Particle Motion in Gases — AQA GCSE Study Guide
Exam Board: AQA | Level: GCSE
Particle Motion in Gases unlocks the microscopic world behind everyday phenomena — from why a tyre inflates to why a bicycle pump gets warm. This topic sits at the heart of AQA GCSE Physics 4.3.4, requiring candidates to apply the kinetic theory model to explain temperature, pressure, and Boyle's Law with precision. Master the key vocabulary and the p₁V₁ = p₂V₂ equation and you will be well-placed to earn marks across all three Assessment Objectives.

## Overview
Particle Motion in Gases (AQA specification reference 4.3.4) is one of the most logical and internally consistent topics on the GCSE Physics course. Once you understand the central idea — that gas particles are in constant, random motion, and that everything else follows from this — the whole topic clicks into place. This guide covers the kinetic theory model, the relationship between temperature and kinetic energy, the origin of gas pressure, and Boyle's Law, including Higher Tier content on doing work on a gas.
This topic connects directly to energy stores (4.1), density (4.3.3), and forces (4.5), meaning examiners can and do set synoptic questions that bridge these areas. Typical exam question styles include two-mark "describe" questions about particle motion, three-mark "explain" questions about pressure changes, and three-mark calculation questions using p₁V₁ = p₂V₂. Higher Tier candidates should also expect questions about the bicycle pump scenario and the thermodynamic relationship between work, internal energy, and temperature.

---
## Key Concepts
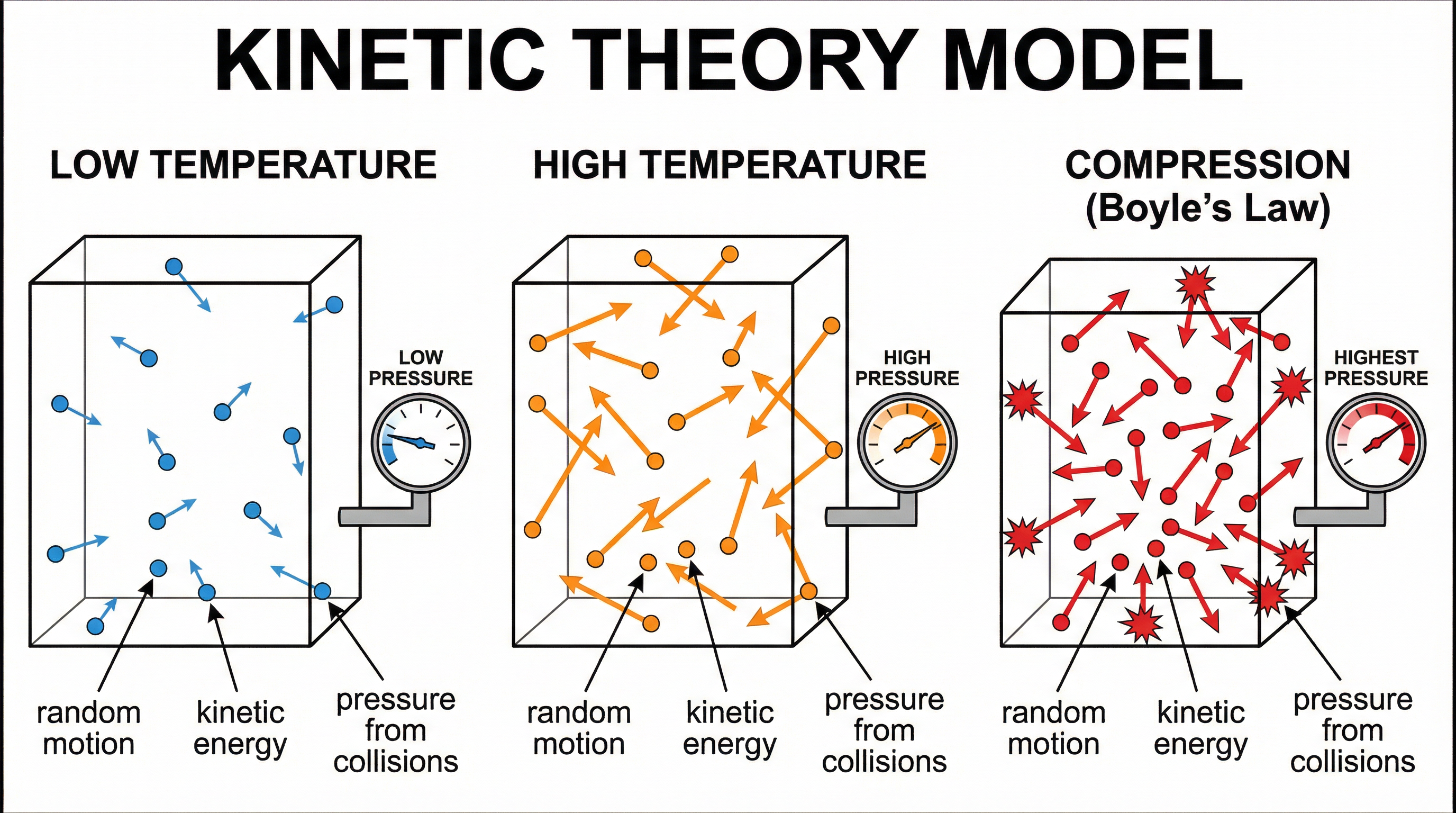
### Concept 1: The Kinetic Theory Model — Random Motion
The kinetic theory of gases rests on one foundational idea: **gas particles are in constant, random motion**. Unlike solids (where particles vibrate in fixed positions) or liquids (where particles flow but remain close), gas particles have sufficient energy to move freely and independently throughout the entire volume of their container.
