The Particle Model — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
This guide covers the OCR GCSE Physics topic of The Particle Model (P1.1), a cornerstone of thermal physics. We'll break down states of matter, density, internal energy, and changes of state, focusing on the language and calculations needed to secure maximum marks in your exam.
## Overview
The Particle Model is fundamental to understanding the physical world. It explains the properties of solids, liquids, and gases by looking at what their constituent particles are doing. For your OCR GCSE exam, mastering this topic is crucial as it provides the foundation for understanding energy transfers, pressure, and thermal physics. Examiners frequently test your ability to describe the arrangement and motion of particles, calculate density, and interpret heating curves. A solid grasp here is essential for linking concepts, such as how energy input affects the kinetic and potential energy stores of particles, which is a common source of confusion for many candidates. Expect to see a mix of short-answer definition questions, calculation-based problems, and longer, 6-mark questions requiring you to describe a practical procedure like measuring the density of an irregular object.

## Key Concepts
### Concept 1: States of Matter and Particle Arrangement
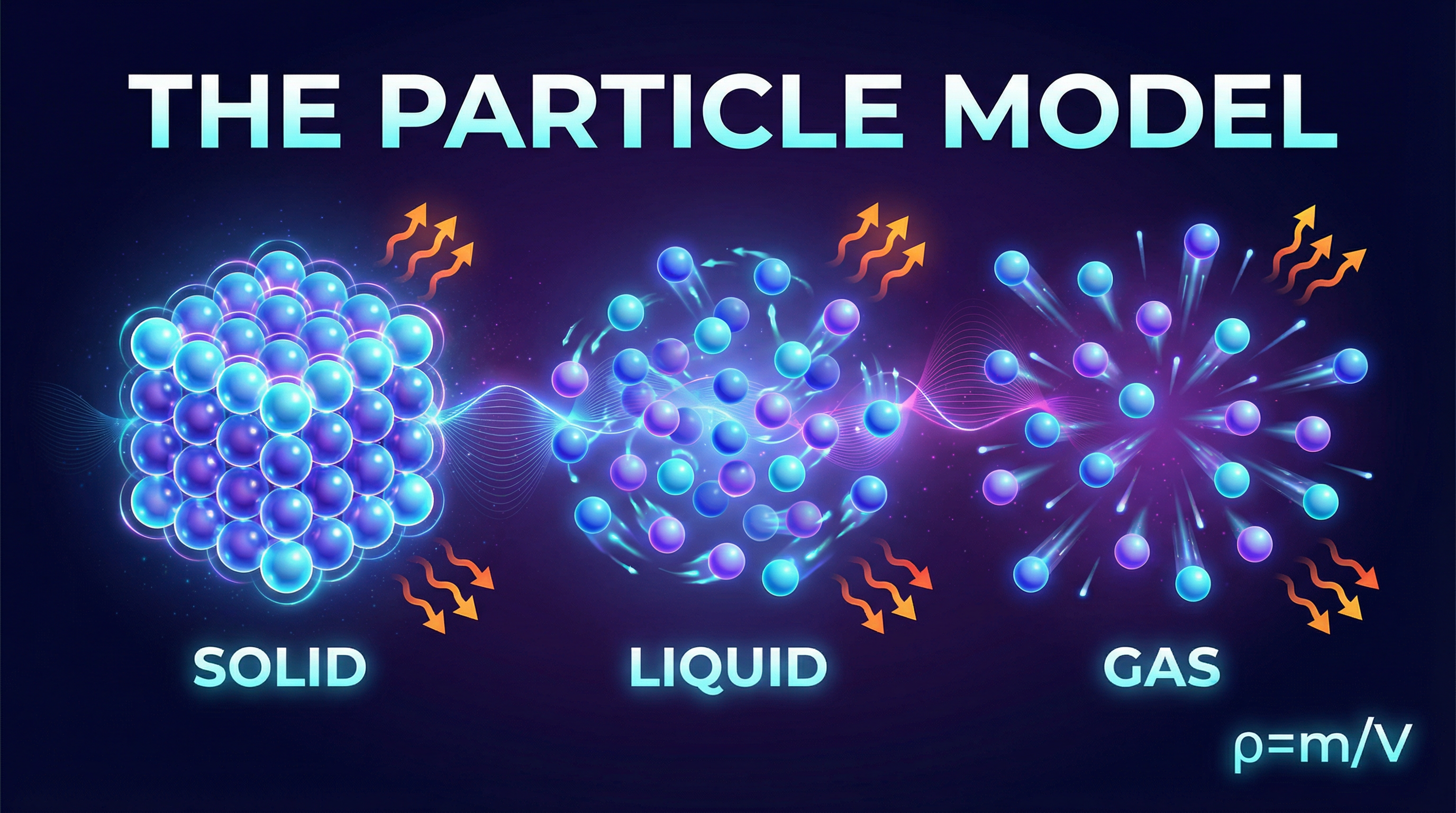
Everything is made of particles, but how these particles are arranged and how they move dictates the properties of a substance. Examiners award marks for precise descriptions.
