Nuclear Power — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
This guide provides a comprehensive overview of Nuclear Power for OCR GCSE Physics (5.8), focusing on the core concepts of fission and fusion. It is designed to help students master the key principles, avoid common mistakes, and excel in their exams by providing detailed explanations, worked examples, and multi-modal learning aids.

## Overview
Nuclear Power is a critical topic in modern physics, dealing with the immense energy stored within atomic nuclei. For your OCR GCSE Physics exam, you will be expected to understand the two primary methods of releasing this energy: **nuclear fission** (the splitting of a large nucleus) and **nuclear fusion** (the joining of two smaller nuclei). This guide will break down the precise mechanics of these processes, explore how a nuclear reactor functions, and evaluate the significant environmental and economic factors involved. A solid grasp of this topic is essential, as it frequently appears in both short-answer and long-form evaluation questions, testing your knowledge (AO1), application (AO2), and analytical skills (AO3). This topic has strong synoptic links to 'Energy' (Topic 2) and 'Radioactivity' (Topic 5.1-5.7), so a deep understanding here will pay dividends across the paper.

## Key Concepts
### Concept 1: Nuclear Fission
Nuclear fission is the process where a large, unstable atomic nucleus, such as Uranium-235, splits into two smaller nuclei, known as **daughter nuclei**. This process is not spontaneous; it must be induced. For Uranium-235, this is achieved by making it absorb a slow-moving neutron, often called a thermal neutron. The absorption makes the nucleus critically unstable (forming Uranium-236 for a fraction of a second), causing it to violently split. This event releases a tremendous amount of energy, primarily as the kinetic energy of the daughter nuclei and the neutrons, along with some gamma radiation. Crucially, the fission of one nucleus also releases 2-3 fast-moving neutrons. These neutrons are the key to a chain reaction.
