States of Matter — AQA GCSE Study Guide
Exam Board: AQA | Level: GCSE
Master the particle model for your AQA GCSE Physics exam. This guide breaks down solids, liquids, and gases, explains the energy changes during melting and boiling, and gives you the tools to tackle tricky density calculations for guaranteed marks.

## Overview
Welcome to the study of States of Matter, a fundamental topic in physics that explains the world around us. In this section (AQA 4.3.2), we explore the **Particle Model of Matter**, a powerful tool for understanding the properties of solids, liquids, and gases. You'll learn how the arrangement and energy of particles determine a substance's state and why, for example, a puddle evaporates or an ice cube melts. This topic is crucial as it forms the basis for understanding energy transfer, pressure, and thermal physics. Examiners frequently test these concepts through data analysis of heating curves and structured 6-mark questions on density practicals, so a solid grasp is essential for a high grade in Paper 1.

## Key Concepts
### Concept 1: The Particle Model of Matter
The particle model states that all matter is made of tiny, constantly moving particles. The energy and forces between these particles determine the state of the matter. It's a simple but powerful idea that explains the macroscopic properties we observe.
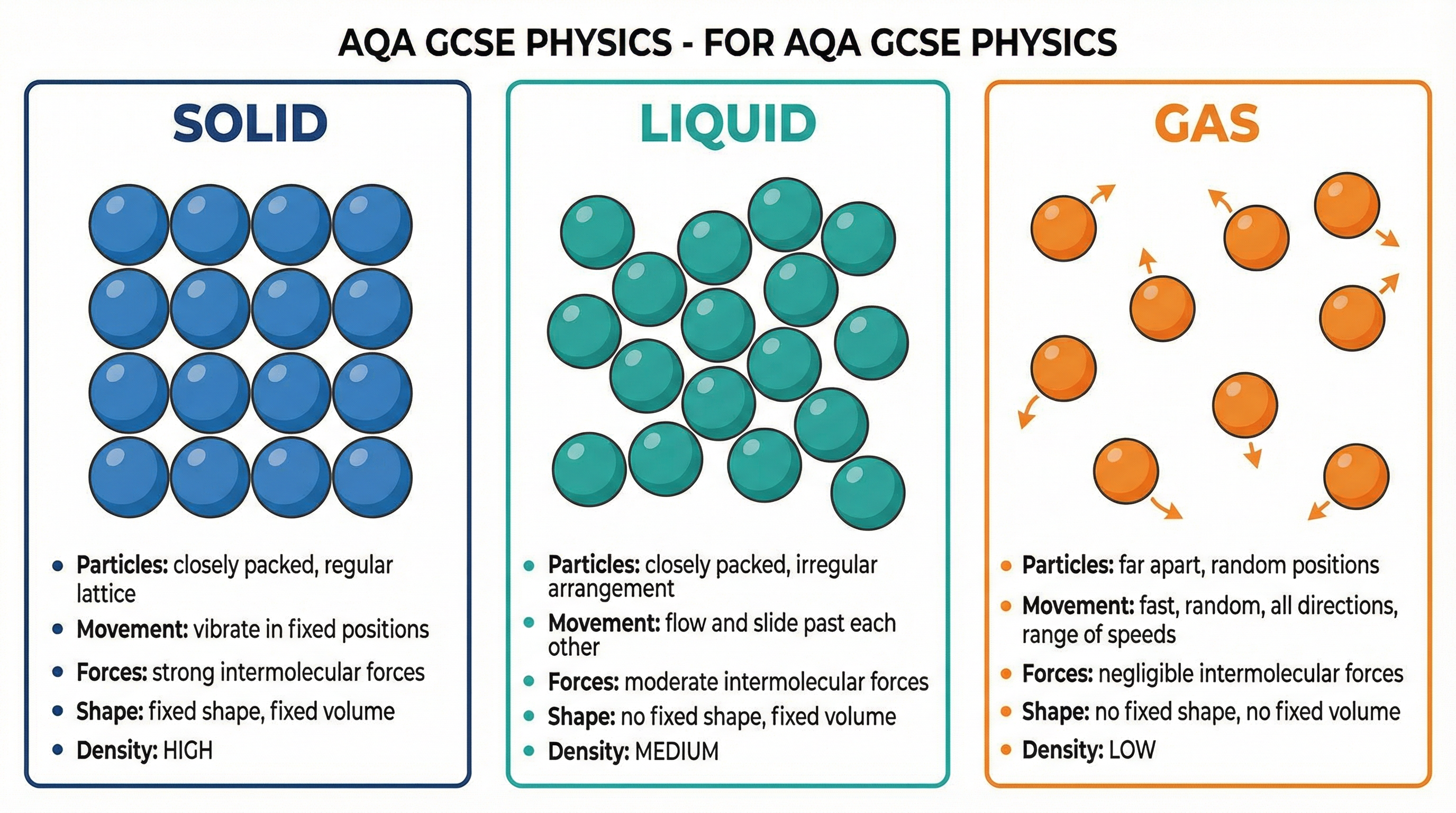
* **Solids**: Particles are held in a fixed, regular lattice. They are closely packed and vibrate about fixed positions. The forces of attraction between particles are very strong, which is why solids have a fixed shape and volume.
