Nuclear and Particle Physics — OCR A-Level Study Guide
Exam Board: OCR | Level: A-Level
Master OCR A-Level Physics Module 6.4: Nuclear and Particle Physics. This guide deconstructs the Standard Model, radioactive decay, and binding energy, providing examiner insights and multi-modal resources to help you secure top marks on complex quantitative and qualitative questions.

## Overview
Welcome to one of the most fundamental and revealing areas of modern physics. OCR A-Level Physics Module 6.4, Nuclear and Particle Physics, takes us on a journey from the familiar atomic nucleus into the strange and wonderful subatomic world of quarks, leptons, and bosons. This topic is crucial because it underpins our understanding of matter itself, the forces that govern it, and the processes that power stars and nuclear reactors. In the exam, you will face a mix of challenging quantitative problems (like decay modelling and binding energy calculations) and qualitative questions that test your precise understanding of the Standard Model and conservation laws. Candidates who can navigate these concepts with fluency, applying conservation laws rigorously and handling mass-energy conversions with precision, are consistently rewarded with high marks. This guide will equip you with the conceptual framework and exam technique to do just that.

## Key Concepts
### Concept 1: The Nucleus & Radioactive Decay
The atomic nucleus is comprised of **protons** and **neutrons**, collectively known as **nucleons**. An atom's identity is its proton number (Z), while its mass number (A) is the total count of nucleons. Nuclei of the same element with different numbers of neutrons are called **isotopes**.
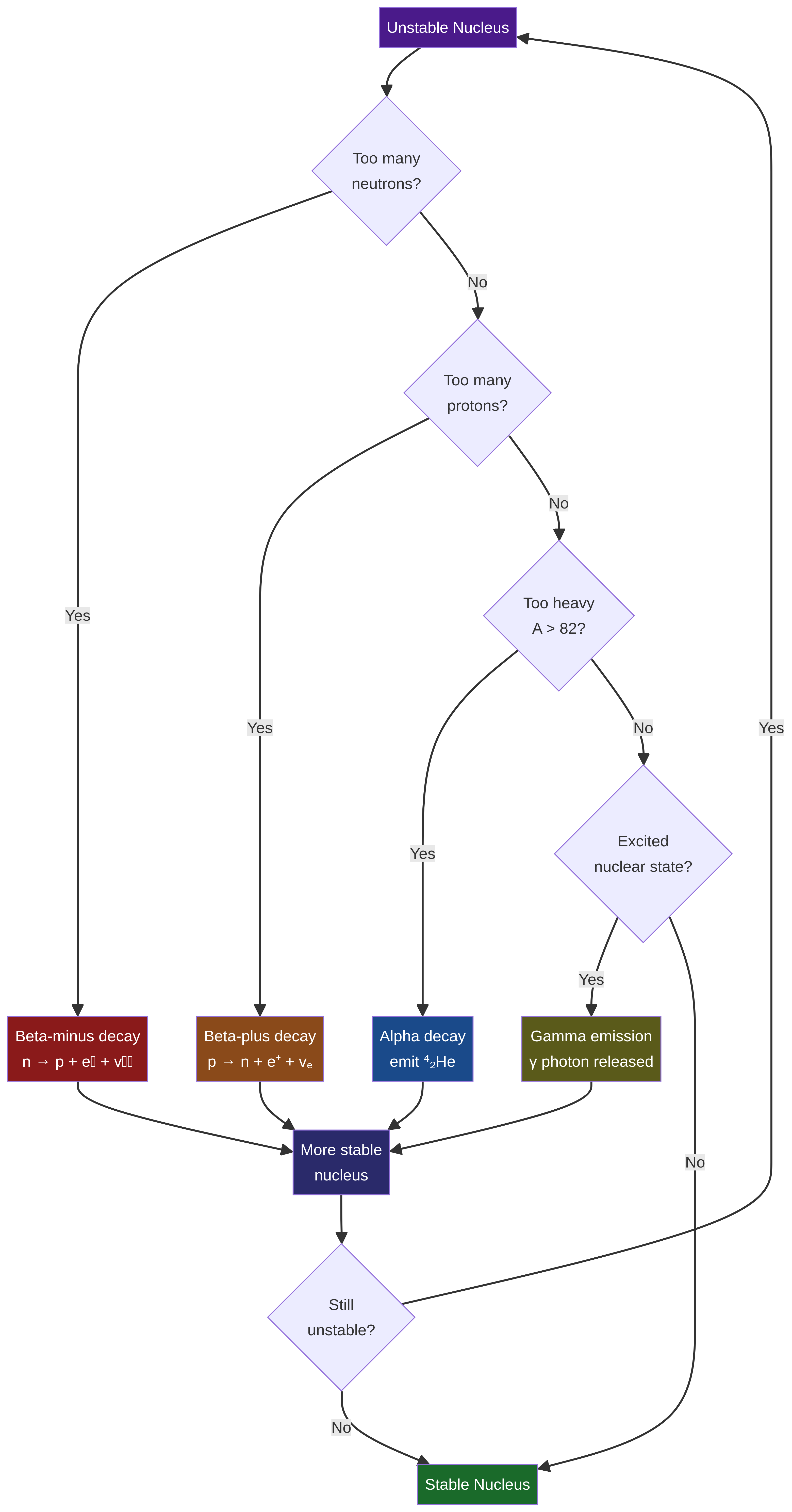
Many nuclei are unstable. They possess an imbalance of neutrons and protons or are simply too large to be held together by the strong nuclear force. To achieve a more stable, lower-energy state, they undergo **radioactive decay**. It's crucial to understand that this process is both **spontaneous** (unaffected by external factors like temperature or pressure) and **random** (it's impossible to predict when a specific nucleus will decay, but there is a constant probability of decay per unit time). Examiners frequently test the distinction between these two terms.
