Waves — OCR A-Level Study Guide
Exam Board: OCR | Level: A-Level
Dive into the quantum world where light behaves like particles and particles behave like waves. This essential module covers the photoelectric effect, electron energy levels, and wave-particle duality — key concepts that bridge classical physics and quantum mechanics.

## Overview
Module 4: Electrons, Waves and Photons is a pivotal section of the A-Level Physics specification. It marks the transition from classical Newtonian physics to the counter-intuitive world of quantum mechanics. You will explore how light can behave as a stream of particles (photons) and how matter can behave as a wave.
This topic is fundamental because it explains phenomena that classical wave theory simply cannot — most notably, the photoelectric effect. It connects deeply with electricity (current and voltage), mechanics (momentum and kinetic energy), and even astrophysics (stellar spectra).
Examiners frequently test this module with multi-step calculation questions involving powers of ten, unit conversions (eV to Joules), and 'show that' questions requiring clear logical chains. Expect to be asked to explain *why* wave theory fails to explain the photoelectric effect, and to perform calculations linking energy, frequency, and wavelength.
## Key Concepts
### Concept 1: The Photon Model
Classical physics treats light purely as a continuous wave. However, to explain interactions with matter, we must model light as discrete packets of energy called **photons**.
The energy of a single photon is proportional to its frequency:
**E = hf**
Think of a beam of light not as a continuous stream of water, but as a stream of individual bullets. A brighter light (higher intensity) means *more* bullets per second, but it doesn't change the energy of each individual bullet. Changing the colour to blue or violet (higher frequency) increases the energy of *each individual bullet*.