* **Liquids**: Particles are closely packed but are in an irregular arrangement. They can move and slide past one another, allowing liquids to flow and take the shape of their container. The forces between particles are weaker than in solids but still strong enough to keep the particles touching, giving liquids a fixed volume.
* **Gases**: Particles are far apart and move quickly and randomly in all directions, with a range of speeds. The forces between particles are negligible. This is why gases have no fixed shape or volume and will expand to fill any container.
**Examiner's Note**: A common mistake is to say liquid particles are 'far apart'. This is incorrect. Credit is only given for descriptions like 'closely packed' or 'touching' in an irregular arrangement.
### Concept 2: Internal Energy, Temperature, and State Changes
**Internal energy** is a crucial concept defined as **the total kinetic energy and potential energy of all the particles within a system**. It's the sum of all the microscopic energy stores.
* **Kinetic Energy (KE)**: This is the energy of movement. The faster the particles vibrate, move, or slide, the higher their kinetic energy. **Temperature is a measure of the average kinetic energy of the particles.** So, if the temperature of a substance increases, its particles are, on average, moving faster.
* **Potential Energy (PE)**: This is the energy stored in the bonds and forces between particles. To move particles further apart and overcome these intermolecular forces, energy is required. This energy increases the potential energy store.
When a substance is heated, its internal energy increases. This energy can either increase the kinetic energy of the particles (raising the temperature) OR increase the potential energy of the particles (changing the state), but not both at the same time.
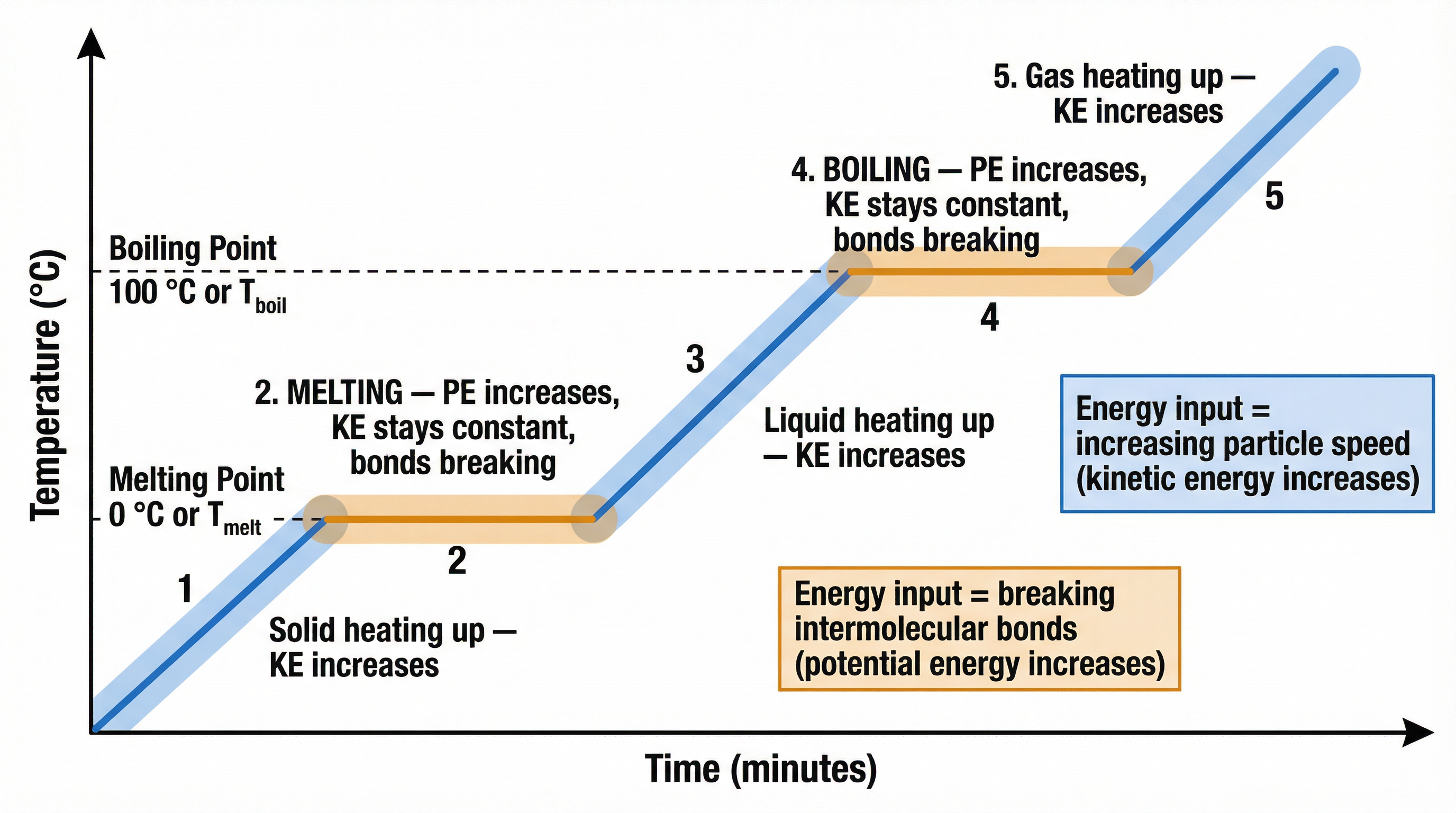
This is best shown by a **heating curve**. When a solid is heated, its temperature rises as particles gain kinetic energy. At the melting point, the temperature stops rising. The energy being supplied (the latent heat of fusion) is used to break the bonds holding the particles in their fixed lattice, increasing their potential energy. Once all the solid has melted, the temperature of the liquid begins to rise again. The same process occurs at the boiling point, where the energy supplied (the latent heat of vaporisation) is used to overcome the remaining forces and separate the particles completely into a gas.
