Uses of Radiation — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
This guide provides a comprehensive, exam-focused breakdown of OCR GCSE Physics topic 7.6: Uses of Radiation. It covers the critical applications of alpha, beta, and gamma radiation in industry and medicine, explaining exactly how to justify the choice of isotope to secure maximum marks.

## Overview
Welcome to one of the most applied and frequently examined topics in GCSE Physics. 'Uses of Radiation' (OCR specification reference 7.6) moves beyond the theoretical nature of radioactive decay and into the real world. This is where your understanding of penetrating power, ionising ability, and half-life is put to the test in practical scenarios. Examiners rigorously assess your ability to justify why a specific isotope is chosen for a job, from smoke detectors in our homes to life-saving medical scanners in hospitals. A strong grasp of this topic is crucial as it often features in higher-mark, structured questions that require you to link multiple concepts together. You will be expected to clearly distinguish between the physical processes of irradiation and contamination, a common source of confusion for many candidates. This guide will equip you with the precise language and conceptual understanding needed to tackle any question on this topic with confidence.
## Key Concepts
### Concept 1: Properties of Ionising Radiation
To understand the uses of radiation, you must first be an expert on the tools themselves: alpha (α), beta (β), and gamma (γ) radiation. Their properties directly determine their applications. Examiners expect you to compare their range, penetrating power, and ionising ability.
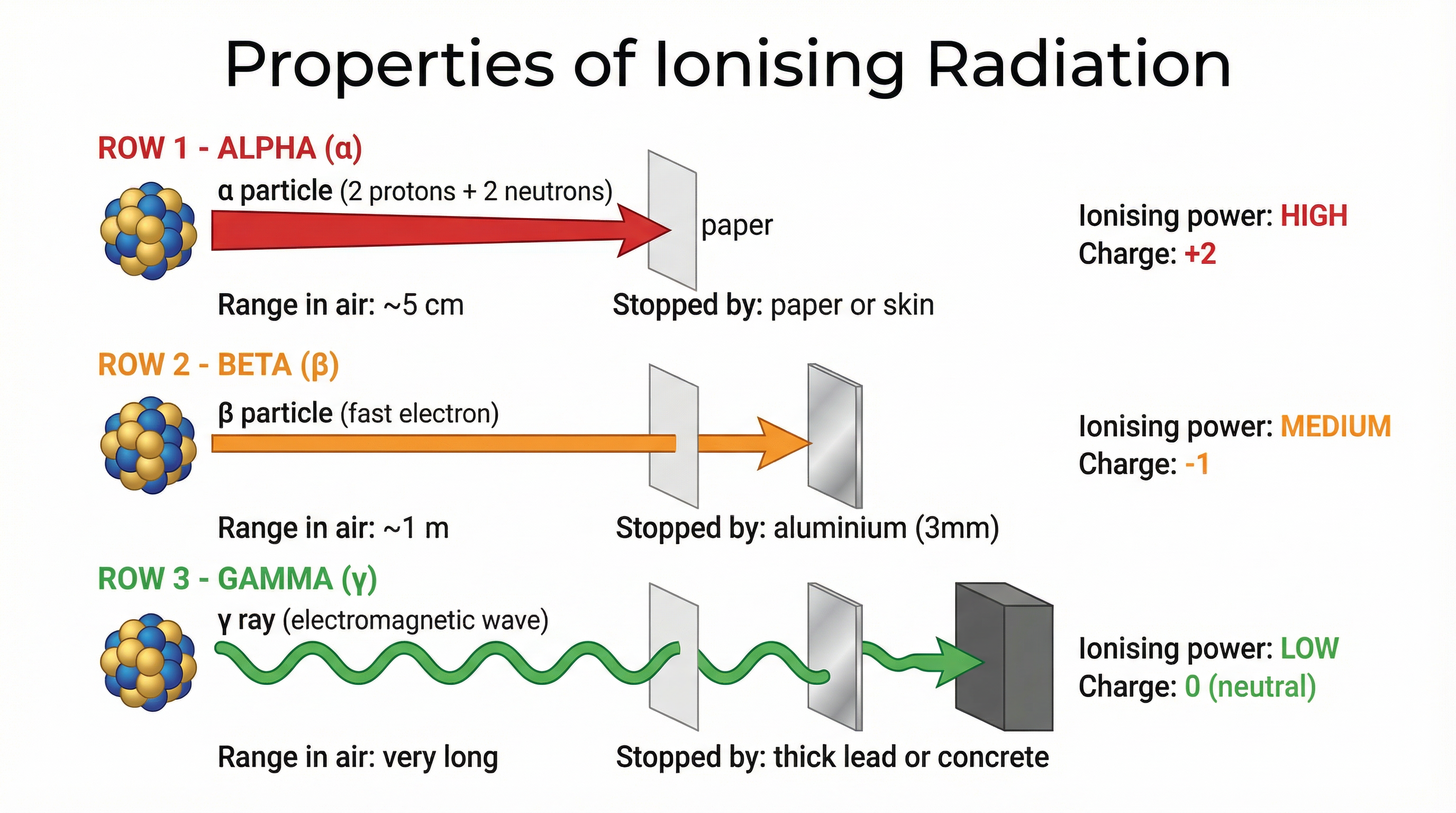
* **Alpha (α):** A helium nucleus (2 protons, 2 neutrons). It is large, slow, and has a +2 charge. This makes it a highly effective ioniser – it is very good at knocking electrons off other atoms. However, this high interaction rate means it loses its energy very quickly and has a very short range (a few cm in air) and low penetrating power (stopped by paper or skin).