* **Solids**: Particles are held in fixed positions within a regular, repeating pattern known as a **lattice**. They are tightly packed and have strong intermolecular forces between them. The particles can only **vibrate** about their fixed positions. This is why solids have a fixed shape and volume.
* **Liquids**: Particles are still closely packed but are arranged randomly. The intermolecular forces are weaker than in solids, allowing the particles to **move past one another**. This is why liquids can flow and take the shape of their container, but have a fixed volume.
* **Gases**: Particles are far apart with very weak intermolecular forces between them. They move randomly and rapidly in all directions. This is why gases have no fixed shape or volume and will fill any container they are in.
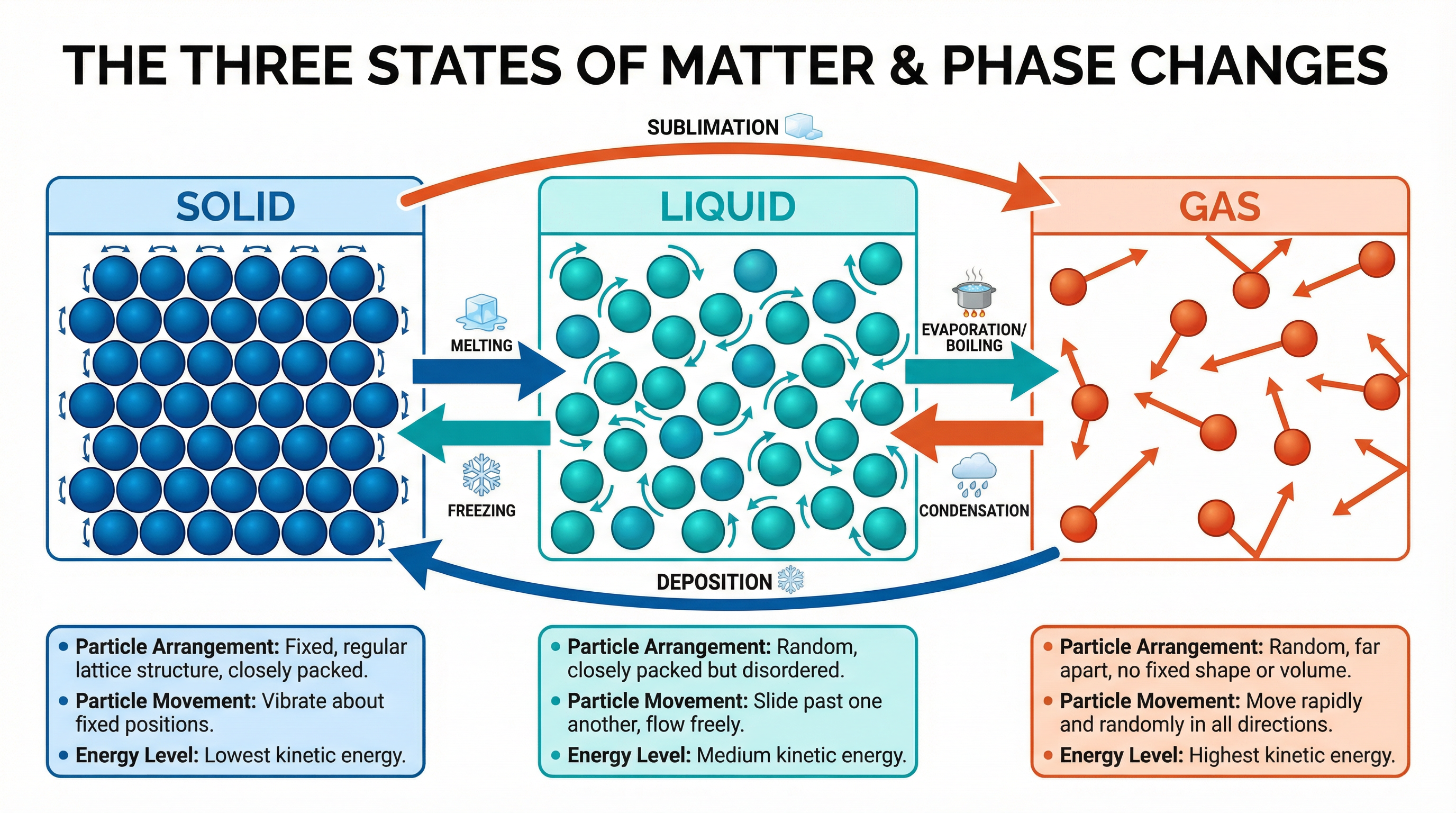
**Key Exam Point**: When a substance changes state, the number of particles does not change. This means **mass is conserved** during changes of state. If you melt an ice cube, the mass of the water is the same as the mass of the ice.
