Nuclear Fusion — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
This guide provides a comprehensive, exam-focused breakdown of Nuclear Fusion (OCR GCSE Physics 7.9). It covers the fundamental process, the extreme conditions required, and the critical differences between fusion and fission, all tailored to help you secure maximum marks.

## Overview
Nuclear Fusion is the process that powers our Sun and every star in the universe. It involves the joining of light atomic nuclei to form a heavier nucleus, releasing a vast amount of energy in the process. For your OCR GCSE Physics exam, understanding fusion is not just about memorising a definition; it’s about explaining *why* it happens and the immense challenges in replicating it on Earth. This topic connects directly to your understanding of atomic structure (P7.1), energy transfers (P1), and radioactivity (P7.2). Examiners will test your ability to use precise scientific language, compare fusion with its counterpart, fission, and, for Higher Tier candidates, explain the underlying physics of the conditions required. Expect to see structured questions, comparisons, and calculation-based problems related to the energy released.

## Key Concepts
### Concept 1: The Fusion Process
At its core, nuclear fusion is the forceful merging of two light nuclei. The most common example, and the one you should remember, is the fusion of hydrogen isotopes (like Deuterium, H-2) to form a helium nucleus. The crucial point is that the mass of the final helium nucleus is slightly *less* than the combined mass of the two original hydrogen nuclei. This ‘missing’ mass is not lost; it is converted directly into a huge amount of energy, as described by Albert Einstein’s famous equation, E=mc².