* **Beta (β):** A fast-moving electron ejected from the nucleus. It is much smaller and faster than alpha, with a -1 charge. Its ionising ability is moderate, and it has a greater range (around 1m in air) and penetrating power (stopped by ~3mm of aluminium).
* **Gamma (γ):** A high-energy electromagnetic wave. It has no mass and no charge. It interacts weakly with atoms, giving it a very low ionising ability but extremely high penetrating power. It can travel long distances in air and requires thick lead or concrete to be significantly absorbed.
### Concept 2: Irradiation vs. Contamination
This is a critical distinction that frequently appears in exams and is worth a guaranteed mark if you get it right. Confusing the two is one of the most common mistakes.
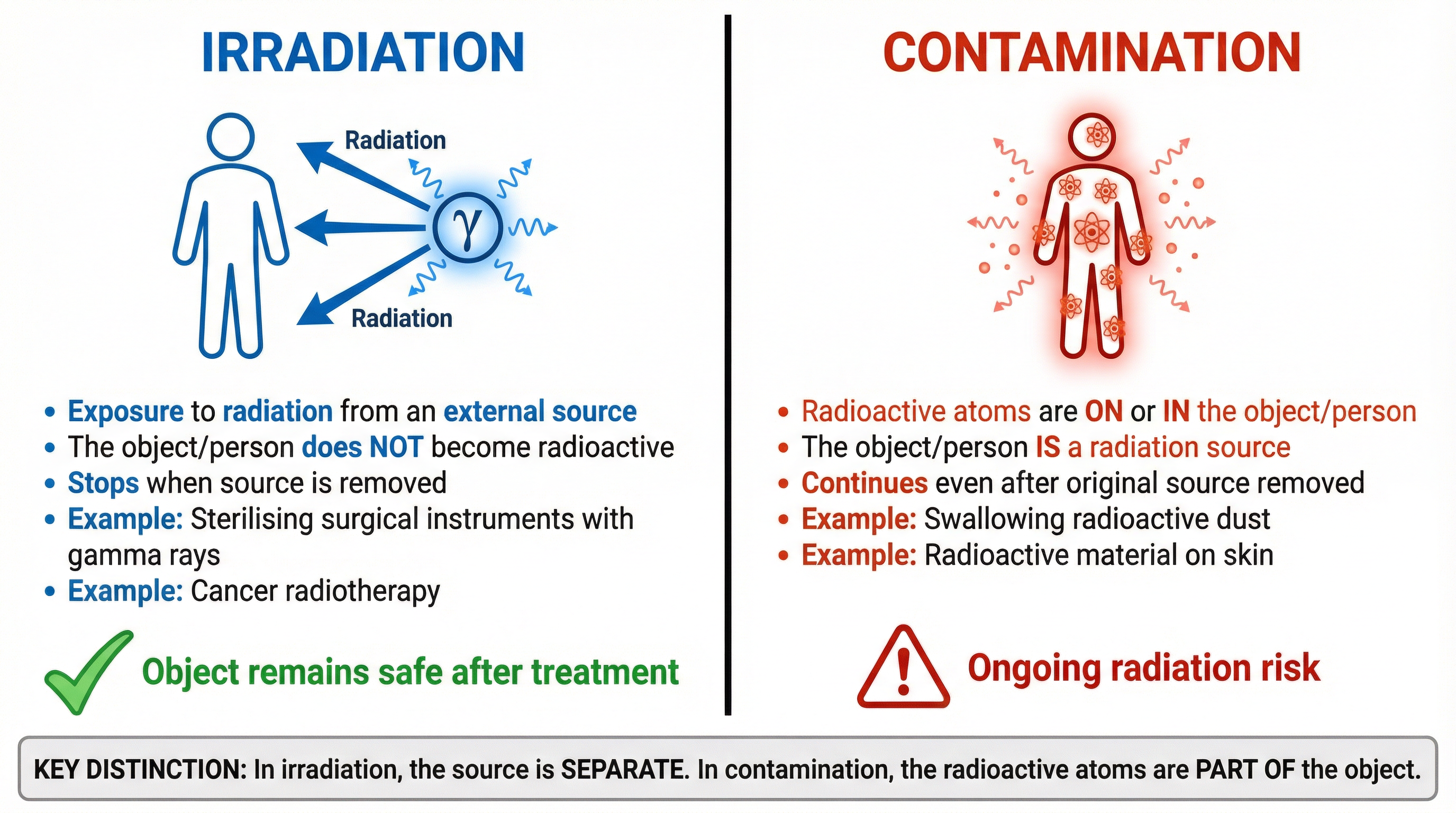
* **Irradiation:** This is the process of an object being exposed to a source of ionising radiation from the outside. The object itself does not become radioactive. Once the source is removed, the irradiation stops. Think of it like standing in the sun – you are being irradiated by UV light, but you don't start glowing when you go indoors.
* **Example:** Sterilising surgical instruments. They are zapped with gamma rays to kill bacteria, but the instruments themselves do not become radioactive sources.
* **Contamination:** This occurs when radioactive atoms or molecules get onto or inside an object. The object itself becomes a source of radiation. The hazard persists even after the original source is removed, as the contaminating atoms continue to decay.
* **Example:** Radioactive dust from a nuclear accident landing on your clothes or being inhaled. Your clothes and lungs are now contaminated and are emitting radiation.
