Density and States of Matter — Edexcel GCSE Study Guide
Exam Board: Edexcel | Level: GCSE
Master Edexcel GCSE Physics Topic 14.1: Density and States of Matter. This guide breaks down the essential particle model, the density formula (ρ = m/V), and the Core Practical for measuring density, providing examiner insights and memory hooks to help you secure top marks.
## Overview
Welcome to your deep dive into Density and States of Matter, a cornerstone of your Edexcel GCSE Physics course. This topic is all about the **particle model** — a powerful idea that explains the properties of solids, liquids, and gases by looking at how their tiny particles behave. You'll learn why a block of steel is denser than the same-sized block of wood, and what actually happens when ice melts into water. This isn't just abstract theory; it connects directly to other key areas like **Energy** (internal energy and latent heat) and **Forces** (pressure in gases). Examiners frequently test this topic through a mix of calculations, particle diagrams, and a 6-mark question on the **Core Practical** for measuring density. Mastering this topic means you'll be confident in tackling a wide range of question styles, from simple definitions to complex practical analysis.
## Key Concepts
### Concept 1: The Particle Model
The particle model is the central idea that all matter is made of tiny, constantly moving particles. The arrangement and energy of these particles determine whether a substance is a solid, liquid, or gas. Examiners expect you to describe and draw these arrangements with precision.
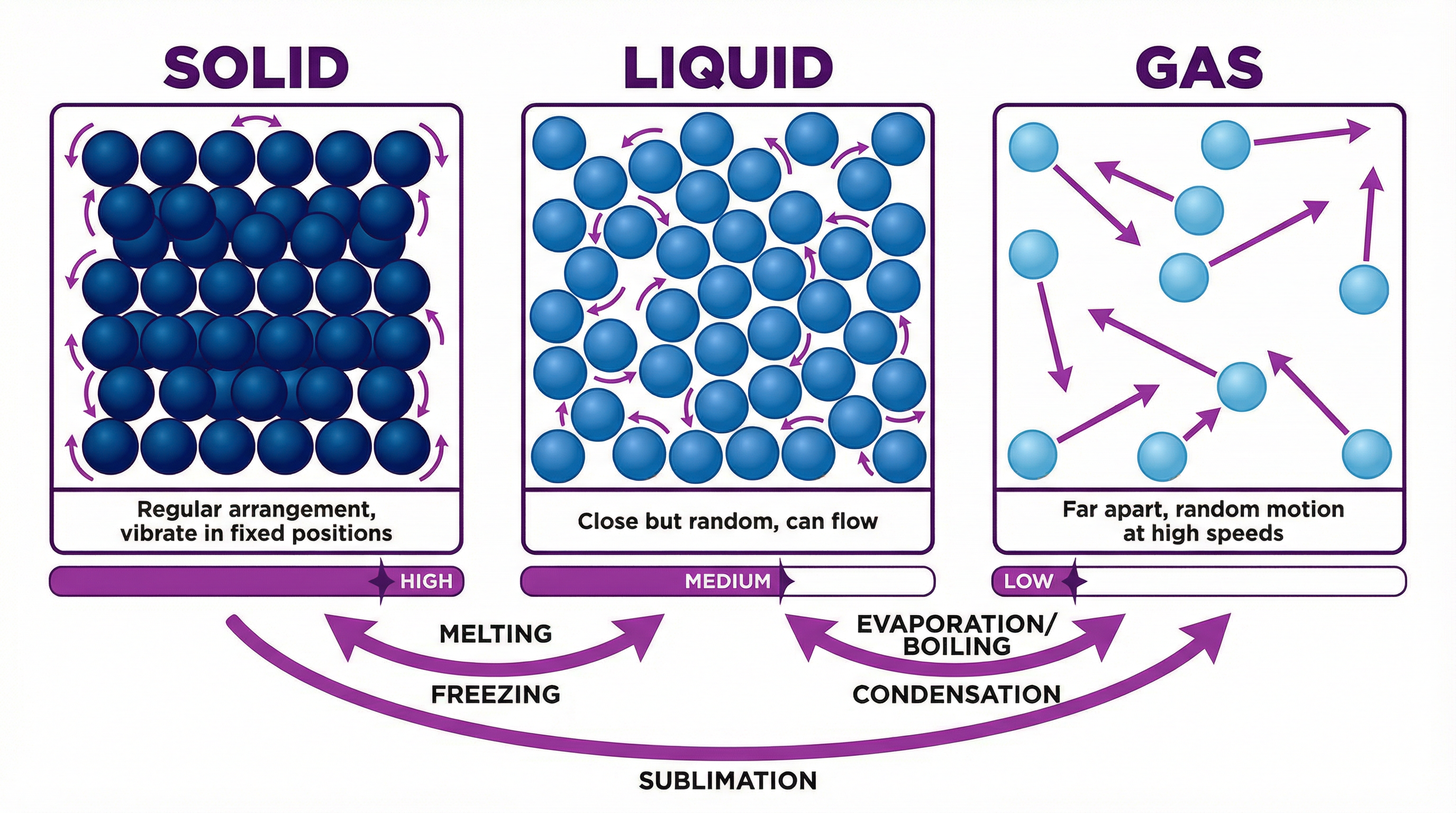
* **Solids**: Particles are arranged in a **regular lattice structure**. They are held in **fixed positions** by strong intermolecular forces and can only **vibrate**. Because the particles are packed tightly together, solids have a **high density**.
