Models of the Atom — AQA GCSE Study Guide
Exam Board: AQA | Level: GCSE
Unlock top marks in AQA GCSE Physics by mastering the story of the atom. This guide breaks down the historical models, from Thomson's Plum Pudding to Rutherford's nuclear atom, focusing on the crucial alpha scattering experiment that changed science forever.
## Overview
Welcome to your guide for AQA GCSE Physics topic 4.4.1: Models of the Atom. This topic is a narrative of scientific discovery, tracing how our understanding of the atom has evolved over time. It's a story of evidence leading to new theories, a core principle of science. For your exam, the focus is less on complex calculations and more on your ability to clearly explain the evidence from key experiments and link it logically to the conclusions that were drawn. AQA examiners place a heavy emphasis on the alpha particle scattering experiment, as it provides a perfect case study in how scientific models are tested, refuted, and replaced. Expect questions that ask you to describe and compare different models, explain the scattering experiment, and link specific observations to the features of the nuclear atom. Mastering this topic not only secures marks here but also builds a foundation for understanding radioactivity and nuclear physics later in the specification.

## Key Concepts
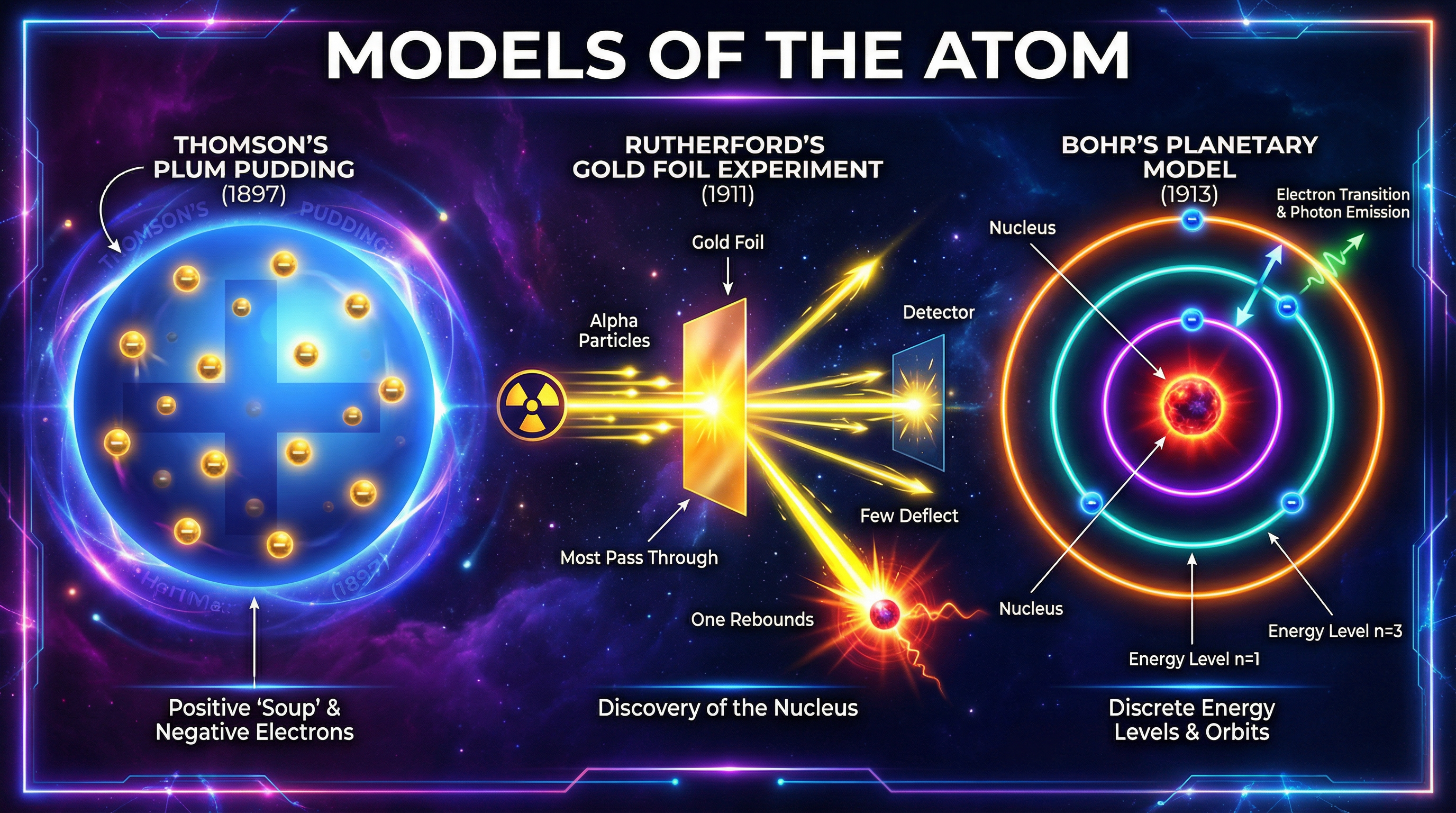
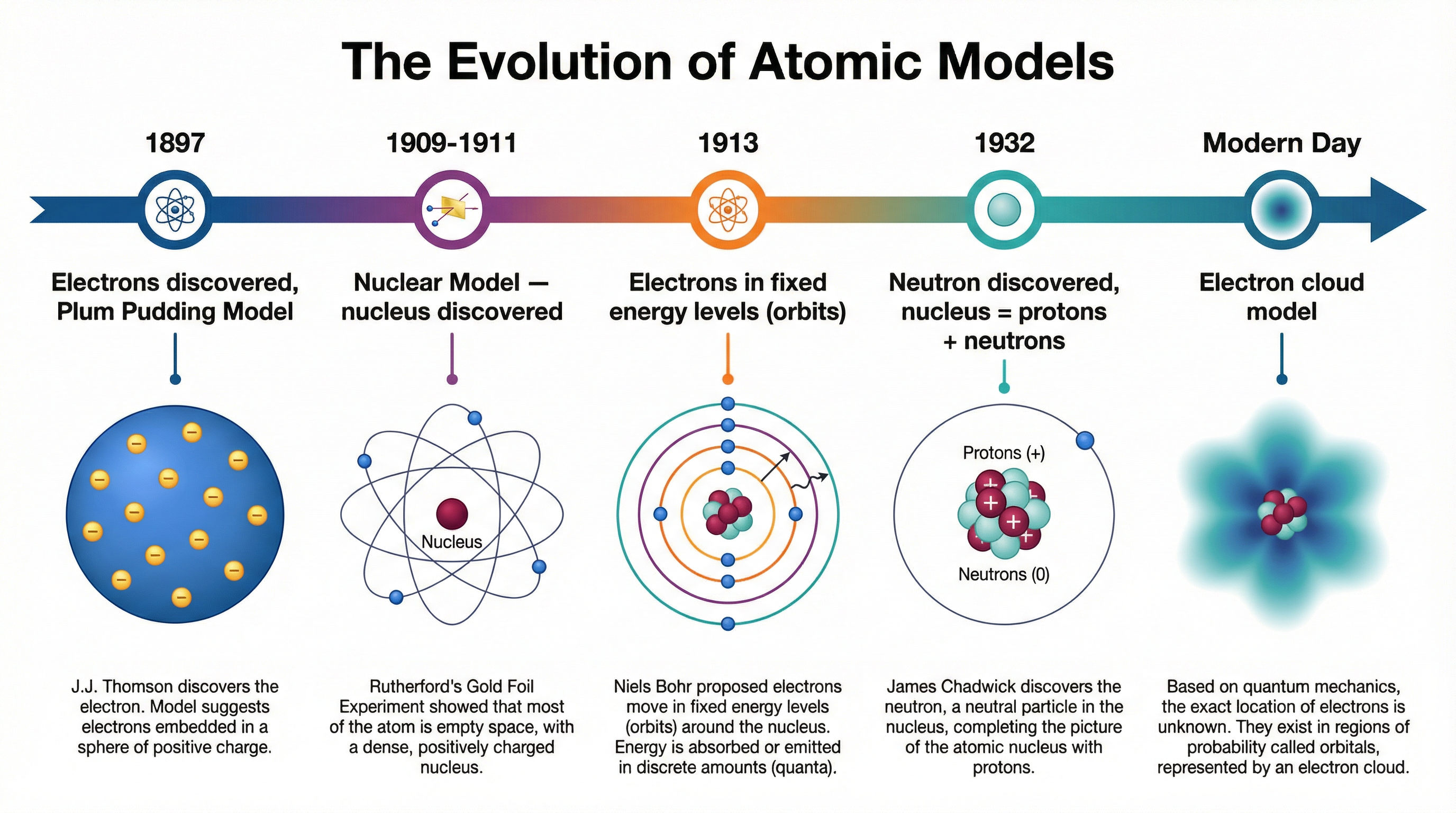
### Concept 1: The Plum Pudding Model (J.J. Thomson, 1897)
Before 1897, scientists like John Dalton imagined atoms as tiny, solid, indivisible spheres. However, J.J. Thomson's experiments with cathode rays led to a groundbreaking discovery: the electron. These were tiny, negatively charged particles that were much smaller than atoms. Since atoms themselves are electrically neutral, Thomson reasoned that if they contain negative electrons, they must also contain a source of positive charge to balance it out.