**Example**: If two Deuterium nuclei fuse, the product is a Helium-3 nucleus and a neutron. The total mass *before* is greater than the total mass *after*. This difference in mass, known as the **mass defect**, is released as kinetic energy of the products.
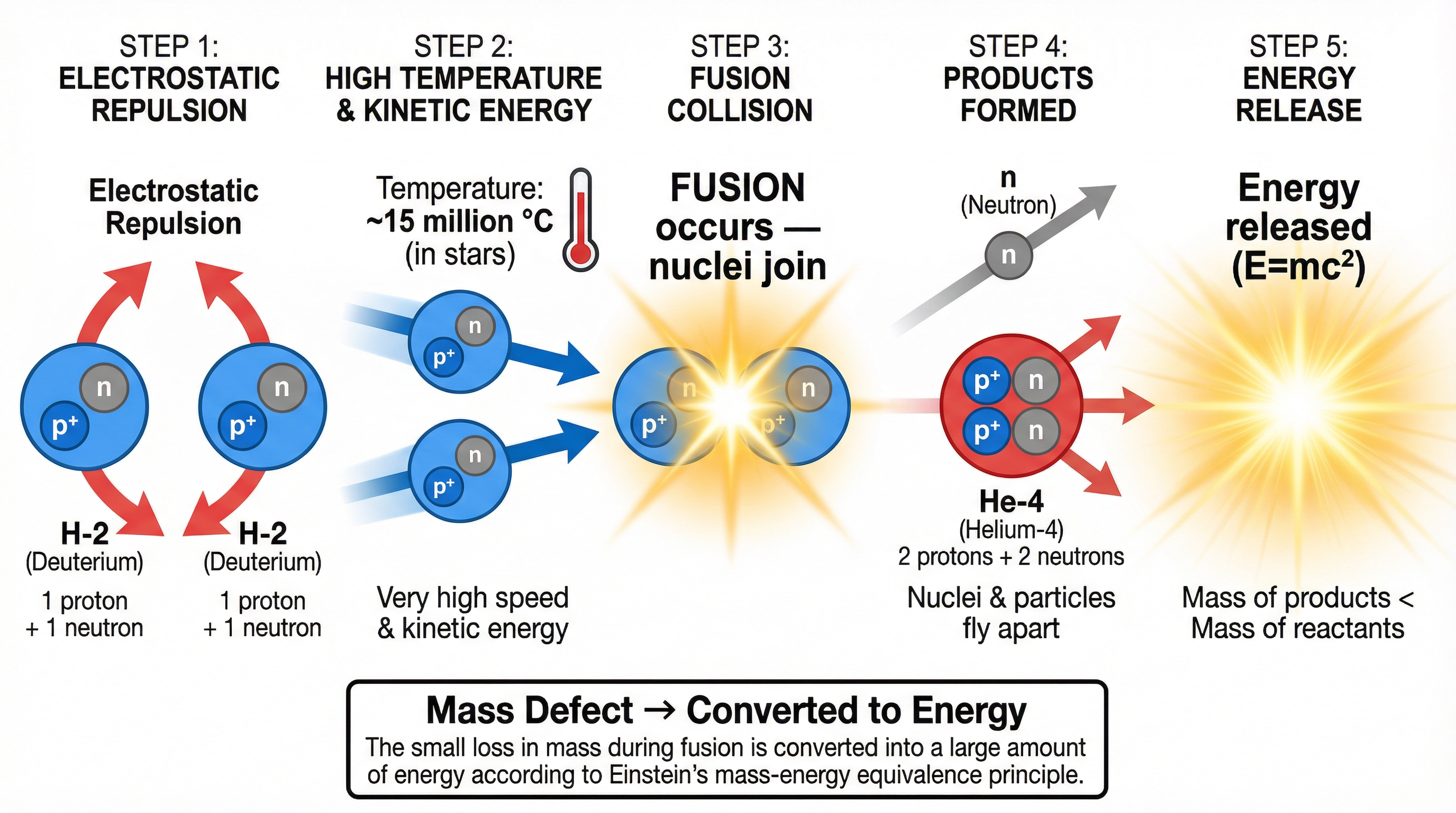
### Concept 2: The Conditions for Fusion (Higher Tier)
Fusion doesn’t happen spontaneously. The reason is that atomic nuclei are positively charged (due to their protons). When you try to bring them close together, a powerful force of **electrostatic repulsion** pushes them apart. To overcome this repulsion, the nuclei must be moving at incredibly high speeds. This is achieved by heating them to extreme temperatures—over 100 million degrees Celsius in experimental reactors on Earth, even hotter than the Sun's core. At these temperatures, the material becomes a **plasma**, and the nuclei have enough kinetic energy to overcome the electrostatic barrier and get close enough for the short-range **strong nuclear force** to pull them together.
* **High Temperature**: Gives nuclei high kinetic energy.
* **High Pressure/Density**: Increases the collision rate, making fusion more likely.
### Concept 3: Fusion vs. Fission
A common 6-mark question requires candidates to compare and contrast nuclear fusion with nuclear fission. It is vital to be able to describe both similarities and differences clearly.
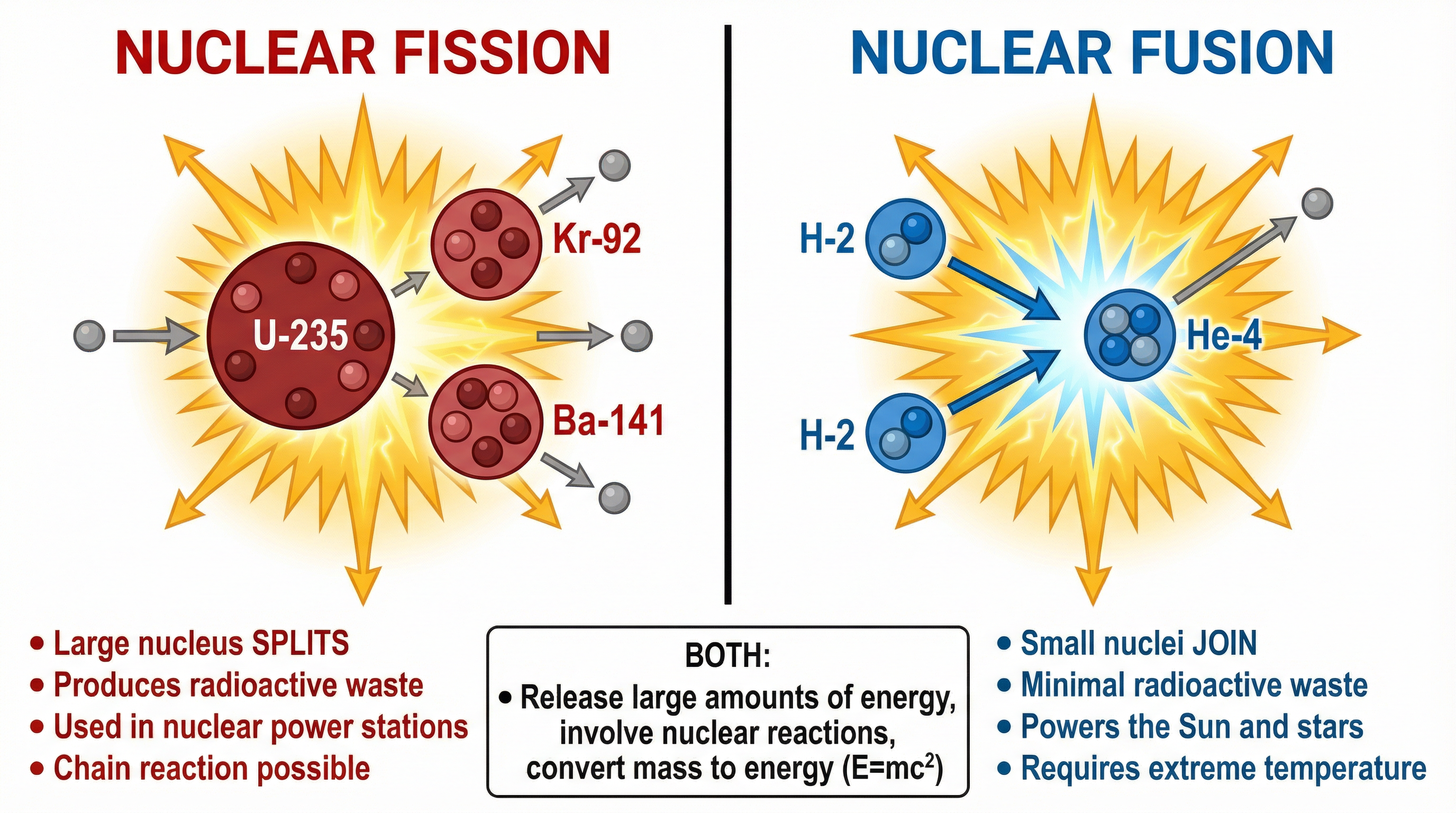
| Feature | Nuclear Fission | Nuclear Fusion |
| :--- | :--- | :--- |
| **Process** | A large, unstable nucleus **splits** into two smaller nuclei. | Two small, light nuclei **join** (fuse) to form a heavier nucleus. |
| **Nuclei Involved** | Heavy nuclei (e.g., Uranium-235, Plutonium-239). | Light nuclei (e.g., Hydrogen isotopes like Deuterium and Tritium). |
| **Conditions** | Can occur at normal temperatures; initiated by a slow neutron. | Requires extremely high temperatures and pressures. |
| **Waste Products** | Produces highly radioactive waste with a long half-life. | Produces very little long-lived radioactive waste (Helium is inert). |
