Changes in Energy — AQA GCSE Study Guide
Exam Board: AQA | Level: GCSE
Master the essential energy calculations for your AQA GCSE Physics exam. This guide breaks down kinetic, potential, and elastic energy, showing you how to secure every mark with step-by-step worked examples and examiner insights.

## Overview
Welcome to one of the most fundamental topics in your AQA GCSE Physics course: **Changes in Energy (4.1.2)**. This isn't just about memorising formulas; it's about understanding the core principle that governs the universe: energy cannot be created or destroyed, only transferred between different stores. A solid grasp of this topic is crucial, as it forms the bedrock for understanding everything from electricity to mechanics. In the exam, you will be expected to perform multi-step calculations, often involving unit conversions and equation rearrangements. Questions frequently combine concepts, for example, asking you to calculate the speed of a falling object by equating its loss in gravitational potential energy to its gain in kinetic energy. This guide will equip you with the skills to tackle these calculation-heavy questions and describe the associated required practicals with the precision examiners are looking for.

## Key Concepts
### Concept 1: Energy Stores and Systems
An 'energy store' is exactly what it sounds like: a way of storing energy. Think of it like a bank account for energy. A 'system' is just an object or a group of objects that we are interested in. When something happens, energy is transferred from one store to another. For your exam, you need to be familiar with the main energy stores.
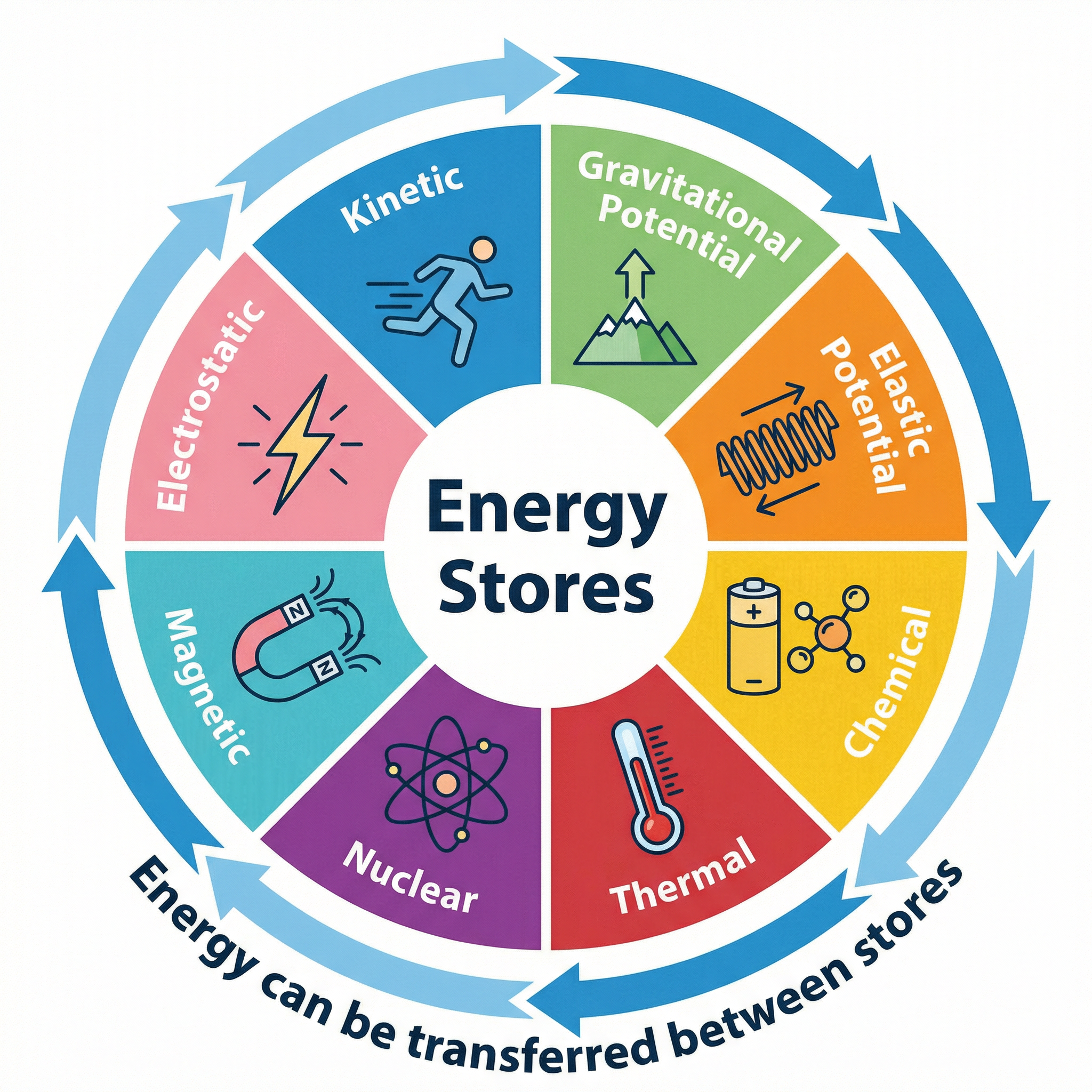
* **Kinetic:** The energy of a moving object.
* **Gravitational Potential:** Energy stored by an object due to its position in a gravitational field.
* **Elastic Potential:** Energy stored when an object is stretched or compressed.
* **Thermal:** The total kinetic and potential energy of the particles in an object.
* **Chemical:** Energy stored in the bonds between atoms.
* **Nuclear:** Energy stored in the nucleus of an atom.
* **Magnetic:** Energy stored when repelling poles have been pushed closer together or attracting poles have been pulled further apart.
* **Electrostatic:** Energy stored when repelling charges have been moved closer together or attracting charges have been pulled further apart.