This led him to propose the **Plum Pudding Model**. Imagine a blueberry muffin or a traditional plum pudding. The 'pudding' or 'muffin batter' itself represents a diffuse sphere or 'ball' of positive charge. The electrons (the 'plums' or 'blueberries') are then scattered or embedded within this positive sphere.
**Key Features of the Plum Pudding Model:**
- The atom is a sphere of positive charge.
- Negative electrons are embedded within it.
- There is no nucleus.
- The mass and charge are spread out evenly throughout the entire atom.
This was the accepted model for over a decade, but it had a fatal flaw: it couldn't explain the results of Rutherford's next big experiment.
### Concept 2: The Alpha Particle Scattering Experiment (Rutherford, Geiger & Marsden, 1909-1911)
This is the most important part of the topic. Ernest Rutherford, along with his assistants Hans Geiger and Ernest Marsden, designed an experiment to probe the structure of the atom. Their findings would completely overturn the Plum Pudding model.
**The Setup:**
1. **Alpha Source:** A radioactive source was used to emit a beam of alpha (α) particles. Alpha particles are small, dense, and have a positive charge (+2).
2. **Gold Foil:** The alpha particles were fired at an incredibly thin sheet of gold foil, only a few atoms thick.
3. **Detector Screen:** A circular screen coated with zinc sulfide was placed around the foil. This screen would produce a tiny flash of light (a scintillation) whenever it was struck by an alpha particle, allowing the scientists to see where the particles went.
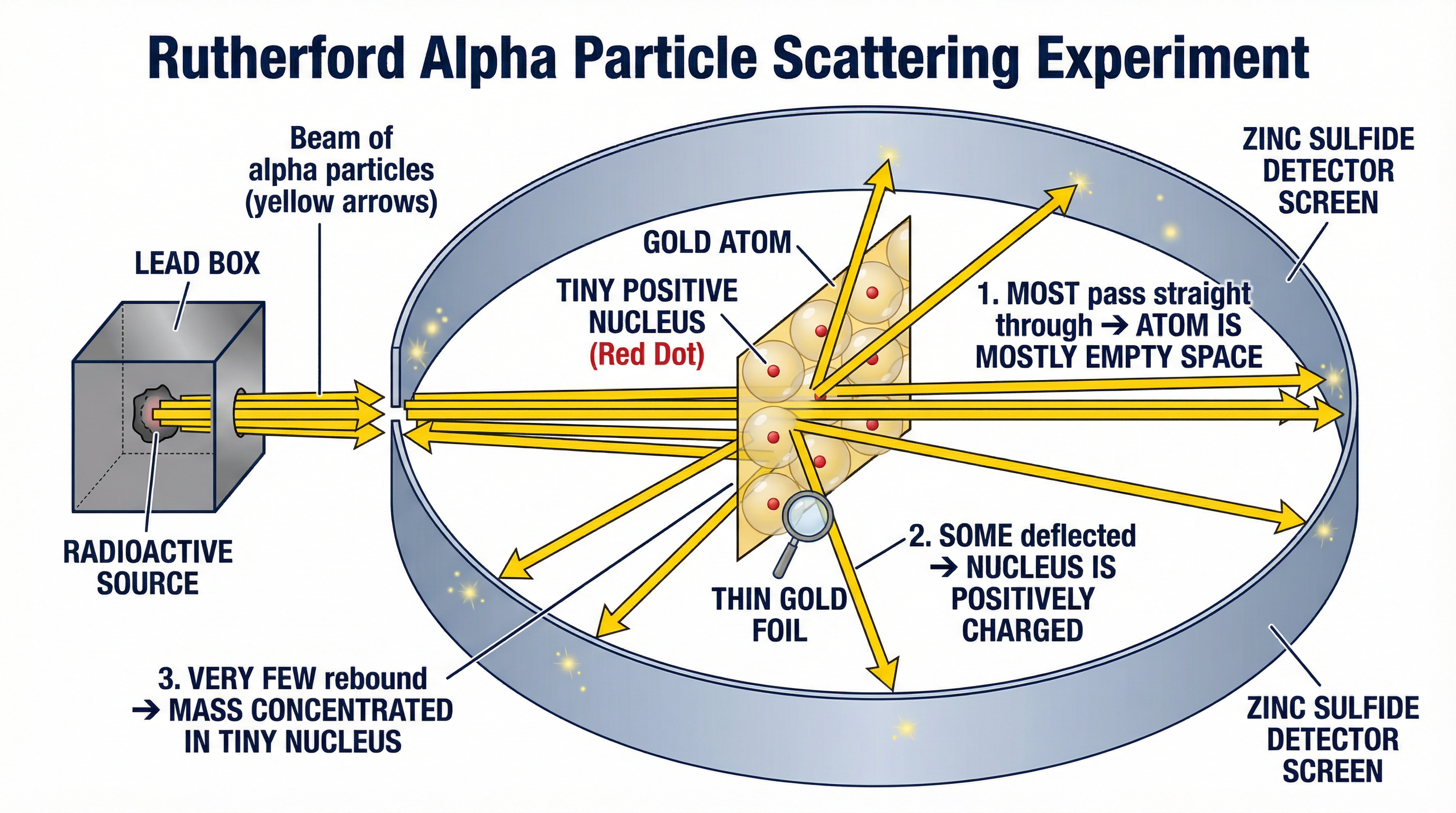
**The Three Key Observations and Conclusions:**
This is the core knowledge you must have. Examiners expect you to link each specific observation to its corresponding conclusion.
| Observation | Conclusion | Explanation |
| -------------------------------------------------- | ----------------------------------------------------------------------- | -------------------------------------------------------------------------------------------------------------------------------------- |
| 1. **Most** alpha particles passed straight through. | The atom is **mostly empty space**. | The vast majority of the atom contains nothing that can stop or deflect the alpha particles, so they pass through unaffected. |