### Concept 3: Half-Life in Practical Applications
Half-life is the time it takes for the number of radioactive nuclei in a sample to halve, or for the count rate from the sample to fall to half its initial level. When choosing an isotope for a job, the half-life is just as important as the type of radiation it emits.
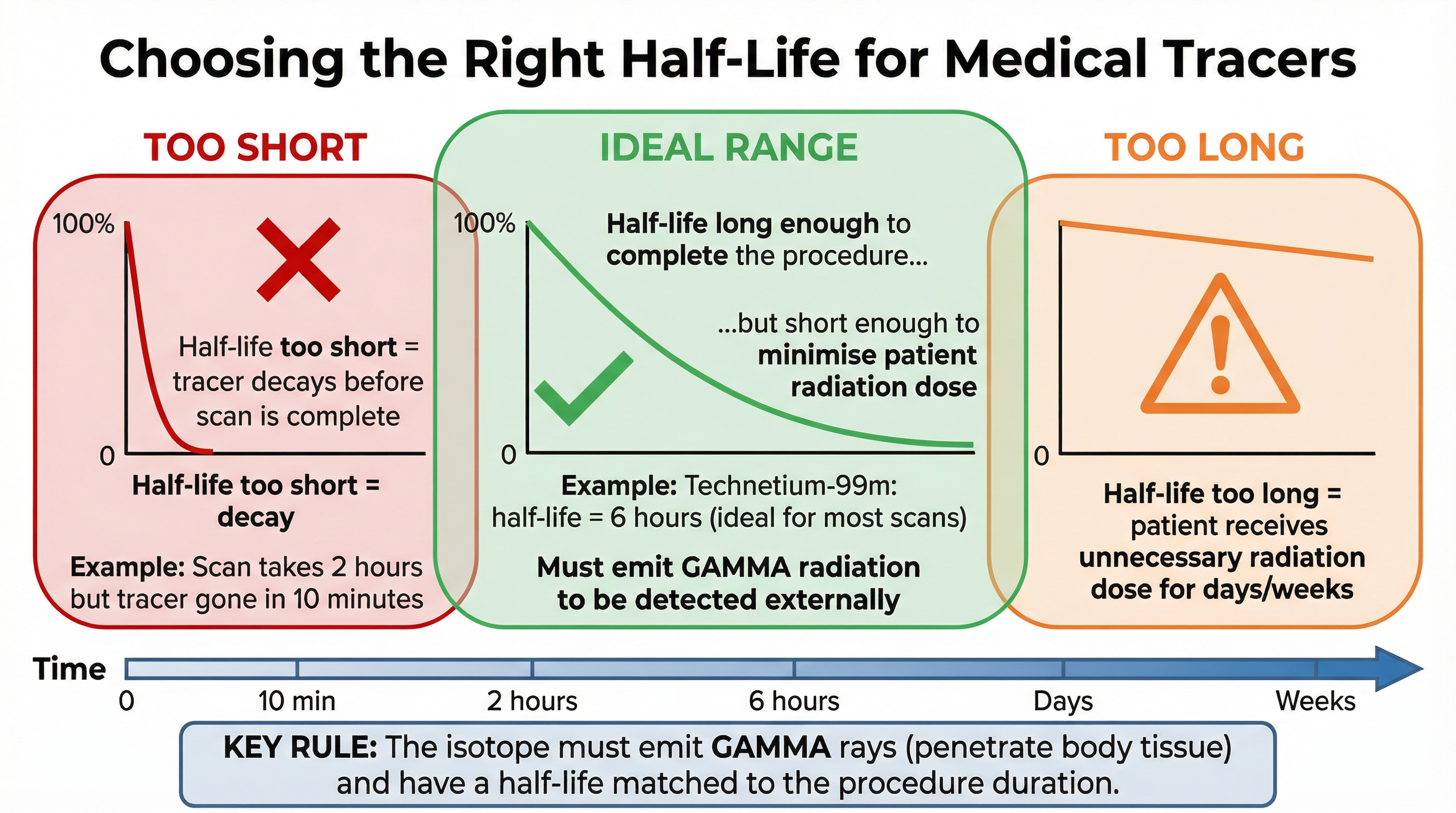
* **For Medical Tracers:** The choice is a trade-off. The half-life must be **long enough** for the medical procedure to be carried out (e.g., for the tracer to travel to the target organ and for the scan to be completed). However, it must be **short enough** to minimise the radiation dose to the patient and ensure the radioactivity disappears from their body in a reasonable time. A half-life of a few hours is often ideal (e.g., Technetium-99m, half-life of 6 hours).
* **For Industrial Gauges & Smoke Detectors:** These require a very **long half-life**. The source needs to provide a consistent and predictable count rate for many years without needing frequent replacement. Americium-241, used in smoke detectors, has a half-life of 432 years.
## Practical Applications
### Smoke Detectors (Alpha Emitter)
An alpha source (Americium-241) is placed in a chamber with two electrodes, creating a small air gap. The alpha particles ionise the air molecules in the gap, allowing a small, constant electric current to flow. If smoke particles enter the chamber, they absorb the alpha particles, preventing them from reaching the air molecules. The ionisation stops, the current drops, and an alarm is triggered.
**Examiner's Checklist:**
1. State it uses **alpha** radiation.
2. Explain alpha has **high ionising power** to create a current.
3. Explain alpha has **low penetrating power** so it is stopped by smoke particles.
4. State that alpha **cannot penetrate the plastic casing**, making it safe.