**Example**: A laser emits red light with a wavelength of 650 nm. To find the energy of one photon, we first convert wavelength to metres ($650 \times 10^{-9}$ m), then use $E = hc/\lambda$. This gives $E = (6.63 \times 10^{-34} \times 3.00 \times 10^8) / (650 \times 10^{-9}) = 3.06 \times 10^{-19}$ J.
### Concept 2: The Photoelectric Effect
This is the phenomenon where electrons are emitted from a metal surface when electromagnetic radiation shines on it. Crucially, this only happens if the radiation is above a certain **threshold frequency ($f_0$)**.
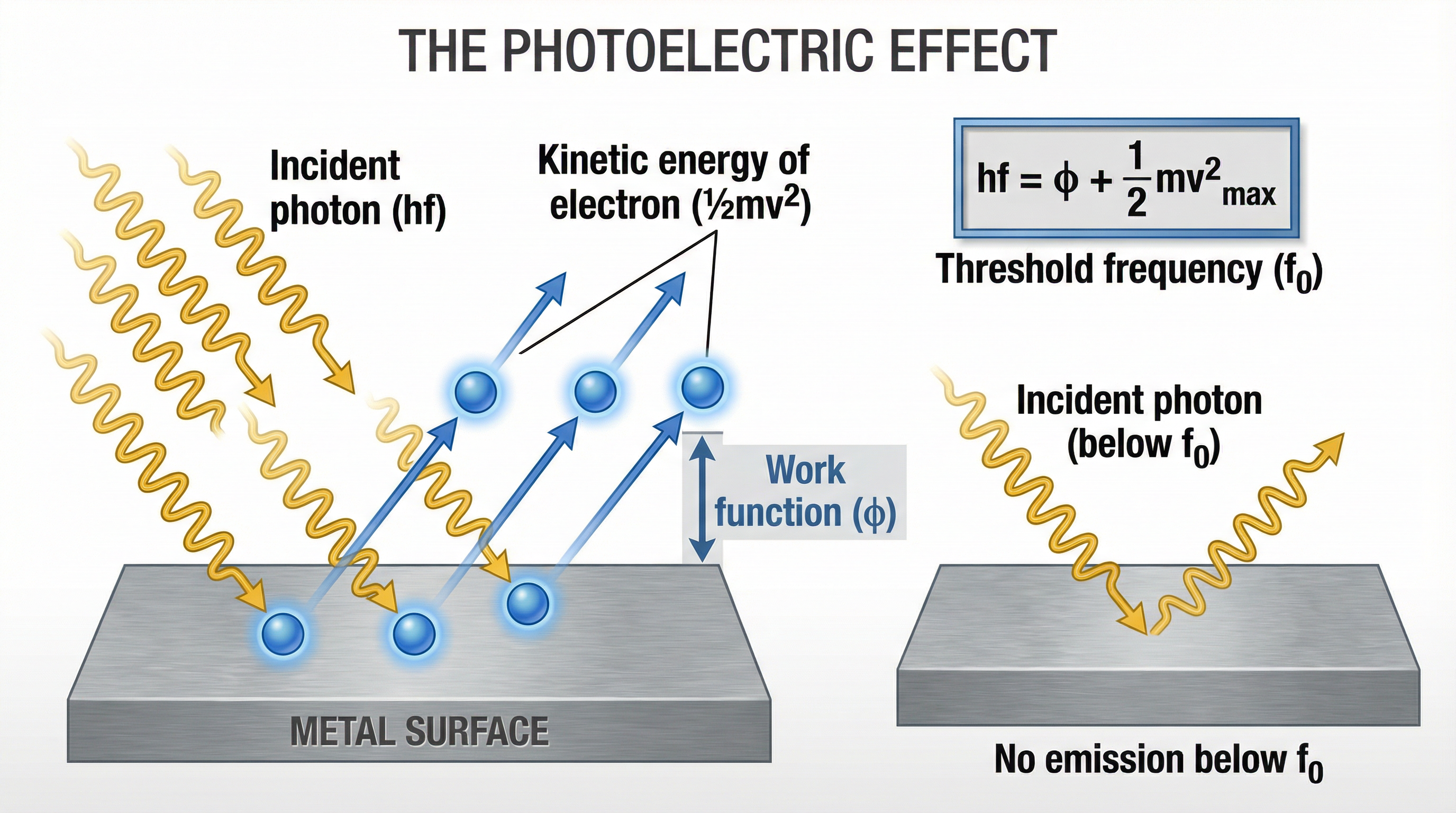
Why does this prove the particle nature of light?
- **Wave theory prediction**: Any light, if bright enough or shone for long enough, should eventually give electrons enough energy to escape.
- **Experimental reality**: If frequency is below $f_0$, *no* electrons are emitted, even with the brightest floodlight. Emission is instantaneous.
Einstein explained this by proposing a one-to-one interaction: **one photon transfers all its energy to one electron**. If that photon's energy ($hf$) is less than the energy needed to escape (the work function, $\phi$), the electron stays put.
### Concept 3: Wave-Particle Duality
We have seen light behaving as a particle. But we know light also diffracts and interferes — classic wave behaviours. This is **wave-particle duality**: light exhibits both wave-like and particle-like properties depending on the experiment.
In 1924, Louis de Broglie proposed that if waves can behave like particles, particles (like electrons) should behave like waves.
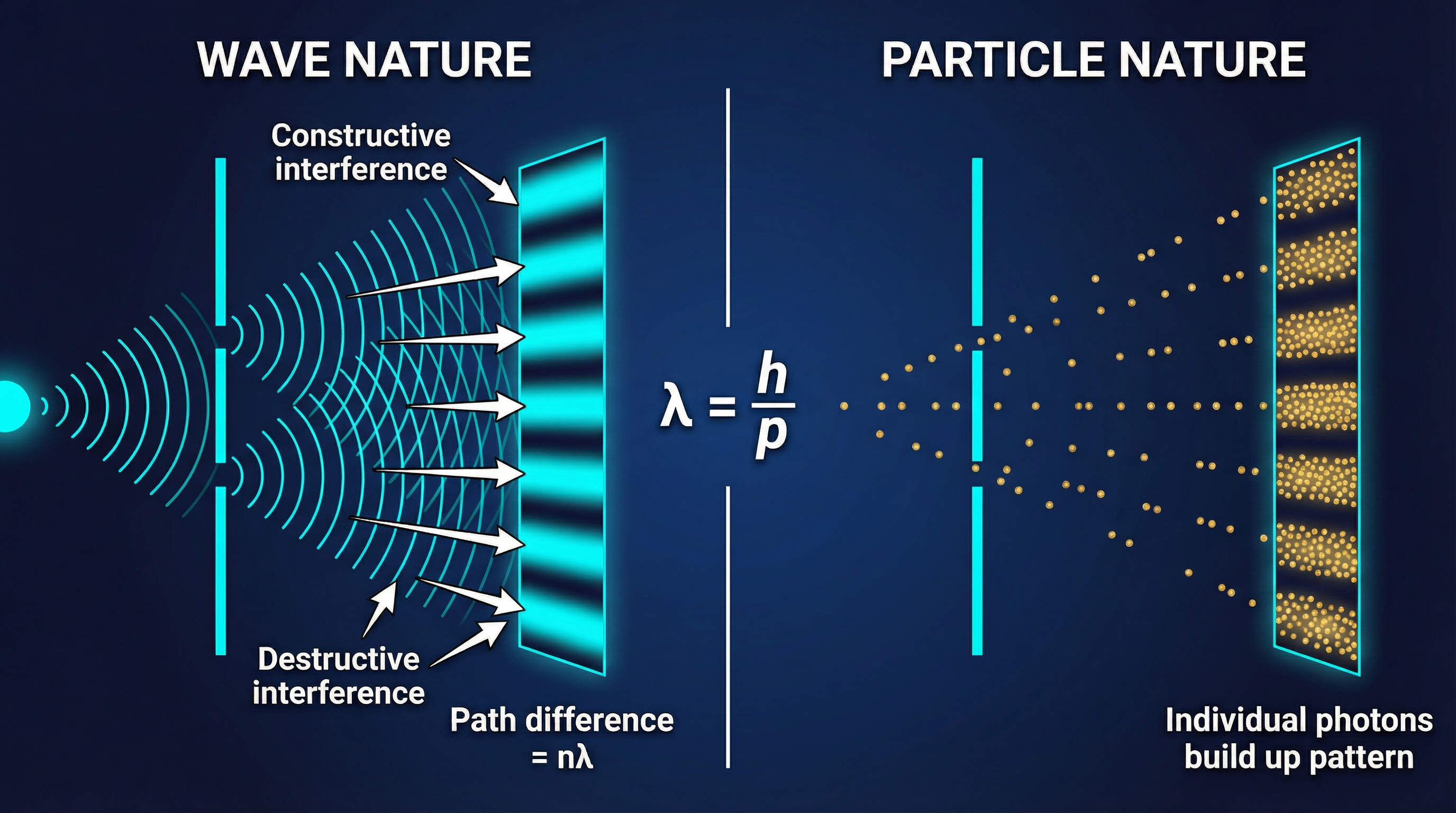
This was confirmed when electrons were fired at a crystal lattice and produced a diffraction pattern — something only waves do. The wavelength of a particle is inversely proportional to its momentum.