### Concept 2: Density
Density is a measure of how much 'stuff' (mass) is packed into a given space (volume). It explains why a block of iron is heavier than a block of wood of the same size.
* **Definition**: Density is the mass per unit volume.
* **Formula**: You must memorise this equation.
`ρ = m / V`
Where:
* `ρ` (rho) is density, measured in kilograms per metre cubed (kg/m³).
* `m` is mass, measured in kilograms (kg).
* `V` is volume, measured in metres cubed (m³).
**Example**: A block of aluminium has a mass of 5.4 kg and a volume of 0.002 m³. To find its density, you substitute the values into the formula: `ρ = 5.4 kg / 0.002 m³ = 2700 kg/m³`.
Generally, solids are denser than liquids, and liquids are denser than gases. This is because the particles in a solid are packed most tightly, while in a gas they are most spread out.
### Concept 3: Internal Energy (Higher Tier)
Internal energy is a crucial concept for understanding heat. It is **not** just another word for temperature.
* **Definition**: The internal energy of a system is the **total energy that its particles have in their kinetic and potential energy stores**.
* **Kinetic Energy Store**: Related to the movement of the particles. The faster the particles move or vibrate, the higher their kinetic energy. Temperature is a measure of the average kinetic energy of the particles.
* **Potential Energy Store**: Related to the position of the particles and the forces between them. When you pull particles apart against their intermolecular forces, you increase their potential energy.
Heating a substance increases its internal energy. This energy can either increase the kinetic energy of the particles (raising the temperature) or increase the potential energy of the particles (changing the state).
### Concept 4: Changes of State & Latent Heat
When a substance changes state, its temperature remains constant, even though energy is being supplied. This energy is called **latent heat**.
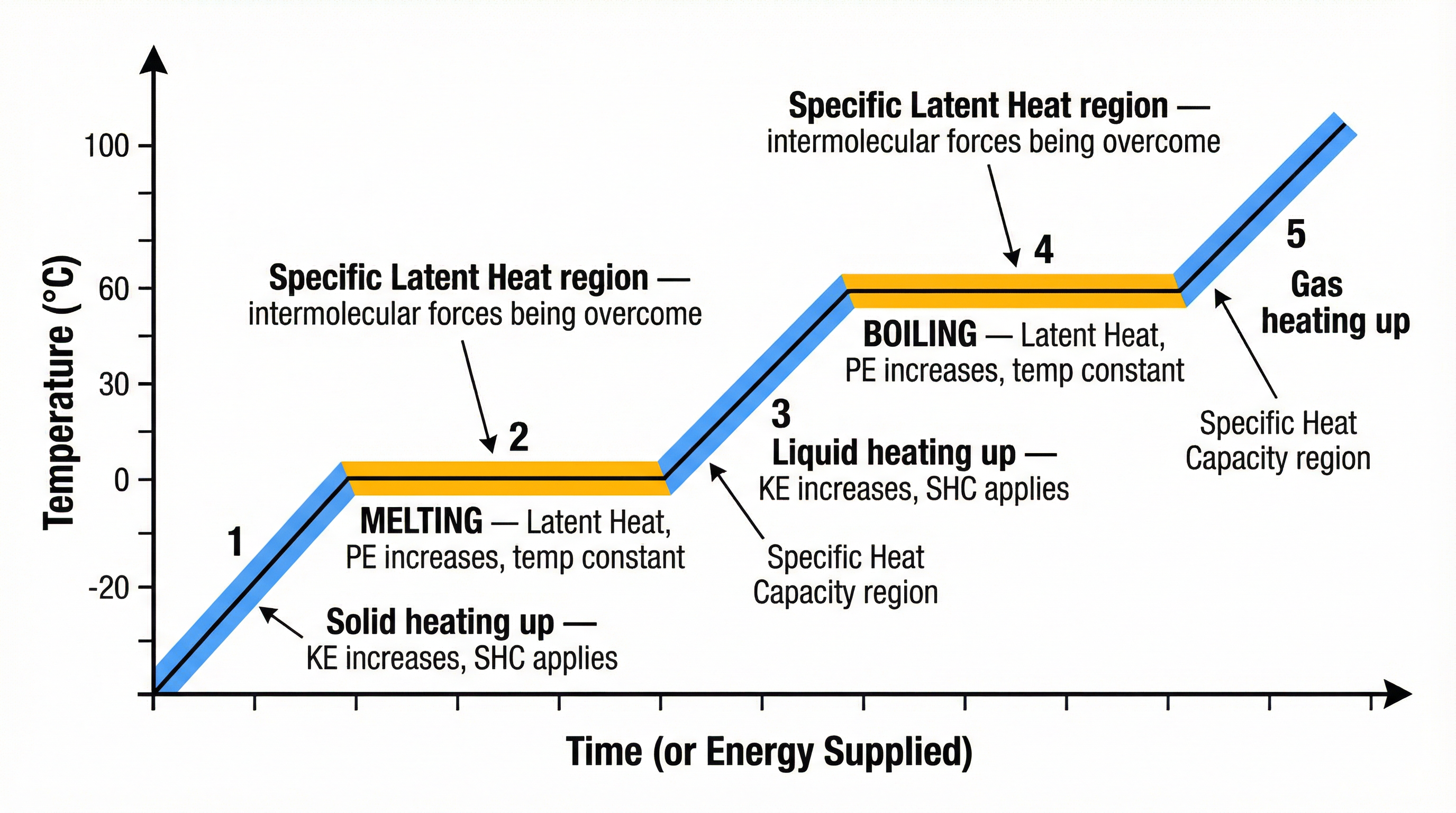
* **Specific Latent Heat (L)**: The energy required to change the state of 1 kg of a substance **without any change in temperature**.