**Example**: A single Uranium-235 nucleus absorbs a slow neutron. It splits into a Barium-141 nucleus and a Krypton-92 nucleus, releasing three fast neutrons and approximately 200 MeV of energy.
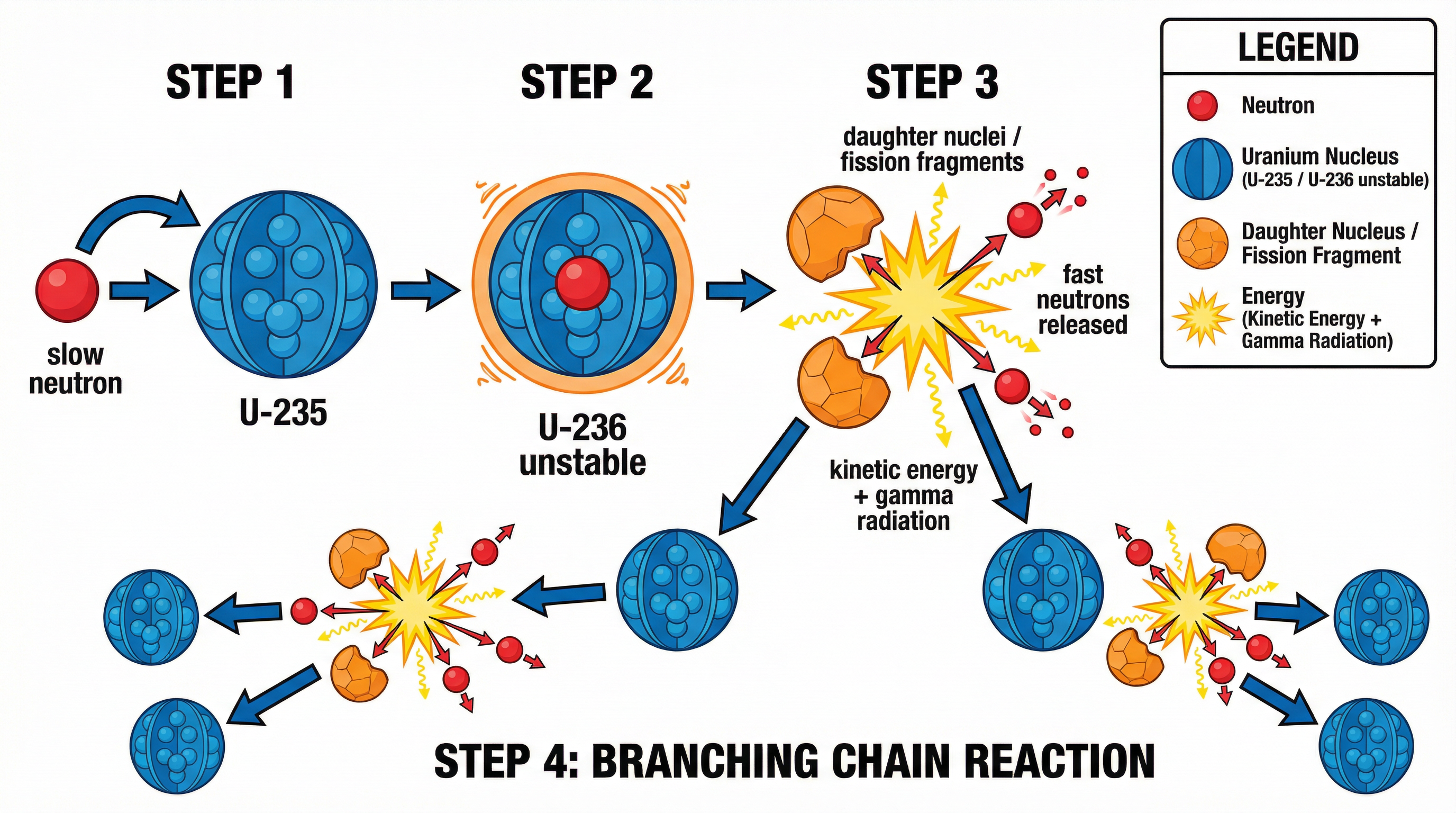
### Concept 2: The Chain Reaction
A chain reaction is a self-sustaining process where the products of one reaction go on to trigger subsequent reactions. In a nuclear reactor, the 2-3 neutrons released from a fission event are slowed down and then absorbed by other Uranium-235 nuclei. If, on average, exactly one neutron from each fission causes another fission, the reaction is stable and self-sustaining, producing energy at a constant rate. This is a **controlled chain reaction**. If more than one neutron causes further fissions, the reaction rate increases exponentially, leading to an uncontrolled release of energy, as in a nuclear weapon.
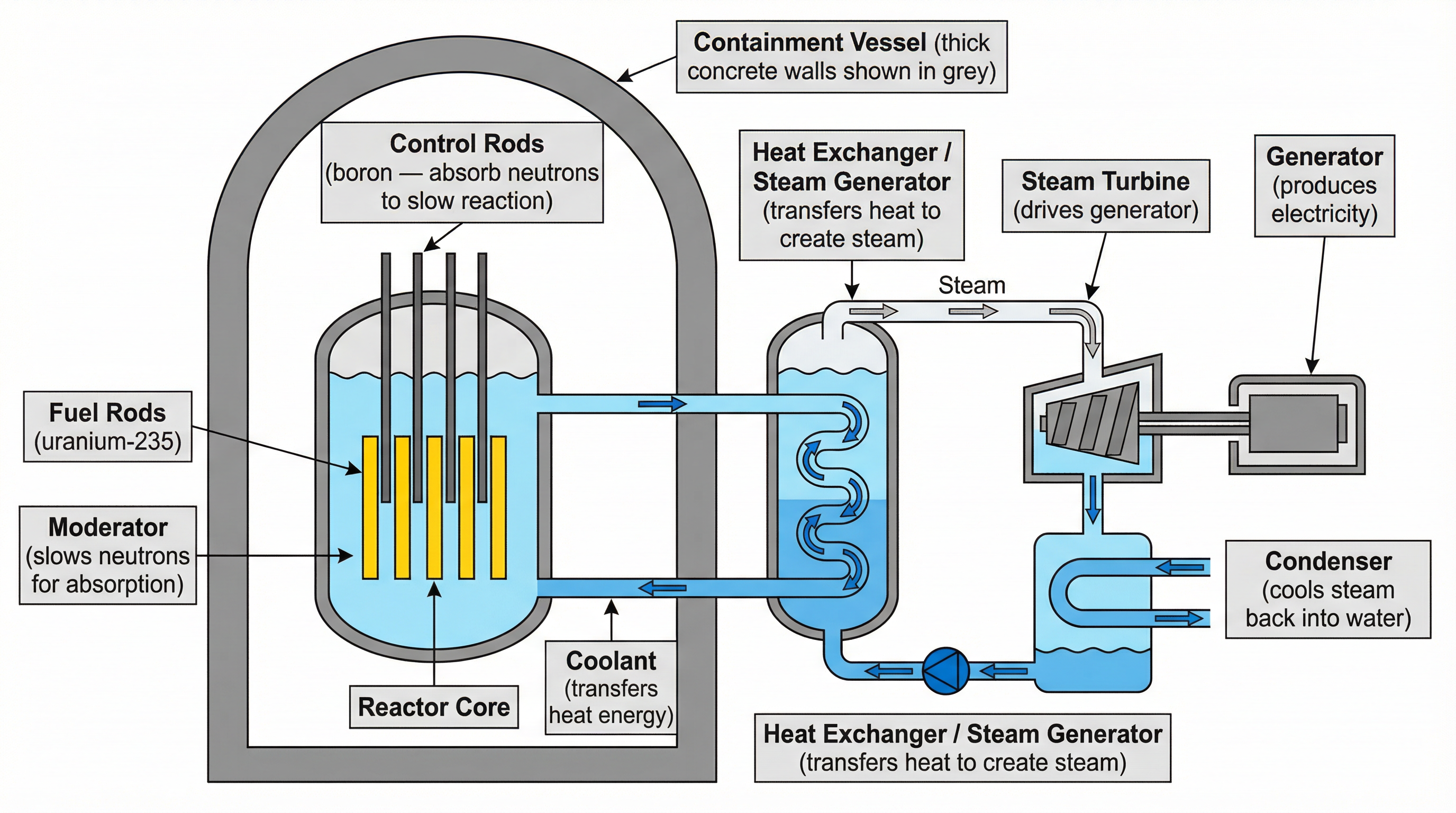
### Concept 3: The Role of the Moderator and Control Rods
Controlling a chain reaction requires managing the neutrons. This is done by two key components in a reactor core:
* **The Moderator**: The neutrons released during fission are moving too fast to be efficiently absorbed by other U-235 nuclei. The moderator, typically made of water or graphite, surrounds the fuel rods. The fast neutrons collide with the moderator's atoms, transferring kinetic energy and slowing down to become thermal neutrons. This is essential for the chain reaction to be sustained.
* **The Control Rods**: These are made of a material that readily absorbs neutrons, such as boron. By inserting the control rods into the reactor core, excess neutrons are absorbed, which slows down the rate of fission. By withdrawing them, the rate increases. They are the primary mechanism for controlling the reactor's power output and for shutting it down completely in an emergency.