* **Alpha (α) Decay**: Occurs in very heavy nuclei. An alpha particle (a Helium nucleus, ⁴₂He) is emitted. The result is that A decreases by 4 and Z decreases by 2.
* **Beta-minus (β⁻) Decay**: Occurs in neutron-rich nuclei. A neutron transforms into a proton, emitting a high-energy electron (the β⁻ particle) and an **electron antineutrino (v̄ₑ)**. The antineutrino is essential for conserving lepton number and is a common omission in exam answers, leading to lost marks. A increases by 1, Z increases by 1.
* **Beta-plus (β⁺) Decay**: Occurs in proton-rich nuclei. A proton transforms into a neutron, emitting a positron (the electron's antiparticle, e⁺) and an **electron neutrino (vₑ)**. A decreases by 1, Z decreases by 1.
* **Gamma (γ) Emission**: An excited nucleus releases surplus energy as a high-frequency photon (gamma ray). There is no change in A or Z; the nucleus simply moves to a lower energy state.
### Concept 2: The Mathematics of Decay
Radioactive decay is governed by a key exponential relationship. The number of undecayed nuclei, N, at time t is given by:
`N = N₀ * e^(-λt)`
Where `N₀` is the initial number of undecayed nuclei and `λ` is the **decay constant**. The decay constant represents the probability that an individual nucleus will decay per unit time. The **activity (A)** of a sample, measured in Becquerels (Bq), is the rate of decay:
`A = λN`
This means activity also follows the same exponential decay pattern: `A = A₀ * e^(-λt)`.
A related and more intuitive concept is **half-life (T₁/₂)**, the time taken for half of the undecayed nuclei in a sample to decay (or for the activity to halve). It is related to the decay constant by:
`T₁/₂ = ln(2) / λ ≈ 0.693 / λ`
**Exam Skill**: A common high-mark question involves determining the decay constant from data. Plotting `ln(A)` against `t` produces a straight line with a gradient of `-λ`. Candidates must explicitly state this relationship to gain full credit.
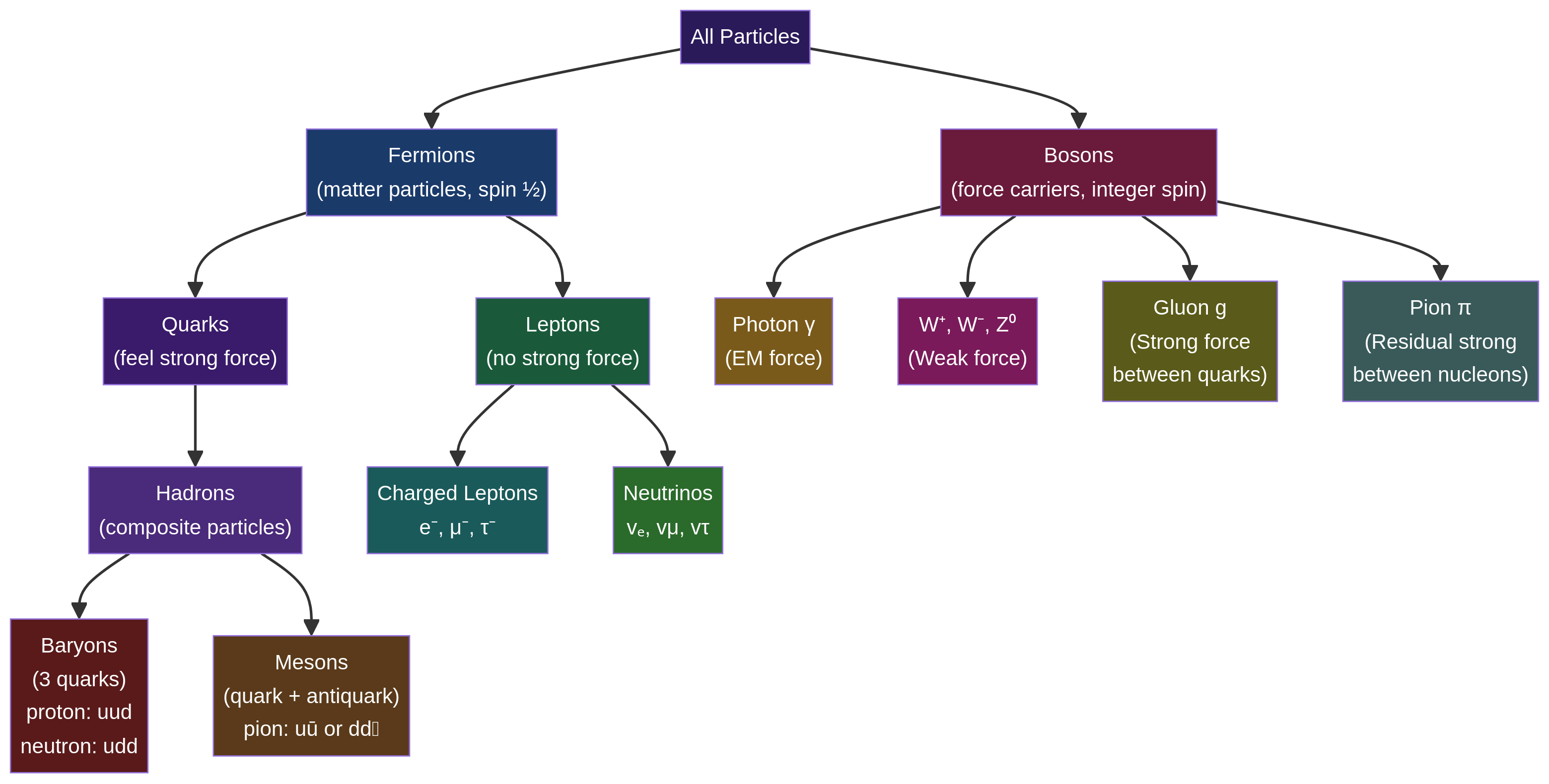
### Concept 3: The Standard Model of Particle Physics
The Standard Model is the physicist's periodic table, classifying all known fundamental particles. It is a cornerstone of the specification.

