Fossil Fuels — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
Fossil fuels — coal, oil, and natural gas — are the dominant global energy source, yet their combustion drives climate change through CO2 emissions and causes acid rain through SO2. OCR examiners assess candidates on their ability to evaluate the trade-offs between fossil fuels' unmatched reliability and energy density against their serious environmental and sustainability drawbacks, making this a high-value topic for both Foundation and Higher tier candidates.

## Overview
Fossil fuels sit at the heart of the global energy debate, and OCR examiners know it. Topic 5.7 requires candidates to move beyond simple description and into genuine scientific evaluation — weighing the advantages of coal, oil, and gas against their profound environmental consequences. This topic connects directly to the Physics of energy transfer, the electromagnetic spectrum (infrared radiation), and the broader themes of sustainability and global resource management. In the exam, candidates can expect questions ranging from 1-mark definitions to 6-mark extended response questions demanding structured evaluation. Command words to watch for include **State**, **Explain**, **Compare**, and — most importantly — **Evaluate**. Mastering this topic means understanding not just what fossil fuels are, but why societies still use them, what the precise consequences of combustion are, and how to communicate that understanding in the language of a senior examiner.

## Key Concepts
### Concept 1: Non-Renewable Resources and Their Formation
Fossil fuels are classified as **non-renewable resources** — a term with a precise, mark-earning definition. A non-renewable resource is one that **is being used faster than it can be replenished**. This definition is critical: it is not sufficient to state that fossil fuels will "run out"; candidates must convey the disparity in rates. Coal, oil, and natural gas formed over hundreds of millions of years from the compressed and chemically altered remains of ancient organisms — marine plankton in the case of oil and gas, and ancient forests in the case of coal. Human civilisation burns these resources in decades. The rate of consumption vastly exceeds any natural rate of formation, making replenishment within any meaningful human timescale impossible.
A common and costly error is to describe renewable energy as something that can be "re-used." This is scientifically incorrect. Fuel undergoes an irreversible chemical change during combustion — bonds are broken, new molecules are formed, and the original fuel cannot be recovered. The correct term is **replenished**: renewable energy sources are naturally restored by ongoing processes (sunlight, wind, rainfall) at a rate sufficient to sustain continued use.
**The three fossil fuels candidates must know**: Coal, Oil (petroleum), and Natural Gas (predominantly methane, CH₄).
### Concept 2: Combustion and Environmental Consequences
When fossil fuels burn, they react with oxygen in the air. The primary products of complete combustion are carbon dioxide (CO₂) and water (H₂O). However, the environmental consequences extend well beyond this simple equation.
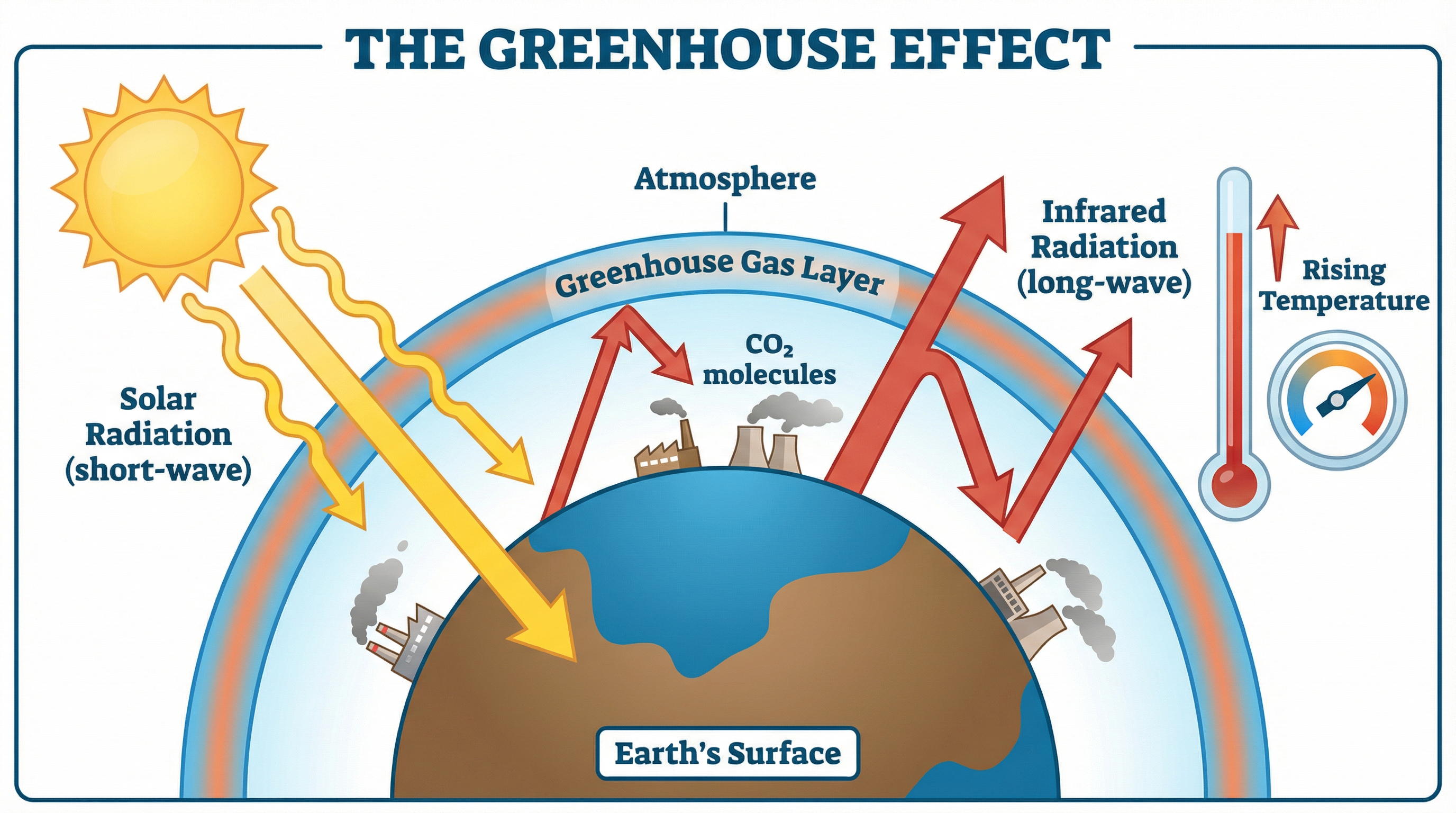
**Carbon Dioxide and the Greenhouse Effect**: CO₂ is a greenhouse gas. Short-wave solar radiation from the Sun passes through the atmosphere relatively unimpeded and warms the Earth's surface. The warmed surface then emits longer-wave infrared radiation. Greenhouse gases, including CO₂, absorb this outgoing infrared radiation and re-emit it in all directions — including back towards Earth. This traps thermal energy in the atmosphere, raising global temperatures. This is the **greenhouse effect**, and enhanced levels of CO₂ from fossil fuel combustion are intensifying it, driving **global warming** and **climate change**.