### Thickness Gauges (Beta Emitter)
In a factory making paper or metal foil, a beta source is placed on one side of the material and a detector (like a Geiger-Müller tube) on the other. The detector measures the **count rate** of beta particles getting through. If the material gets too thick, it absorbs more beta particles, and the count rate drops. If it gets too thin, the count rate rises. This information is fed back to the rollers to adjust the pressure.
**Examiner's Checklist:**
1. State it uses **beta** radiation.
2. Explain beta is used because it is **partially absorbed** by the material. (Alpha would be completely blocked; gamma would pass through unaffected).
3. Use the precise term **'count rate'** changes with thickness.
### Medical Tracers (Gamma Emitter)
A gamma-emitting isotope is attached to a chemical that is absorbed by a specific organ. It is injected into the patient. As it circulates and collects in the target organ, a **gamma camera** outside the body detects the gamma rays. A computer builds up an image showing where the tracer is, indicating how well the organ is functioning.
**Examiner's Checklist:**
1. State it must be a **gamma** emitter.
2. Explain gamma is used because it is **highly penetrating** and can pass out of the body to be detected.
3. Explain the **half-life** must be long enough for the procedure but short enough to minimise patient dose.
### Sterilisation (Gamma Emitter)
Surgical instruments and some foods are sealed in bags and then exposed to a high dose of gamma radiation from a source like Cobalt-60. The gamma rays are highly penetrating and kill all bacteria and microorganisms by damaging their DNA. Because this is **irradiation** and not contamination, the instruments themselves do not become radioactive and are safe to use immediately. The long shelf-life of sterilised medical supplies is thanks to this process.