Internal Energy — AQA GCSE Study Guide
Exam Board: AQA | Level: GCSE
Unlock top marks in AQA GCSE Physics by mastering Internal Energy. This guide breaks down the crucial difference between temperature and energy, showing you how to analyse heating curves and apply the correct formulas (E=mcΔθ vs E=mL) to secure every mark.

## Overview
Welcome to your deep dive into Internal Energy, specification point 4.3.3 for AQA GCSE Physics. This topic is fundamental because it explains what happens to substances at a particle level when we heat them. Examiners love to test this because it requires you to distinguish between temperature and energy, a concept that many candidates find tricky. A typical exam question might present you with a heating graph and ask you to calculate the energy required for a specific section, or explain why the temperature remains constant during melting. Mastering the content in this guide will give you a significant advantage, allowing you to confidently tackle calculation and long-answer explanation questions. This topic links directly to Particle Model of Matter (4.3.1) and Energy Changes (4.1.1), so a strong understanding here will boost your performance across the paper.

## Key Concepts
### Concept 1: Internal Energy (The Total Energy Store)
Every substance is made of particles (atoms and molecules) that are constantly moving and interacting. The **internal energy** of a system is the total energy that its particles have in their kinetic and potential energy stores. It's a measure of the *entire* energy content at the particle level.
- **Kinetic Energy**: This is due to the movement of the particles. In a solid, they vibrate around fixed positions. In a liquid, they move past each other. In a gas, they move randomly and rapidly. **Temperature is a measure of the *average* kinetic energy of the particles.** When you heat a substance and its temperature rises, the particles are moving or vibrating faster.
- **Potential Energy**: This is due to the forces of attraction (bonds) between the particles. To change the state of a substance (e.g., melt a solid or boil a liquid), you need to overcome these forces and push the particles further apart. This increases their potential energy. When the state changes, the potential energy store is altered.
