Density — AQA GCSE Study Guide
Exam Board: AQA | Level: GCSE
Master AQA GCSE Physics Density (4.3.1) with this comprehensive guide. We break down the core concepts, calculations, and the crucial Required Practical 5, giving you examiner insights to secure top marks."

## Overview
Density is a fundamental property of matter that describes how much 'stuff' is packed into a given space. For your AQA GCSE Physics exam, a solid understanding of density is crucial, not just for calculation questions but also for explaining the properties of solids, liquids, and gases using the particle model. This topic, specification point 4.3.1, frequently appears in exams, often featuring high-mark questions related to the required practical. Candidates who can confidently manipulate the density equation, describe experimental procedures with precision, and link microscopic particle behaviour to macroscopic properties will be well-rewarded. This guide will equip you with the knowledge and exam technique to tackle any density question thrown your way, connecting directly to topics like Particle Model (3.3), Energy (3.1), and Forces (3.4).

## Key Concepts
### Concept 1: The Definition of Density
At its core, **density** is the mass of a substance per unit volume. It's a measure of how compact the material is. A small, heavy object is very dense, while a large, light object is not. The key takeaway is that density is an intrinsic property of a material – a small iron nail and a large iron girder have the same density, because the material is the same.
**Example**: A 1 kg bag of feathers and a 1 kg block of iron have the same mass, but the iron block is much smaller. This is because iron is far denser than feathers; more mass is packed into a smaller volume.
### Concept 2: The Particle Model of Matter
The density of a substance is determined by the mass of its individual particles and how closely those particles are packed together. This is where the particle model becomes essential for high-level explanations.