The two features of this motion that AQA examiners specifically test are:
1. Particles move in **random directions**
2. Particles have a **range of speeds**
The word "random" is non-negotiable. Candidates who write "particles move around" or "particles move in all directions" without using the word "random" will not be awarded the AO1 mark. Practise writing this phrase until it is automatic.
**Analogy**: Think of a busy school corridor at break time. Students (particles) move in all sorts of directions, some fast, some slow, bumping into walls and each other — that is a reasonable mental model of a gas.
---
### Concept 2: Temperature and Average Kinetic Energy
Temperature is not just a measure of how hot something feels — it is a direct measure of the **average kinetic energy** of the particles in a substance. This is the key link that connects the macroscopic (temperature you can measure with a thermometer) to the microscopic (individual particle speeds).
**The relationship**: An increase in temperature → an increase in the average kinetic energy of the particles → particles move faster on average.
The word "average" matters because not all particles move at the same speed. There is always a distribution of speeds. At any given temperature, some particles are moving very fast and some are moving slowly, but the average kinetic energy is determined by the temperature.
**Common error to avoid**: Candidates sometimes state that "the particles get hotter" or "the particles expand." This is incorrect. The particles themselves do not change in size. What changes is their **kinetic energy** (and therefore their speed). The space between particles may increase if the container allows it, but the particles themselves remain the same size.
---
### Concept 3: Gas Pressure — Collisions with Container Walls
Gas pressure arises from the cumulative effect of billions of particles colliding with the **walls of the container** every second. Each collision exerts a tiny force on the wall. Pressure is defined as force per unit area, so the total effect of all these collisions is what we measure as pressure.
Two factors determine the pressure:
- The **frequency** of collisions with the walls (how many per second)
- The **force** of each individual collision
When temperature increases, particles move faster, so they collide with the walls **more frequently** and with **greater force**, resulting in higher pressure.
**Critical exam language**: Always specify "collisions with the **walls of the container**" — not just "collisions between particles." The examiner mark scheme specifically requires this distinction. Similarly, write "more **frequent** collisions" rather than just "more collisions."
---
### Concept 4: Boyle's Law — pV = constant
Boyle's Law describes the relationship between pressure and volume for a **fixed mass of gas at constant temperature**. It states that pressure and volume are **inversely proportional**: when one increases, the other decreases proportionally.
**Formula**: pV = constant, or equivalently: **p₁V₁ = p₂V₂**
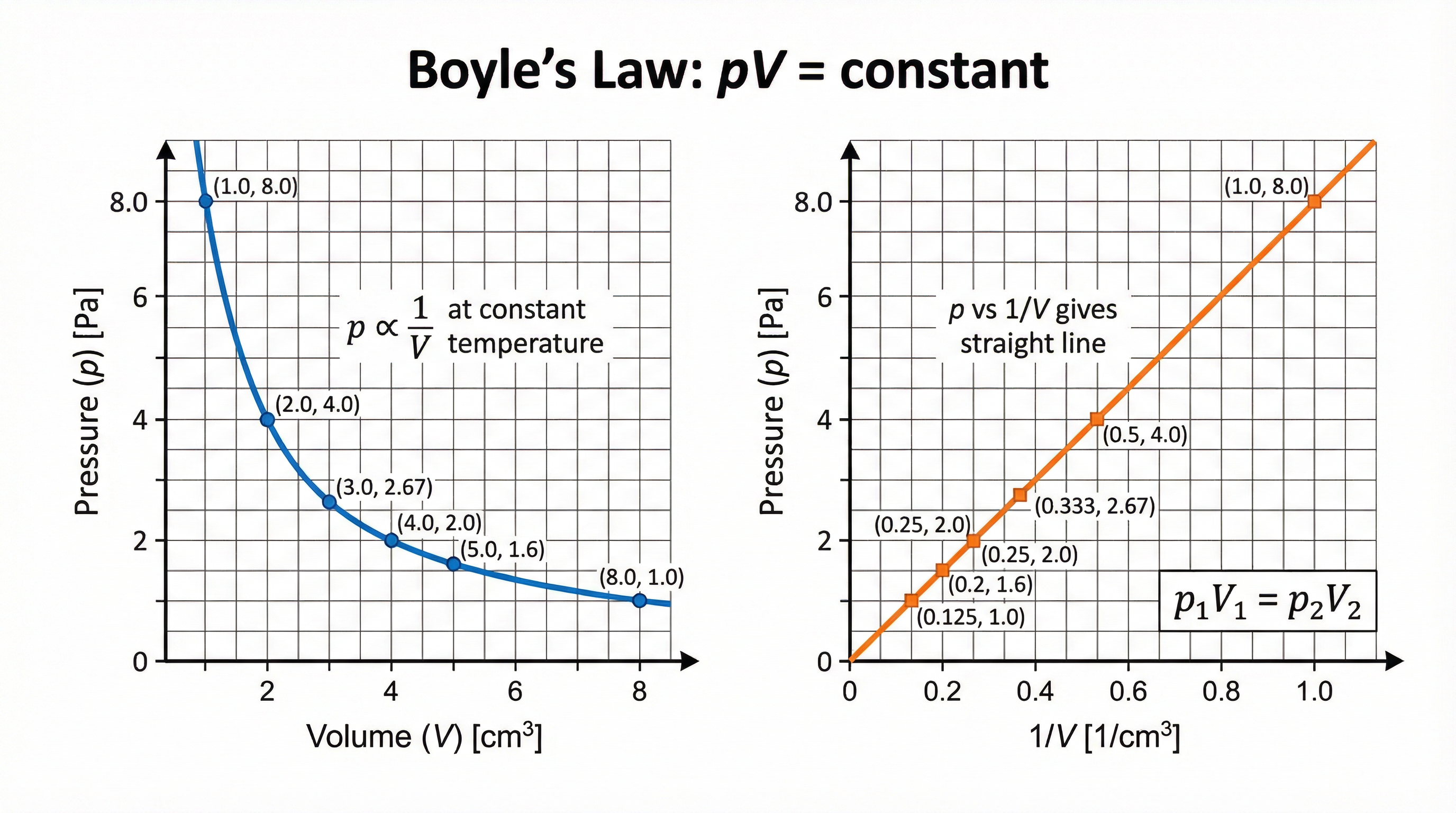