| 2. A **small number** of alpha particles were deflected. | The centre of the atom has a concentration of **positive charge** (the nucleus). | The positive alpha particles were repelled by the positive nucleus via an electrostatic force, causing them to change direction. |
| 3. A **very few** alpha particles rebounded (>90°). | The nucleus is **tiny, dense, and contains most of the mass**. | Only a direct, head-on approach to the nucleus results in a strong enough repulsion to send the particle backwards. This is a rare event. |
These results were completely incompatible with the Plum Pudding model. If mass and charge were spread out, you would expect all the alpha particles to pass through with only minor deflections. The fact that some were deflected at large angles and some even bounced back meant a new model was needed.
### Concept 3: The Nuclear Model (Rutherford, 1911)
Based on the evidence from the scattering experiment, Rutherford proposed the **Nuclear Model** of the atom.
**Key Features of the Nuclear Model:**
- A tiny, dense, positively charged **nucleus** at the centre.
- The nucleus contains almost all of the atom's **mass**.
- The atom is **mostly empty space**.
- Negative **electrons orbit** the nucleus at a distance, like planets around the sun.
This model successfully explained all the observations from the gold foil experiment. It marked the discovery of the nucleus and remains the foundation of our modern understanding.
### Concept 4: Bohr and Chadwick - Refining the Model
While revolutionary, Rutherford's model wasn't perfect. According to the laws of physics at the time, the orbiting electrons should lose energy and spiral into the nucleus, causing the atom to collapse. In 1913, **Niels Bohr** adapted the nuclear model by suggesting that electrons could only orbit the nucleus at specific, fixed distances, known as **energy levels** or **shells**. An electron can exist in one shell or another, but not in between. This explained why atoms were stable.
Finally, in 1932, **James Chadwick** discovered the **neutron**. This was a neutral particle found in the nucleus with a mass similar to a proton. His discovery completed the basic picture of the atom that you use at GCSE: a nucleus containing positive protons and neutral neutrons, surrounded by orbiting negative electrons in specific energy levels.