* **Liquids**: Particles are **close together but randomly arranged**. The forces between them are weaker than in solids, allowing them to **move past each other**. This is why liquids can flow. Their density is typically slightly lower than that of their solid form (a key exception being water).
* **Gases**: Particles are **far apart** with **negligible forces** between them. They move **randomly and at high speeds**. Because of the large spaces between particles, gases have a **very low density** and can be easily compressed.
### Concept 2: Density
Density is a measure of how much 'stuff' (mass) is crammed into a certain amount of space (volume). It's a fundamental property of a material.
> **Definition**: Density is defined as mass per unit volume. A substance with a high density has a lot of mass packed into a small volume.
**Example**: A block of aluminium has a mass of 540g and a volume of 200 cm³. Its density is 540g / 200cm³ = 2.7 g/cm³.
### Concept 3: Changes of State & Internal Energy
When a substance is heated, its particles gain **internal energy** — the total kinetic energy (from their movement) and potential energy (from the bonds between them). This can lead to a change of state.
* **Melting & Boiling**: When energy is supplied, the potential energy of the particles increases, weakening or breaking the bonds holding them together. During a change of state, the temperature remains constant because the energy is being used to change the state (latent heat), not to increase the kinetic energy of the particles.
* **Conservation of Mass**: Crucially, during a physical change of state, **mass is conserved**. The number of particles remains the same; they just rearrange themselves. If you melt 1kg of ice, you get 1kg of water.
* **Physical vs. Chemical Changes**: Changes of state are **physical changes** because they are **reversible**. No new substances are formed. This is a key distinction that can be tested.
## Mathematical/Scientific Relationships
### Density Formula
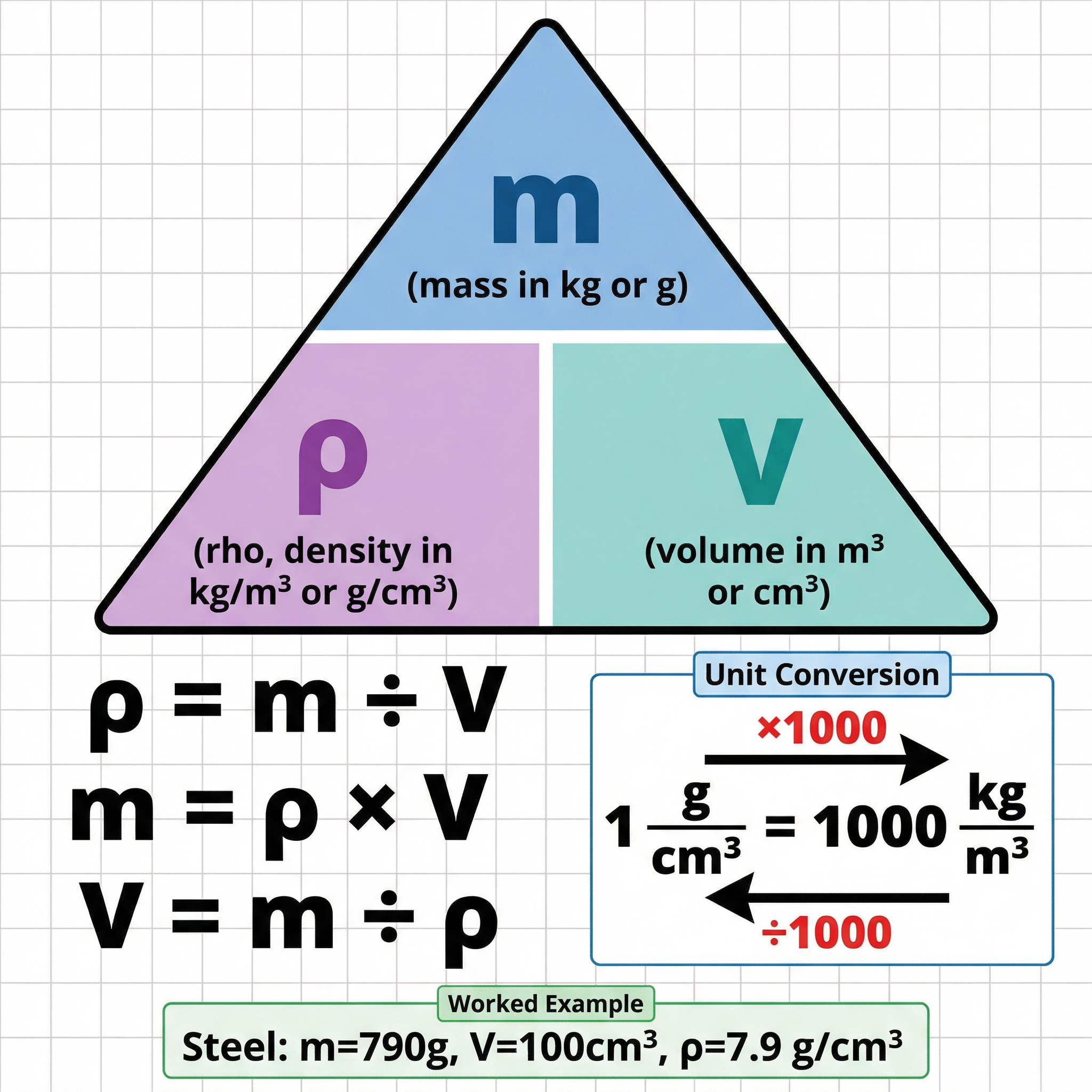
* **Formula**: `ρ = m / V`
* **ρ (rho)**: Density (kg/m³ or g/cm³)
* **m**: Mass (kg or g)
* **V**: Volume (m³ or cm³)
* **Status**: **Given on formula sheet**. You must be able to rearrange it.
### Unit Conversions
* **g/cm³ to kg/m³**: Multiply by 1000. (e.g., 2 g/cm³ = 2000 kg/m³)
* **kg/m³ to g/cm³**: Divide by 1000. (e.g., 800 kg/m³ = 0.8 g/cm³)
* **Status**: **Must memorise**. This is a very common source of error.
## Required Practical: Measuring Density (Core Practical 14.3)
This is a guaranteed area for exam questions, including potential 6-mark questions. You need to know the method for both regular and irregular solids.