### Concept 2: Specific Heat Capacity (Changing Temperature)
When you add energy to a substance and its temperature changes, the relationship is governed by its **Specific Heat Capacity (c)**. This is defined as **the amount of energy required to raise the temperature of 1kg of a substance by 1°C.**
A substance with a high specific heat capacity, like water (c = 4200 J/kg°C), requires a lot of energy to heat up. A substance with a low specific heat capacity, like copper (c = 390 J/kg°C), heats up very quickly. This is why a metal spoon gets hot much faster than the soup it's in!
**When to use it**: Use the Specific Heat Capacity equation for any part of a problem where the temperature is changing (the sloped sections of a heating graph).
### Concept 3: Specific Latent Heat (Changing State)
This is the concept that catches most students out. When a substance is changing state (melting, boiling, freezing, or condensing), you can keep adding energy, but the temperature will **not** change. The graph of temperature against time goes flat.
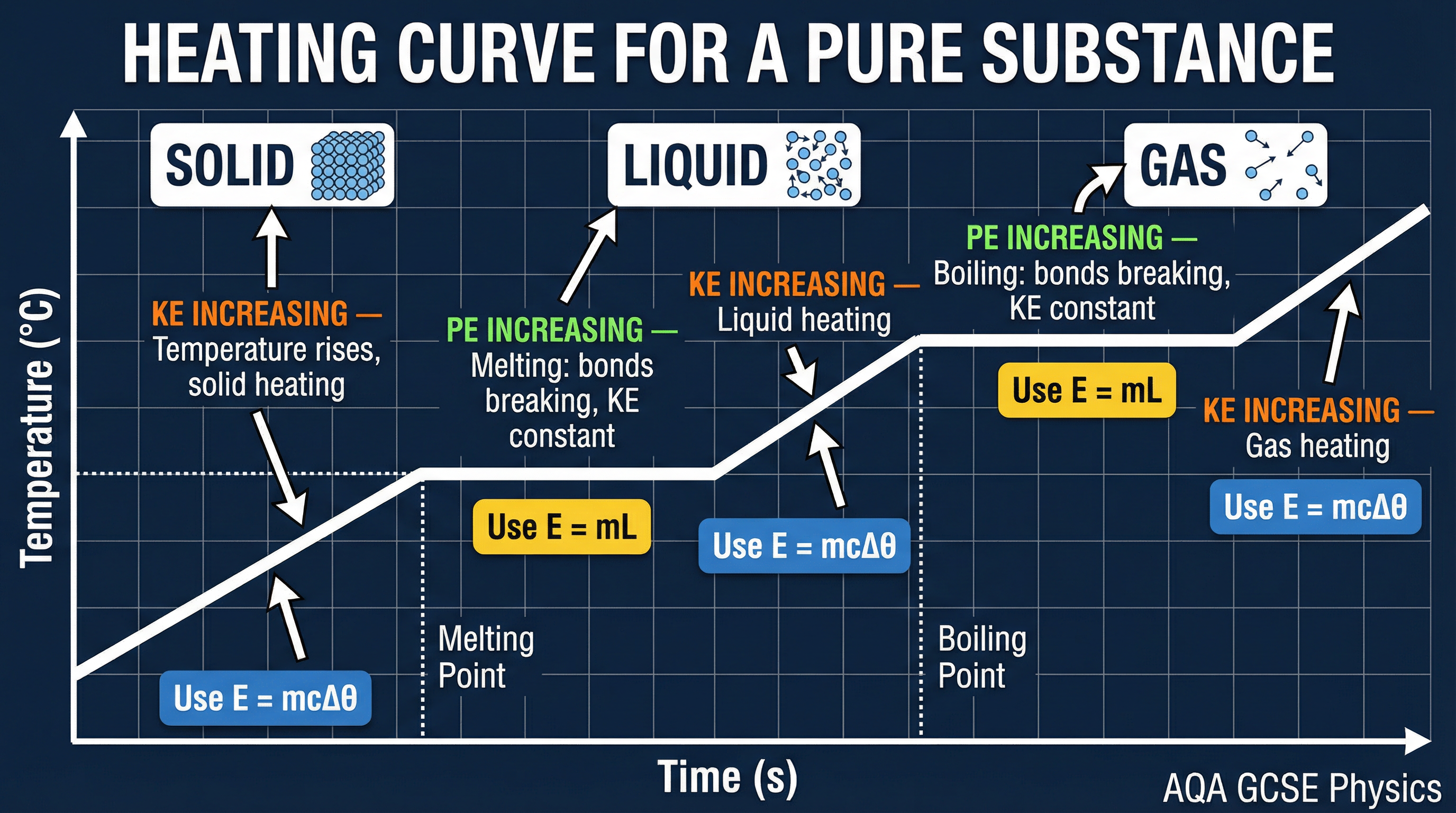
During this process, the energy being supplied is not increasing the kinetic energy of the particles (so temperature stays constant). Instead, it is being used to break the intermolecular bonds and increase the potential energy of the particles. This energy is called **latent heat**, which means 'hidden heat'.
- **Specific Latent Heat of Fusion (Lf)**: The energy required to change the state of 1kg of a substance from solid to liquid (or vice versa) with no change in temperature.
- **Specific Latent Heat of Vaporisation (Lv)**: The energy required to change the state of 1kg of a substance from liquid to gas (or vice versa) with no change in temperature.
**When to use it**: Use the Specific Latent Heat equation for any part of a problem where the state is changing (the flat sections of a heating graph).
## Mathematical/Scientific Relationships
There are two key equations you must be able to select and apply. One is for temperature changes, the other for state changes.