Particles are divided into two main families:
* **Fermions**: The 'matter' particles. They have half-integer spin and are subdivided into **quarks** and **leptons**.
* **Quarks**: Experience the strong nuclear force. They come in six 'flavours': up (u), down (d), charm (c), strange (s), top (t), and bottom (b). They always exist in composite particles called **hadrons**.
* **Baryons**: Made of three quarks (e.g., Proton: uud, Neutron: udd). Baryon Number = +1.
* **Mesons**: Made of one quark and one antiquark (e.g., Pion: uđ). Baryon Number = 0.
* **Leptons**: Do not experience the strong force. They include the electron (e⁻), muon (μ⁻), tau (τ⁻), and their corresponding neutrinos (vₑ, vμ, vτ). Each has a Lepton Number of +1.
* **Bosons**: The 'force-carrying' particles. They have integer spin.
* **Photon (γ)**: Mediates the electromagnetic force.
* **W⁺, W⁻, Z⁰ Bosons**: Mediate the weak nuclear force (responsible for beta decay).
* **Gluon (g)**: Mediates the strong nuclear force that binds quarks together inside hadrons.
* **Higgs Boson**: Responsible for giving particles mass.
### Concept 4: Conservation Laws
In any particle interaction, certain properties must be conserved. To validate a proposed interaction, you must check for the conservation of:
1. **Charge (Q)**
2. **Baryon Number (B)**
3. **Lepton Number (L)** (This is conserved separately for each family: Lₑ, Lμ, Lτ)
If any of these laws are violated, the interaction cannot happen. This is a standard template for many exam questions.
### Concept 5: Binding Energy and Mass Defect
The mass of a nucleus is always *less* than the sum of the masses of its individual, separate protons and neutrons. This difference in mass is called the **mass defect (Δm)**. The 'missing' mass has been converted into energy, known as the **binding energy (BE)**, which holds the nucleus together. This is a direct consequence of Einstein's mass-energy equivalence principle:
`BE = Δm * c²`
* **Mass Defect (Δm)** = (Total mass of separate nucleons) - (Mass of the nucleus)
For calculations, you can either convert the mass defect from atomic mass units (u) to kg and use `c²`, or more directly, use the conversion factor `1 u = 931.5 MeV`.