**Apparatus List**:
* Balance (for measuring mass)
* Ruler or Vernier callipers (for regular solids)
* Eureka (displacement) can
* Measuring cylinder
* String
* The objects to be measured
### Method for an Irregular Solid
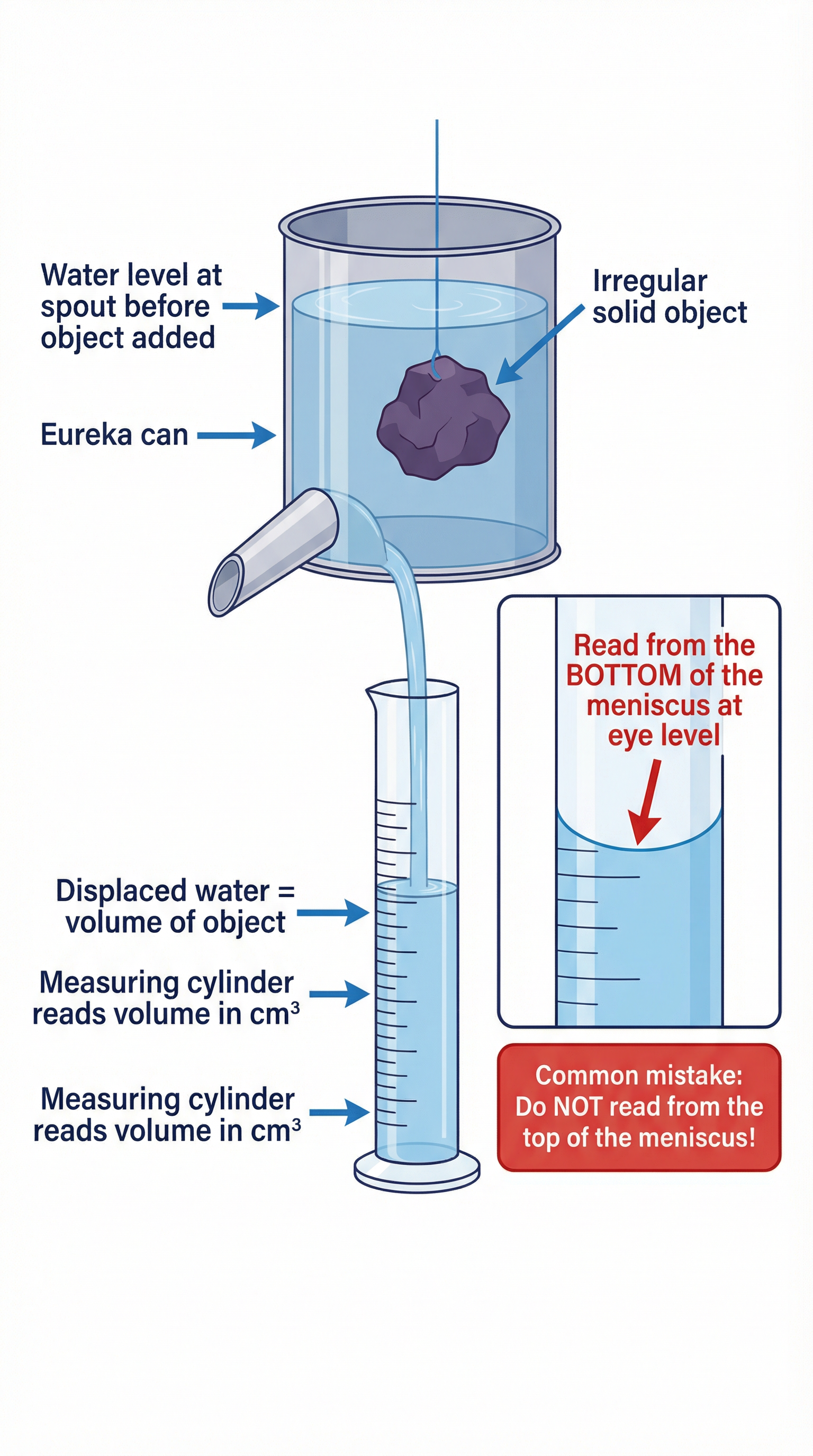
1. **Measure Mass**: Place the dry irregular object on a balance and record its mass (m).
2. **Prepare for Displacement**: Fill the Eureka can with water until it just starts to trickle out of the spout. Wait for it to stop dripping.
3. **Collect Displaced Water**: Place an empty measuring cylinder directly under the spout.
4. **Measure Volume**: Carefully lower the object into the Eureka can using a piece of string until it is fully submerged. The volume of water that overflows into the measuring cylinder is equal to the volume of the object (V).
5. **Read the Volume**: Read the volume from the measuring cylinder at eye level from the bottom of the meniscus.
6. **Calculate Density**: Use the formula `ρ = m / V` to calculate the density.
### Method for a Regular Solid (e.g., a Cube)
1. **Measure Mass**: Place the cube on a balance and record its mass (m).
2. **Measure Dimensions**: Use a ruler to measure the length, width, and height of the cube. For greater precision, use Vernier callipers and take multiple readings to calculate an average.
3. **Calculate Volume**: Calculate the volume using the formula `Volume = length × width × height`.
4. **Calculate Density**: Use the formula `ρ = m / V` to calculate the density.