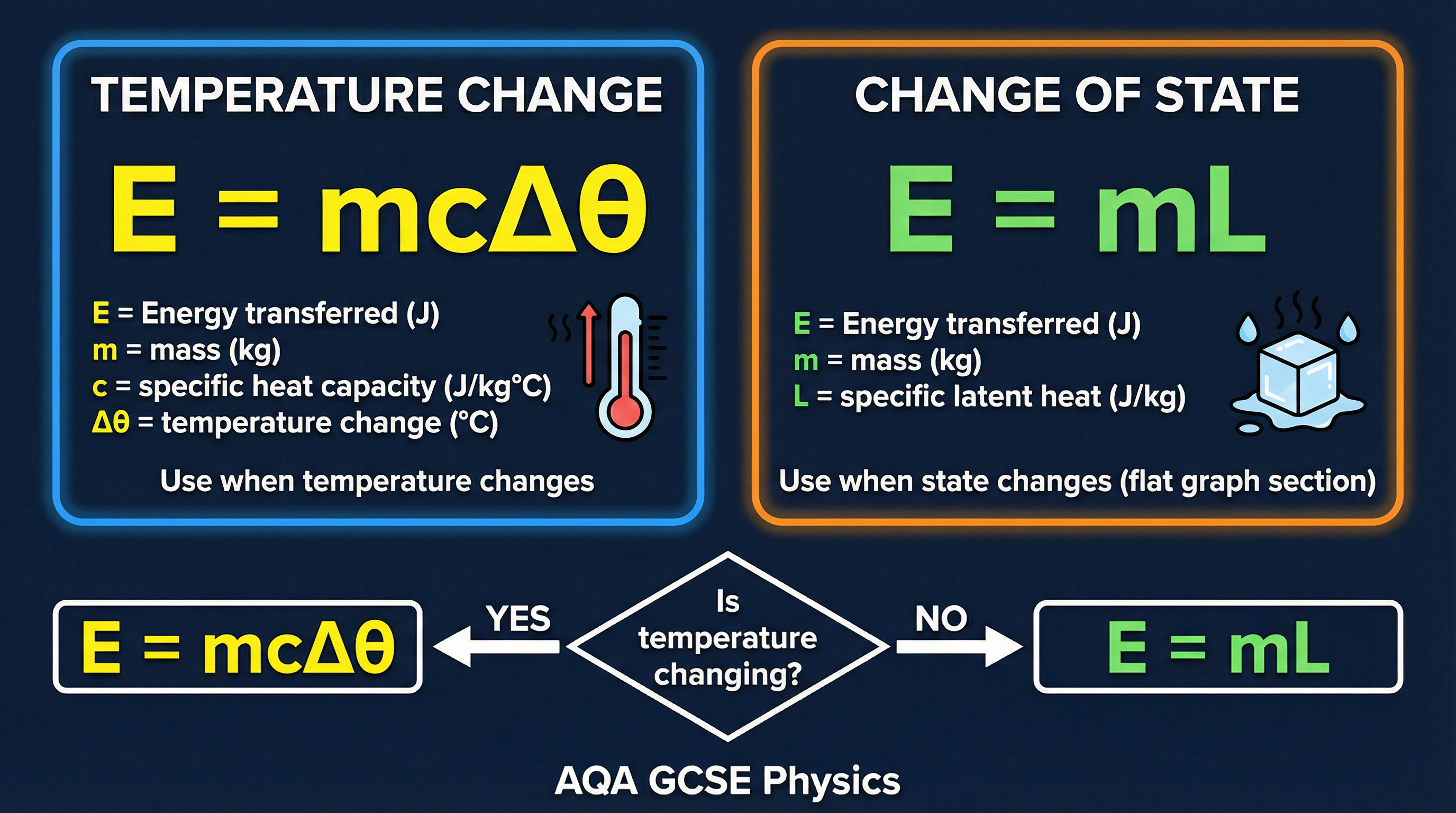
1. **Energy for Temperature Change (Must memorise)**
`E = m c Δθ`
- `E` = Energy transferred, in Joules (J)
- `m` = mass, in kilograms (kg)
- `c` = specific heat capacity, in Joules per kilogram per degree Celsius (J/kg°C)
- `Δθ` (delta theta) = change in temperature, in degrees Celsius (°C)
2. **Energy for a Change of State (Given on formula sheet)**
`E = m L`
- `E` = Energy transferred, in Joules (J)
- `m` = mass, in kilograms (kg)
- `L` = specific latent heat, in Joules per kilogram (J/kg). This will be either `Lf` for fusion or `Lv` for vaporisation.
**Unit Conversions are Critical**: Examiners often give mass in grams (g) or energy in kilojoules (kJ). You MUST convert these to the standard units of kilograms (kg) and Joules (J) before substituting into the equations.
- To convert g to kg: divide by 1000.
- To convert kJ to J: multiply by 1000.
## Practical Applications
This topic is directly tested in the **Required Practical: Specific Heat Capacity**. In this experiment, you typically heat a 1kg block of a specific material (e.g., aluminium) using an electric heater. You measure the energy input from the heater (using a joulemeter or by calculating Power x time) and the temperature change of the block with a thermometer. You can then rearrange `E = mcΔθ` to find the specific heat capacity `c = E / (mΔθ)`. A key source of error is energy loss to the surroundings. To reduce this, the block should be insulated by wrapping it in a layer of foam or cotton wool. Repeating the experiment and calculating an average improves the accuracy of the result.