### Concept 2: The Conservation of Energy
This is the golden rule: **Energy can be transferred usefully, stored or dissipated, but can never be created or destroyed.** In a 'closed system' (one where no energy can enter or leave), the total energy is always constant. However, in reality, energy transfers are rarely 100% efficient. Often, some energy is dissipated, usually as heat (to the thermal store of the surroundings), due to friction or air resistance. Examiners will award credit for using the term 'dissipated' or 'transferred to the thermal store' instead of the less precise term 'lost'.
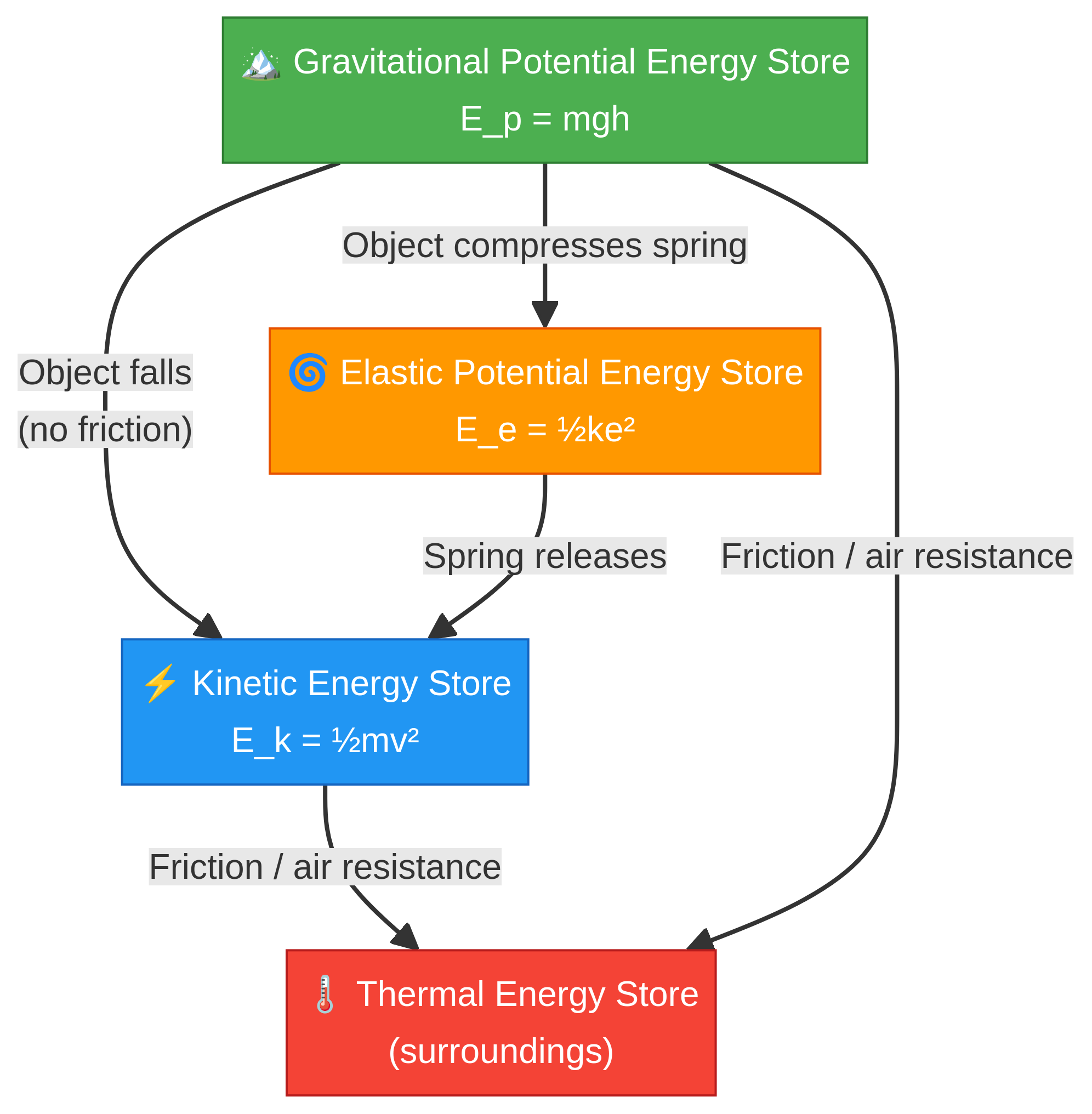
### Concept 3: Work Done
Work is done whenever energy is transferred from one store to another. In mechanics, work is done when a force causes an object to move. The amount of work done is calculated by: **Work Done (J) = Force (N) × Distance (m)**. This means the work done is equal to the energy transferred. For example, the work done to lift an object is equal to the gain in its gravitational potential energy store.