### Concept 4: Nuclear Fusion
Nuclear fusion is the process that powers the Sun. It involves forcing two light atomic nuclei together to form a single, heavier nucleus. For example, two isotopes of hydrogen, Deuterium (H-2) and Tritium (H-3), can be fused to create a Helium (He-4) nucleus and a neutron. This process releases even more energy per unit mass than fission. However, fusion requires overcoming the immense electrostatic repulsion between the two positively charged nuclei. To achieve this, the fuel must be heated to extreme temperatures (over 100 million °C) and pressures, creating a state of matter called plasma. Containing this plasma is a major technological challenge, which is why fusion power is not yet commercially viable.
## Mathematical/Scientific Relationships
While you are not required to memorise complex nuclear equations for GCSE, you should be familiar with the concept of mass-energy equivalence, as described by Einstein's famous equation:
**E = mc²**
* **E**: Energy released (in Joules, J)
* **m**: Mass defect (the small amount of mass converted into energy, in kilograms, kg)
* **c**: The speed of light in a vacuum (approximately 3.00 x 10⁸ m/s)
This equation is **Given on the formula sheet**. It explains why such a tiny amount of mass in nuclear reactions can be converted into such a vast amount of energy. The speed of light squared (c²) is a huge number, which acts as a massive multiplier.
## Practical Applications
* **Electricity Generation**: The primary application of nuclear fission is in nuclear power stations to generate electricity. They provide a reliable, low-carbon source of baseload power, meaning they can operate continuously for long periods.
* **Medical Isotopes**: Nuclear reactors are used to produce radioactive isotopes for medical imaging and cancer treatment.
* **Nuclear Submarines**: Compact nuclear reactors power naval submarines, allowing them to stay submerged for months at a time without needing to refuel.
* **Research**: Experimental fusion reactors like the Joint European Torus (JET) in the UK are pushing the boundaries of science, aiming to create a clean, virtually limitless energy source for the future.