**Critical examiner warning**: The greenhouse effect is entirely distinct from ozone layer depletion. The ozone layer is damaged by chlorofluorocarbons (CFCs), not by CO₂. Examiners will award zero marks to any response that links fossil fuel combustion to ozone layer damage. These are different atmospheric phenomena with different causes.
**Sulfur Dioxide and Acid Rain**: Coal and oil contain sulfur impurities. When burned, these produce sulfur dioxide (SO₂). SO₂ dissolves in atmospheric moisture to form sulfuric acid (H₂SO₄), which falls as **acid rain**. Acid rain acidifies lakes and rivers (killing aquatic life), damages forests, and corrodes limestone buildings and metal structures. Candidates must link SO₂ specifically to acid rain — not to global warming. The distinction between CO₂ (global warming) and SO₂ (acid rain) is a frequent exam focus.
**Particulates and Global Dimming**: Coal combustion in particular releases fine solid particles — particulates — into the atmosphere. These reflect incoming solar radiation back into space, reducing the amount reaching Earth's surface. This phenomenon is called **global dimming** and is more commonly assessed at Higher tier, though Foundation candidates benefit from awareness of it.
### Concept 3: Advantages of Fossil Fuels — Why We Still Use Them
For an Evaluate question to earn full marks, candidates must present both sides of the argument. The advantages of fossil fuels are substantial and must be articulated precisely.
**High Energy Density**: Fossil fuels release a large quantity of energy per unit mass. Coal, oil, and gas have significantly higher energy density than most renewable alternatives such as biofuels. This makes them economically efficient and practical for large-scale power generation and transport.
**Reliability and Base-Load Electricity**: Fossil fuel power stations can generate electricity continuously, at any time of day, in any weather conditions. This makes them ideal for providing **base-load electricity** — the constant minimum level of power that a national grid must supply at all times. By contrast, wind turbines only generate power when wind speeds are sufficient, and solar panels only generate during daylight hours. This intermittency is a significant disadvantage of many renewable sources.
**Start-Up Time**: Gas-fired power stations have a relatively fast start-up time and can be brought online quickly to meet sudden increases in demand (peak demand). Coal-fired stations have a slower start-up time, requiring hours to reach operating temperature. Both are more responsive than nuclear power stations, which take the longest to start up.
**Existing Infrastructure**: Decades of investment in power stations, pipelines, refineries, and distribution networks mean that fossil fuel energy systems are deeply embedded in national economies. Transitioning to alternatives requires enormous capital investment and time.
### Concept 4: Nuclear Power — A Common Misconception
Nuclear power is frequently misclassified by candidates. Nuclear power is **non-renewable** — uranium is a finite mineral resource — but it is **not** a fossil fuel. Fossil fuels are exclusively coal, oil, and gas, formed from ancient organic matter. Uranium is a metal ore extracted from the Earth's crust. Nuclear fission releases energy from atomic nuclei, not from chemical combustion. Candidates who classify nuclear as a fossil fuel will lose marks.
## Mathematical and Scientific Relationships
While Topic 5.7 is primarily conceptual and evaluative rather than calculation-based, candidates should be familiar with the following relationships:
| Relationship | Formula | Notes |
|---|---|---|
| Energy released by fuel | E = m × specific energy value | Specific energy values are given in data; units are J/kg |
| Efficiency of power station | Efficiency = (useful output energy ÷ total input energy) × 100% | Must memorise; expressed as a percentage |
| Combustion of methane | CH₄ + 2O₂ → CO₂ + 2H₂O | Qualitative understanding required; balancing not assessed in this topic |
Note: The efficiency formula is assessed across multiple topics. For fossil fuel power stations, typical efficiencies range from approximately 35% (coal) to 55% (combined-cycle gas). The remainder is wasted as thermal energy to the surroundings.
## Practical Applications
This topic does not include a required practical, but candidates should be able to interpret data from graphs and tables comparing energy sources. Common data-handling tasks include:
- Reading bar charts showing CO₂ emissions per unit of electricity generated for different fuels.
- Interpreting line graphs showing changes in global temperature over time alongside rising atmospheric CO₂ concentrations.
- Evaluating tables comparing start-up times, reliability, and energy density of different power sources.
When interpreting such data, candidates should describe the trend, quote specific values from the graph, and link the data to the underlying scientific explanation. For example: "The graph shows that coal produces approximately 820 g of CO₂ per kWh, compared to 490 g for gas. This is because coal has a higher carbon content per unit of energy released."