## Mathematical/Scientific Relationships
These are the three critical equations for this topic. You must be able to recall and apply them. The kinetic and GPE equations are for both tiers, while the elastic potential energy equation is for Higher Tier only.
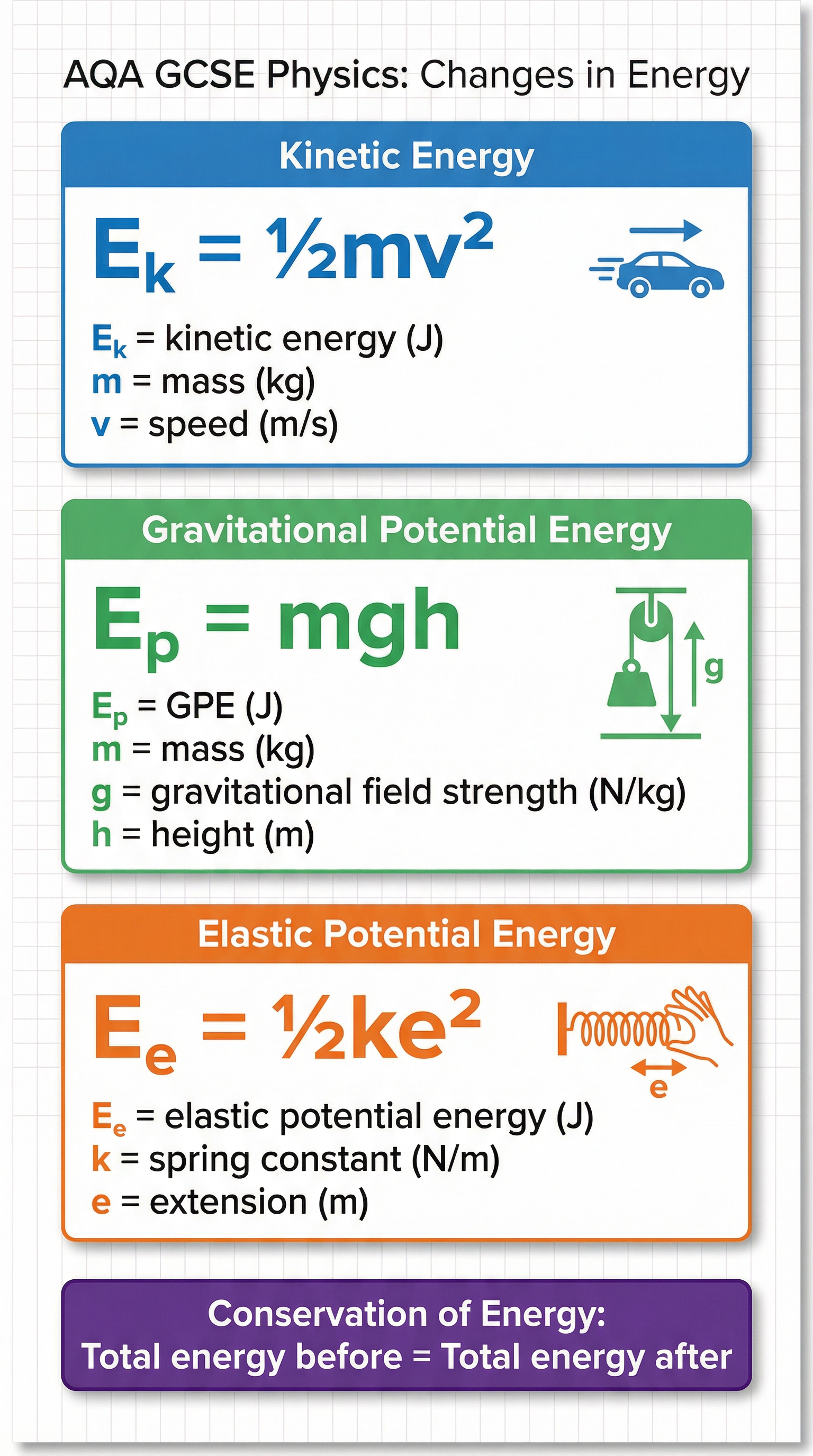
| Formula | What it Means | Tier | Status |
| :--- | :--- | :--- | :--- |
| **E_k = ½mv²** | Kinetic Energy = 0.5 × mass × (speed)² | Both | Must memorise |
| **E_p = mgh** | Gravitational Potential Energy = mass × gravitational field strength × height | Both | Must memorise |
| **E_e = ½ke²** | Elastic Potential Energy = 0.5 × spring constant × (extension)² | Higher Only | Must memorise |
| **ΔE = mcΔθ** | Change in Thermal Energy = mass × specific heat capacity × temperature change | Both | Given on formula sheet |
**Unit Conversions are Crucial:**
* Mass must be in **kilograms (kg)**. To convert grams to kg, divide by 1000.
* Speed must be in **metres per second (m/s)**.
* Height and extension must be in **metres (m)**. To convert cm to m, divide by 100.
* Energy must be in **joules (J)**. To convert kJ to J, multiply by 1000.
## Required Practical: Specific Heat Capacity
This practical is a common source of exam questions, including 6-mark questions asking for a detailed method.