* **Solids**: Particles are arranged in a fixed, regular lattice and vibrate about fixed positions. They are packed very tightly, meaning there are a large number of particles per unit volume. This results in high density.
* **Liquids**: Particles are in close contact but are free to move past one another. The spacing is slightly larger than in solids, so most liquids are slightly less dense than their solid form (a key exception being water).
* **Gases**: Particles are far apart and move randomly and rapidly. There is a large amount of empty space between particles, meaning there are very few particles per unit volume. This results in very low density.
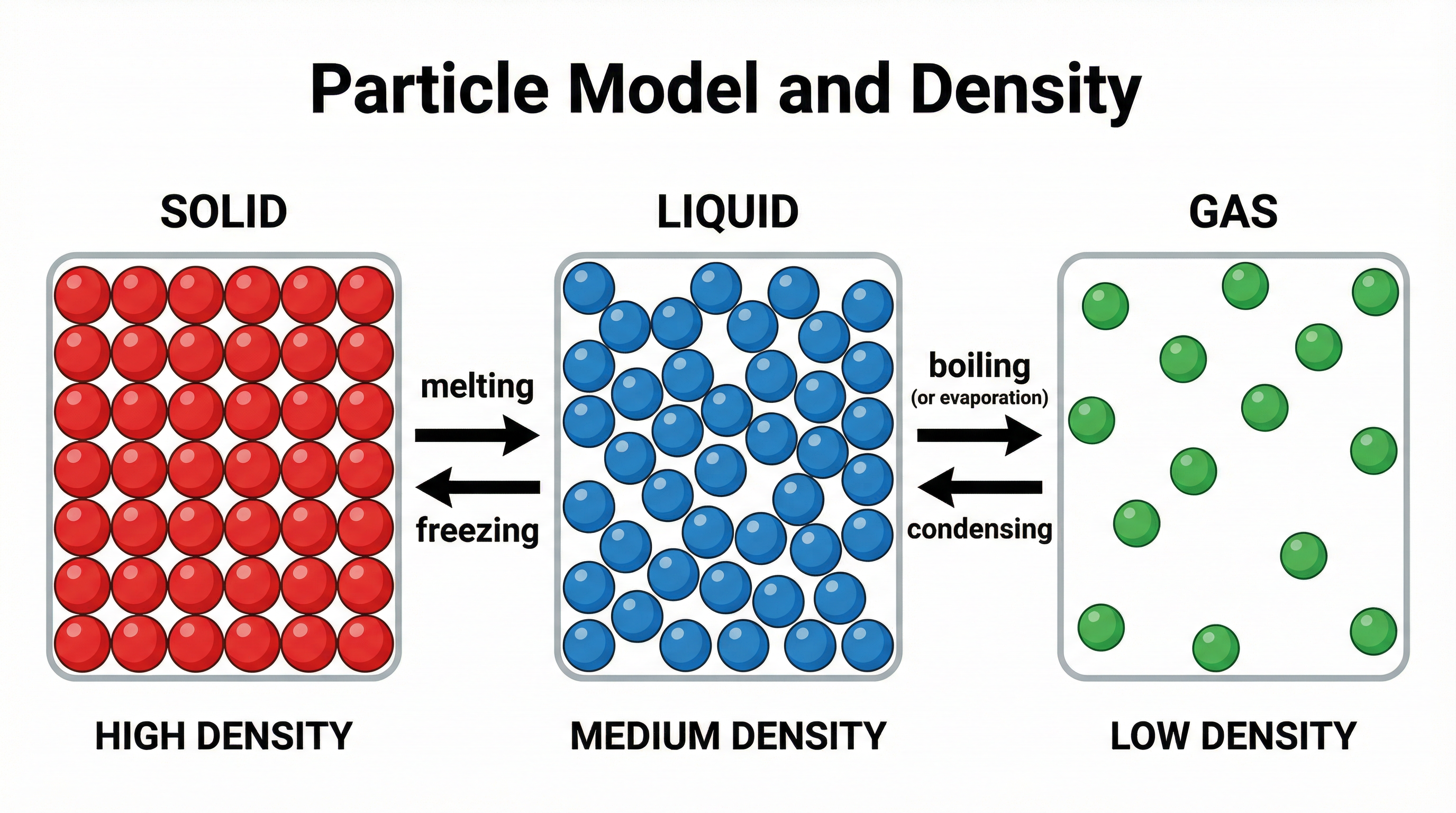
**Examiner Tip**: When explaining differences in density, you must use the phrase **'number of particles per unit volume'** to gain full credit. Simply saying 'particles are closer' is often not enough for the mark.
## Mathematical/Scientific Relationships
The cornerstone of this topic is the density equation. You are expected to know it, rearrange it, and use it confidently.
**The Density Formula (Must memorise)**
\[
\rho = \frac{m}{V}
\]
Where:
* **ρ (rho)** is the density, measured in kilograms per metre cubed (kg/m³) or grams per centimetre cubed (g/cm³).
* **m** is the mass, measured in kilograms (kg) or grams (g).
* **V** is the volume, measured in metres cubed (m³) or centimetres cubed (cm³).
**Rearranging the Formula**
You must also be able to rearrange the formula to solve for mass or volume:
* To find mass: **m = ρ × V**
* To find volume: **V = m / ρ**
**Unit Conversions (Common Pitfall)**
Examiners often test unit conversions. Be ready for them.
* **g/cm³ to kg/m³**: Multiply by 1000. (e.g., 2.7 g/cm³ = 2700 kg/m³)
* **cm³ to m³**: Multiply by 10⁻⁶ or divide by 1,000,000. (e.g., 100 cm³ = 0.0001 m³)
## Required Practical 5: Determining Density
This practical is a major source of exam questions, including 6-mark 'describe a method' questions. You need to know the procedure for three different types of object.