* **Formula**: `E = m × L` (Given on the formula sheet)
* `E` is energy for a change of state (J)
* `m` is mass (kg)
* `L` is specific latent heat (J/kg)
There are two types of specific latent heat:
1. **Specific Latent Heat of Fusion (L_f)**: Energy needed to melt (solid to liquid) or freeze (liquid to solid).
2. **Specific Latent Heat of Vaporisation (L_v)**: Energy needed to boil (liquid to gas) or condense (gas to liquid).
During a change of state, the energy supplied is used to **overcome intermolecular forces**, which increases the potential energy store of the particles. The kinetic energy store does not change, which is why the temperature remains constant.
### Concept 5: Specific Heat Capacity
When you heat a substance and its temperature rises (i.e., it is not changing state), the energy required is determined by its specific heat capacity.
* **Specific Heat Capacity (c)**: The energy required to raise the temperature of 1 kg of a substance by 1°C.
* **Formula**: `ΔE = m × c × Δθ` (Given on the formula sheet)
* `ΔE` is the change in thermal energy (J)
* `m` is mass (kg)
* `c` is specific heat capacity (J/kg°C)
* `Δθ` (delta theta) is the temperature change (°C)
Substances with a high specific heat capacity, like water, require a lot of energy to heat up. This is why water is used in central heating systems.
## Mathematical/Scientific Relationships
| Formula | Symbol Meanings | When to Use | Given or Memorise? |
| :--- | :--- | :--- | :--- |
| `ρ = m / V` | `ρ`: density (kg/m³)<br>`m`: mass (kg)<br>`V`: volume (m³) | Calculating density, mass, or volume. | **Must memorise** |
| `ΔE = m × c × Δθ` | `ΔE`: change in thermal energy (J)<br>`m`: mass (kg)<br>`c`: specific heat capacity (J/kg°C)<br>`Δθ`: temperature change (°C) | Calculating energy change when temperature changes (no state change). | Given on formula sheet |
| `E = m × L` | `E`: energy for state change (J)<br>`m`: mass (kg)<br>`L`: specific latent heat (J/kg) | Calculating energy change during a state change (melting, boiling, etc.) at constant temperature. | Given on formula sheet |
**Unit Conversions**: Examiners love to test these!
* **Mass**: 1 kg = 1000 g
* **Volume**: 1 m³ = 1,000,000 cm³. To convert cm³ to m³, you must **divide by 1,000,000**.
## Practical Applications
### Required Practical: Determining Density
This is a classic 6-mark question. You need to describe a method to find the density of a regular object, an irregular object, and a liquid.
**Apparatus**:
* Top-pan balance (for measuring mass)
* Ruler or vernier calipers (for regular object)
* Displacement (Eureka) can and measuring cylinder (for irregular object)
* Measuring cylinder (for liquid)
**Method for an Irregular Solid (e.g., a rock)**:
1. **Measure Mass**: Place the rock on a top-pan balance and record its mass in kg.
2. **Measure Volume**:
a. Fill a displacement can with water until the water is just about to flow out of the spout.
b. Place an empty measuring cylinder under the spout.
c. Carefully lower the rock into the can, ensuring it is fully submerged. Do not splash.
d. Collect the water that flows out of the spout into the measuring cylinder. This volume of water is equal to the volume of the rock.
e. Record the volume in cm³, then convert to m³ by dividing by 1,000,000.
3. **Calculate Density**: Use the formula `ρ = m / V` with your measured mass and volume.
**Common Errors**:
* Forgetting to convert volume from cm³ to m³.
* Splashing water when lowering the object, leading to an inaccurate volume measurement.
* When measuring the density of a liquid, forgetting to subtract the mass of the empty measuring cylinder from the total mass to find the mass of the liquid alone.