**Physical explanation**: If you compress a gas (reduce its volume), the same number of particles are now in a smaller space. They hit the walls more frequently, so pressure increases. If you expand the volume, particles have more space, hit the walls less often, and pressure decreases. The product p × V always stays the same.
**Unit guidance**: When using p₁V₁ = p₂V₂, you do **not** need to convert units provided you use the same units on both sides of the equation. For example, if p₁ is in kPa, then p₂ must also be in kPa. If V₁ is in cm³, then V₂ must also be in cm³. However, if the question specifically asks for an answer in Pa or m³, you must convert.
**Common error**: Candidates frequently attempt to divide pressure by volume (p/V) rather than multiply (p × V). Remember: Boyle's Law is a **product** — multiply, do not divide.
---
### Concept 5: Doing Work on a Gas (Higher Tier)
When a gas is compressed by an external force — such as pushing the plunger of a bicycle pump — **work is done on the gas**. In physics, doing work is a mechanism of energy transfer. The mechanical work done by the person pushing the plunger is transferred to the **internal energy store** of the gas.
This increase in internal energy means the particles move faster on average — their kinetic energy increases — which means the **temperature of the gas rises**.
**The bicycle pump example**: When you pump up a tyre, the pump barrel gets warm. This is not because the air outside is warm — it is because you are doing work on the gas inside, transferring energy to it and raising its temperature. This is a direct, real-world demonstration of the first law of thermodynamics at GCSE level.
**Exam answer structure for Higher Tier**: "Pushing the plunger does work on the gas → energy is transferred to the gas's internal energy store → the average kinetic energy of the particles increases → the temperature of the gas rises."
---
## Mathematical Relationships
| Formula | Meaning | Status | When to Use |
|---|---|---|---|
| pV = constant | Boyle's Law (single state) | **Must memorise** | Confirming Boyle's Law applies |
| p₁V₁ = p₂V₂ | Boyle's Law (two states) | **Must memorise** | Calculating new pressure or volume after a change |
| p = F/A | Definition of pressure | Given on formula sheet | Converting between force, pressure, and area |
**Step-by-step method for Boyle's Law calculations:**
1. Write down p₁, V₁, and the known value from the new state (either p₂ or V₂).
2. Write the equation: p₁V₁ = p₂V₂
3. Substitute the known values.
4. Rearrange to find the unknown.
5. Check: if volume decreased, pressure should increase (and vice versa). If not, you have made an error.
6. State the answer with correct units.
---
## Practical Applications
**Bicycle tyres and pumps**: Pumping up a tyre compresses air into a smaller volume, increasing pressure. The pump getting warm demonstrates the Higher Tier concept of doing work on a gas.
**Aerosol cans in the sun**: A sealed aerosol left in direct sunlight heats up. The gas inside gains kinetic energy, particles collide with the can walls more frequently and forcefully, and pressure increases — potentially to dangerous levels. This is why aerosols carry warnings not to expose them to heat.
**Scuba diving**: As a diver descends, water pressure increases. The gas in the diver's lungs (and equipment) is subject to higher external pressure. Boyle's Law governs how the volume of gas in the equipment changes with depth.
**Weather balloons**: As a weather balloon ascends, atmospheric pressure decreases. By Boyle's Law, the volume of gas inside the balloon increases — which is why weather balloons expand as they rise and eventually burst at high altitude.