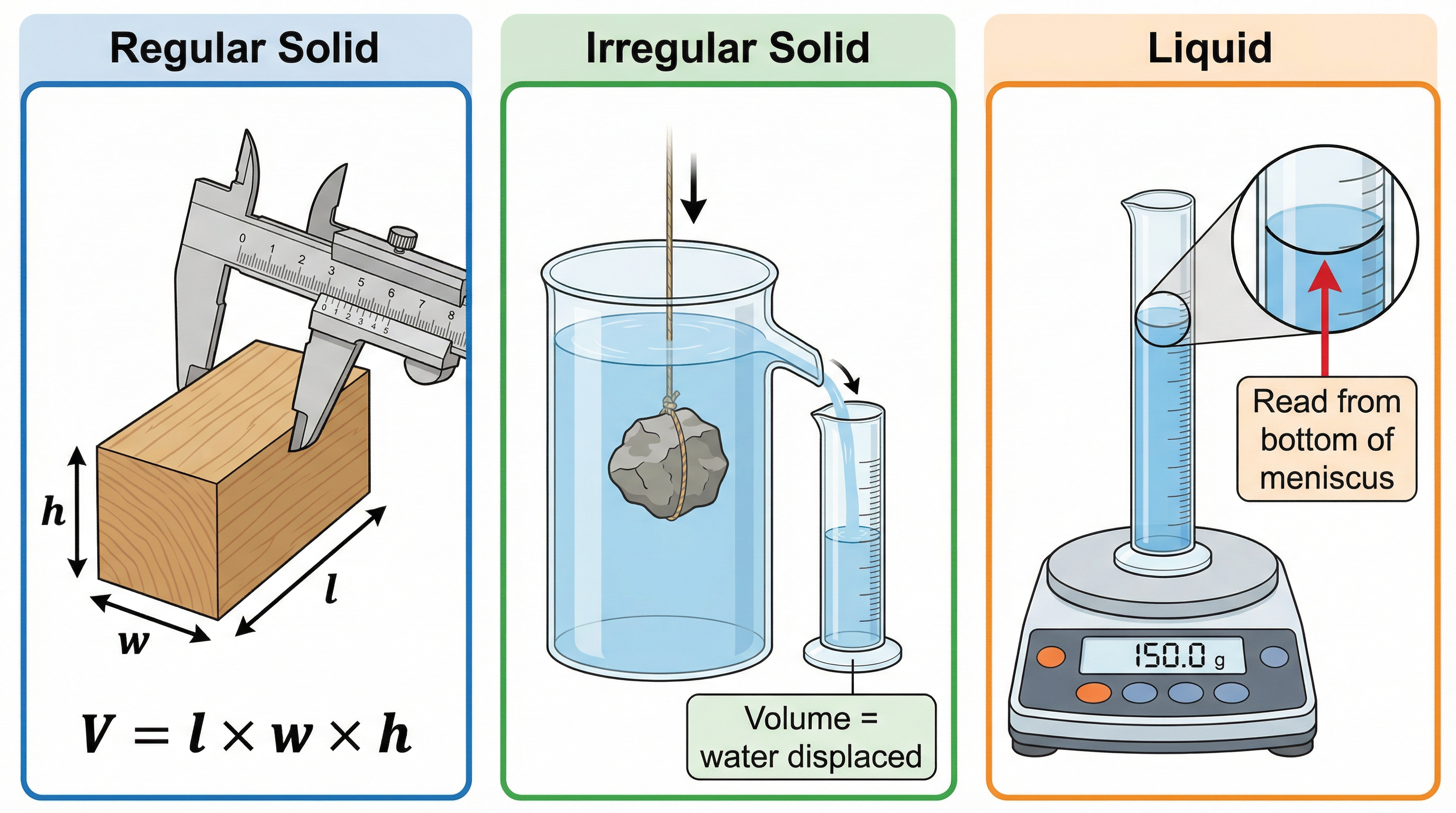
**Apparatus List**
* Top-pan balance (or digital balance)
* Ruler / Vernier callipers / Micrometer
* Displacement can (Eureka can)
* Measuring cylinders
* The objects/liquids to be measured
**Method 1: Regular Solid (e.g., a cube)**
1. **Measure Mass**: Place the object on a top-pan balance and record its mass in kg.
2. **Measure Dimensions**: Use a ruler or vernier callipers to measure the length, width, and height of the object in metres. Take at least three readings for each dimension and calculate a mean to improve accuracy.
3. **Calculate Volume**: Calculate the volume using the appropriate formula (e.g., V = l × w × h for a cuboid).
4. **Calculate Density**: Use the formula ρ = m/V to calculate the density.
**Method 2: Irregular Solid (e.g., a rock)**
1. **Measure Mass**: Measure the mass of the rock using a top-pan balance.
2. **Measure Volume**: Fill a displacement can with water until it is level with the bottom of the spout. Place a measuring cylinder under the spout. Carefully lower the rock into the can. The volume of water that is displaced into the measuring cylinder is equal to the volume of the rock. Record this volume.
3. **Calculate Density**: Use the formula ρ = m/V to calculate the density.
**Method 3: Liquid**
1. **Measure Mass of Empty Cylinder**: Place an empty measuring cylinder on a top-pan balance and record its mass.
2. **Measure Volume of Liquid**: Pour the liquid into the measuring cylinder and record the volume. Ensure you read from the bottom of the meniscus to avoid parallax error.
3. **Measure Total Mass**: Measure the combined mass of the measuring cylinder and the liquid.
4. **Calculate Mass of Liquid**: Subtract the mass of the empty cylinder from the total mass to find the mass of the liquid alone.
5. **Calculate Density**: Use the formula ρ = m/V to calculate the density of the liquid."