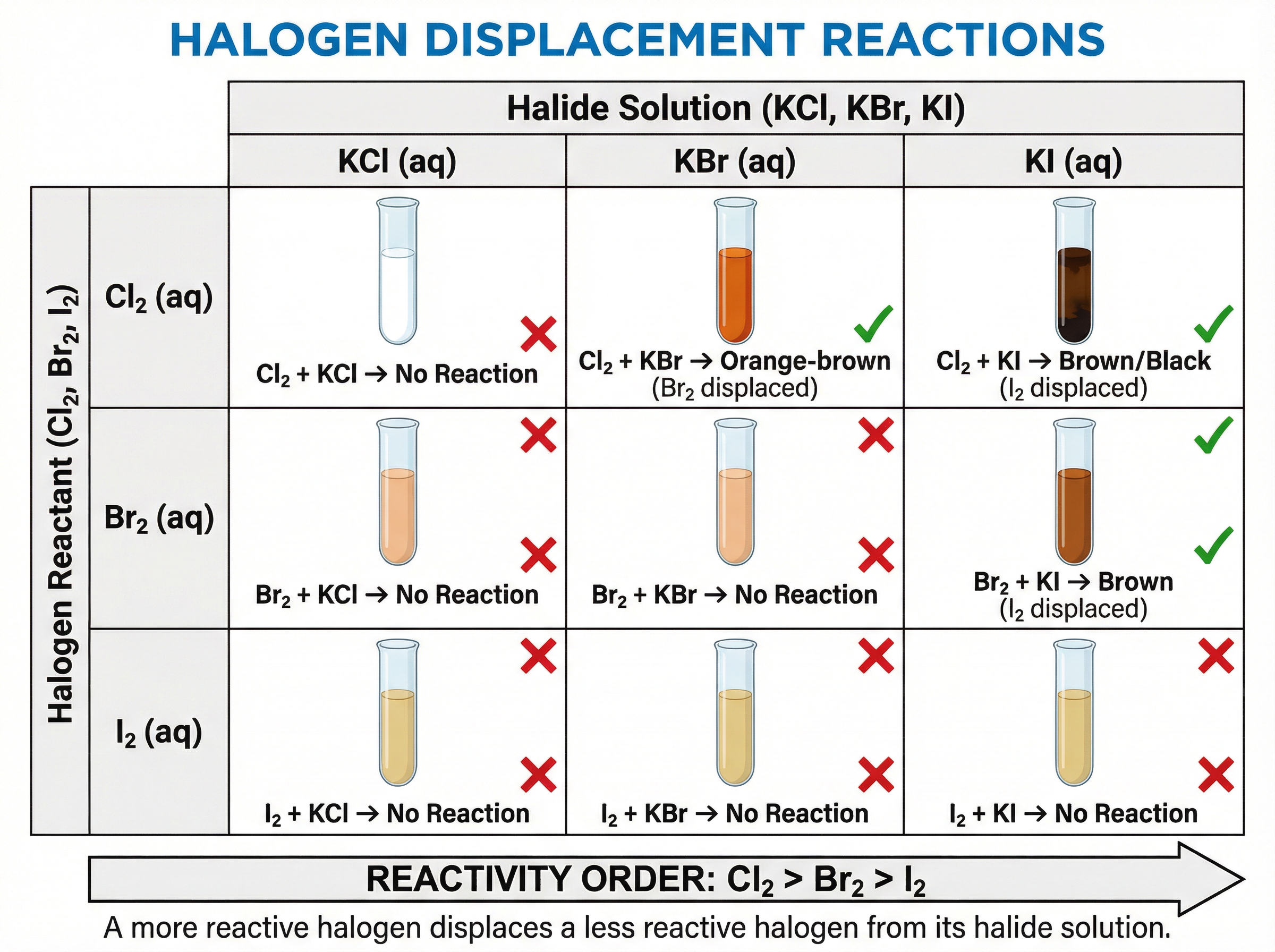
Halogens undergo **displacement reactions**, where a more reactive halogen will displace a less reactive halogen from an aqueous solution of its salt.
**Example**: If chlorine water (Cl₂) is added to potassium bromide (KBr) solution, the more reactive chlorine displaces the bromide ions, forming potassium chloride and releasing bromine (Br₂), which turns the solution orange-brown. The ionic equation is: Cl₂(aq) + 2Br⁻(aq) → 2Cl⁻(aq) + Br₂(aq).
### Concept 3: Transition Metals
The transition metals form the central block of the Periodic Table. Compared to the Group 1 alkali metals, they are harder, stronger, denser, and have much higher melting points (with the exception of mercury). They are also significantly less reactive.
From an exam perspective, transition metals have three unique chemical properties that you must memorise:
1. They can form ions with different charges (variable oxidation states), such as Fe²⁺ and Fe³⁺.
2. They form coloured compounds (e.g., copper(II) sulfate is blue).
3. They, and their compounds, act as excellent **catalysts** (e.g., iron in the Haber process).
### Concept 4: Qualitative Analysis (Flame Tests)
Chemists use specific tests to identify the presence of certain metal ions (cations). The most common method tested at GCSE is the flame test. When a metal compound is heated in a roaring Bunsen flame, the electrons are excited to higher energy levels and then emit specific wavelengths of light as they drop back down, producing characteristic colours.

While traditional chemical tests are useful, modern chemistry increasingly relies on **instrumental analysis** (such as flame emission spectroscopy). Instrumental methods are advantageous because they are highly **sensitive** (can detect tiny amounts), highly **accurate**, and very **fast**.
## Mathematical/Scientific Relationships
While this topic is heavily qualitative, you must be comfortable writing balanced symbol equations and ionic equations for reactions.
**General Equation for Alkali Metal + Water:**
2M(s) + 2H₂O(l) → 2MOH(aq) + H₂(g)
*(Where M represents any Group 1 metal)*
**General Equation for Halogen Displacement:**
X₂(aq) + 2Y⁻(aq) → 2X⁻(aq) + Y₂(aq)
*(Where X is the more reactive halogen and Y is the less reactive halide)*
## Practical Applications
Understanding these properties has profound real-world applications. The unreactive nature of Group 0 noble gases makes argon perfect for filling incandescent light bulbs to prevent the filament from burning. The variable oxidation states of transition metals allow platinum and palladium to be used in catalytic converters, reducing toxic emissions from cars. Meanwhile, the distinct colours produced by metal ions are exactly what give fireworks their brilliant displays—strontium for red, barium for green, and copper for blue.