## Mathematical/Scientific Relationships
### 1. Photon Energy
$$E = hf = \frac{hc}{\lambda}$$
- $h$ = Planck's constant ($6.63 \times 10^{-34}$ J s)
- $c$ = speed of light ($3.00 \times 10^8$ m/s)
- **Tip**: Always convert $\lambda$ to metres (nm $\to \times 10^{-9}$ m).
### 2. Einstein's Photoelectric Equation
$$hf = \phi + \frac{1}{2}mv^2_{max}$$
- $hf$ = Energy of incident photon
- $\phi$ = Work function (minimum energy to escape surface)
- $\frac{1}{2}mv^2_{max}$ = Maximum kinetic energy of emitted electron
- **Rearranged**: $KE_{max} = hf - \phi$ (resembles $y = mx + c$ for graphical analysis)
### 3. Threshold Frequency
At the threshold, kinetic energy is zero, so:
$$hf_0 = \phi$$
### 4. The Electronvolt (eV)
Energy on the atomic scale is often given in eV.
$$1 \text{ eV} = 1.60 \times 10^{-19} \text{ J}$$
- To convert eV $\to$ J: Multiply by $1.60 \times 10^{-19}$
- To convert J $\to$ eV: Divide by $1.60 \times 10^{-19}$
### 5. De Broglie Wavelength
$$\lambda = \frac{h}{p} = \frac{h}{mv}$$
- $p$ = momentum ($kg \cdot m/s$)
- $m$ = mass ($9.11 \times 10^{-31}$ kg for electron)
- $v$ = velocity (m/s)
## Practical Applications
- **Photocells**: Used in solar panels and light sensors (e.g., automatic doors). They rely on the photoelectric effect to convert light energy directly into electrical current.
- **Electron Microscopes**: Use the wave nature of electrons. Because electrons can be accelerated to high speeds, their de Broglie wavelength is thousands of times shorter than visible light, allowing us to resolve tiny structures like viruses that are invisible to optical microscopes.
- **Fluorescent Tubes**: Use excitation and de-excitation of electrons in mercury vapour to produce UV light, which is then absorbed by a phosphor coating to emit visible light.