The **binding energy per nucleon** (BE/A) is a measure of a nucleus's stability. The higher this value, the more stable the nucleus.
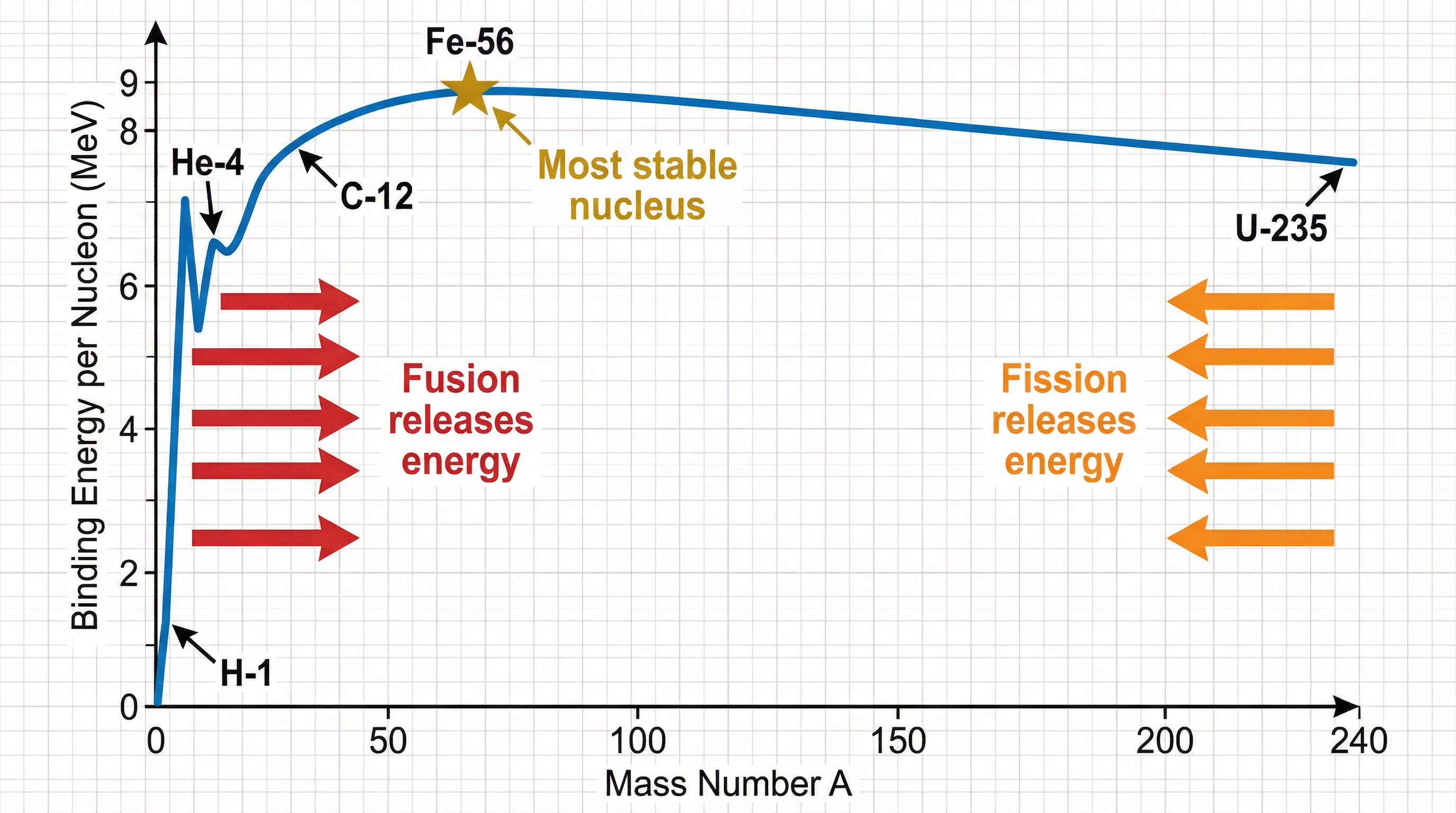
The binding energy curve shows that Iron-56 (Fe-56) has the highest binding energy per nucleon, making it the most stable nucleus. Energy is released when nuclei move towards this peak:
* **Fusion**: Light nuclei combine, increasing their binding energy per nucleon.
* **Fission**: A heavy nucleus splits, increasing the binding energy per nucleon of the fragments.
**Exam Skill**: In 'Show that' questions involving energy release, it is vital to carry intermediate mass values to at least 5 decimal places to avoid rounding errors that will cause your final answer to differ from the given value, resulting in a lost mark.