**Aim:** To determine the specific heat capacity of a material.
**Apparatus:**
* A block of the material (e.g., aluminium) with two holes
* A thermometer
* An immersion heater
* A power supply
* A joulemeter (or a voltmeter and ammeter to calculate energy)
* Insulating material (e.g., cotton wool)
* A balance for measuring mass
* A stopwatch
**Method:**
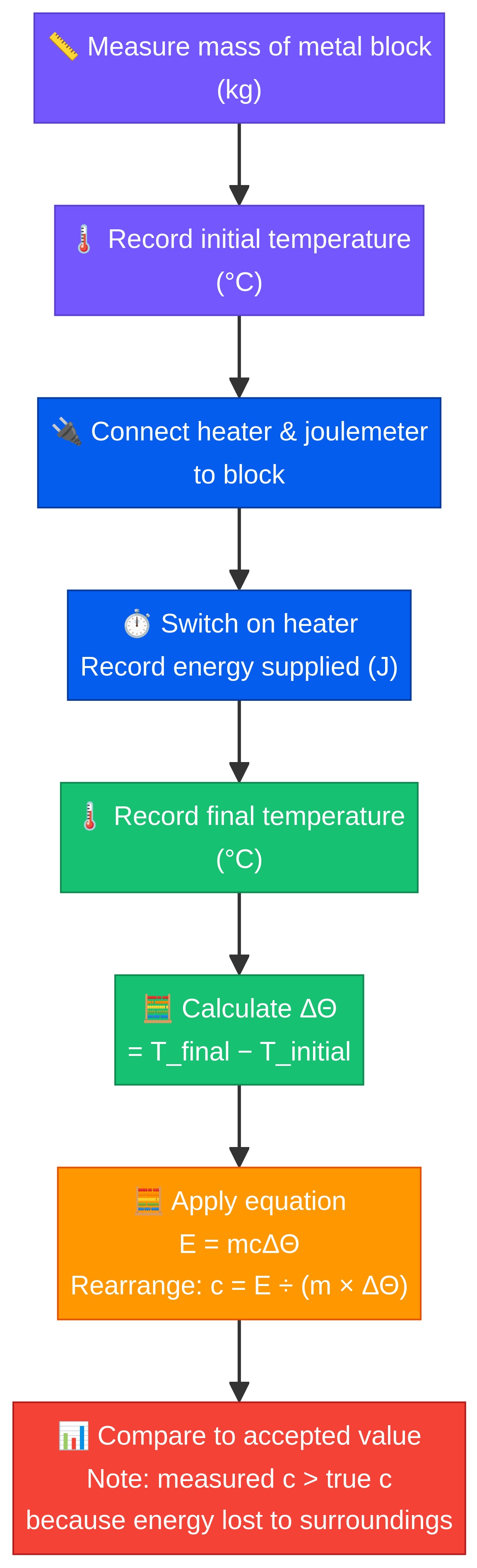
1. **Measure the mass** of the block using the balance.
2. **Assemble the apparatus**, placing the heater in one hole and the thermometer in the other. A small amount of water in the thermometer hole ensures good thermal contact.
3. **Wrap the block in insulation** to reduce energy transfer to the surroundings.
4. **Record the initial temperature** of the block.
5. **Switch on the power supply** and start the stopwatch. Record the energy supplied using the joulemeter (or calculate it using E = IVt).
6. After a set amount of energy has been supplied (or after a set time), **switch off the power**.
7. **Monitor the thermometer**. The temperature will continue to rise for a short while. Record the **highest temperature reached**.
8. **Calculate the temperature change (Δθ)**.
9. **Calculate the specific heat capacity (c)** using the formula c = ΔE / (m × Δθ).
**Common Errors & Improvements:**
* **Error:** Energy is transferred to the surroundings, not just the block. This makes the calculated specific heat capacity higher than the true value.
* **Improvement:** Use insulation and a lid to minimise this effect.
* **Error:** The thermometer may not be in good thermal contact with the block.
* **Improvement:** Place a drop of water in the hole with the thermometer.
* **Error:** Not waiting for the maximum temperature to be reached after switching off the heater.
* **Improvement:** Observe the thermometer carefully and record the peak temperature.