### Concept 3: Density
Density is a measure of how much 'stuff' (mass) is packed into a certain amount of space (volume). A substance is dense if it has a lot of mass for its volume.
**Example**: A kilogram of lead is much smaller than a kilogram of feathers. This is because lead is much denser.
Generally, solids are the densest state, as the particles are packed most closely together. Liquids are slightly less dense. Gases are the least dense by a large margin, as there are huge spaces between the particles. A key exception is water, which is densest as a liquid at 4°C, not as a solid (ice floats).
## Mathematical/Scientific Relationships
### Density Formula (Must memorise)
**ρ = m / V**
* **ρ** (rho) = density, measured in kilograms per cubic metre (kg/m³)
* **m** = mass, measured in kilograms (kg)
* **V** = volume, measured in cubic metres (m³)

### Specific Latent Heat Formula (Given on formula sheet)
**E = m L**
* **E** = Energy for a change of state, measured in Joules (J)
* **m** = mass, measured in kilograms (kg)
* **L** = Specific Latent Heat, measured in Joules per kilogram (J/kg). This is specific to the substance and the change of state (fusion for melting/freezing, vaporisation for boiling/condensing).
## Required Practical: Determining Density
This is a common 6-mark question. You need to be able to describe how to find the density of a regular solid, an irregular solid, and a liquid.
**Apparatus**: Digital balance, ruler, Eureka (displacement) can, measuring cylinder, water.
**Method for a Regular Solid (e.g., a cube)**:
1. **Measure Mass**: Place the object on a digital balance and record its mass in kg.
2. **Measure Volume**: Use a ruler to measure the length, width, and height in metres. Calculate the volume (V = l × w × h).
3. **Calculate Density**: Use ρ = m / V.
**Method for an Irregular Solid (e.g., a rock)**:
1. **Measure Mass**: Place the object on a digital balance and record its mass in kg.
2. **Measure Volume**: Fill a Eureka can with water until it flows from the spout. Place an empty measuring cylinder under the spout. Carefully lower the object into the can. The volume of water displaced into the measuring cylinder is equal to the volume of the object. Record this volume and convert to m³ (1 ml = 1 cm³, 1,000,000 cm³ = 1 m³).
3. **Calculate Density**: Use ρ = m / V.
**Common Errors**: Forgetting to zero the balance; misreading the measuring cylinder scale (read from the bottom of the meniscus); splashing water when lowering the object.