| **Current Use** | Used in nuclear power stations to generate electricity. | Powers the Sun and stars; experimental on Earth (e.g., JET, ITER). |
**Similarity**: Both processes release a very large amount of energy per reaction and both involve a conversion of mass into energy (mass defect).
## Mathematical/Scientific Relationships
### E = mc² (Must memorise)
This is the cornerstone equation for both fission and fusion. It states that energy (E) is equal to mass (m) multiplied by the speed of light squared (c²).
* **E**: Energy released (in Joules, J)
* **m**: The mass defect (the mass that is converted into energy, in kilograms, kg)
* **c**: The speed of light in a vacuum (approximately 3 x 10⁸ m/s). You will be given this value.
You will not be asked to perform complex calculations with this at GCSE, but you MUST be able to state that the energy released is due to a loss in mass.
## Practical Applications
* **Stellar Energy**: All stars, including our Sun, are powered by nuclear fusion. The immense gravitational pressure in their cores creates the conditions necessary for fusion to occur, providing the energy that sustains them for billions of years.
* **Fusion Research (Tokamaks)**: On Earth, scientists are developing fusion reactors to generate clean electricity. The most common design is the **tokamak**, a doughnut-shaped device that uses powerful magnetic fields to confine and heat a hydrogen plasma to extreme temperatures. The Joint European Torus (JET) in the UK is a leading example. The goal is to achieve a net energy gain, where the reactor produces more energy than it consumes.
* **Nuclear Weapons**: Hydrogen bombs (thermonuclear weapons) use a fission bomb as a trigger to create the intense temperatures and pressures needed to initiate a massive, uncontrolled fusion reaction.