Nuclear Decay and Particles — WJEC A-Level Study Guide
Exam Board: WJEC | Level: A-Level
Unlock top marks in WJEC A-Level Physics by mastering the fundamental forces that govern the universe. This guide breaks down the complex world of nuclear decay, particle physics, and exponential change into clear, exam-focused concepts, complete with worked examples and examiner insights.

## Overview
Welcome to the study of the very small and the very powerful. Topic 3.3, Nuclear Decay and Particles, is a cornerstone of modern physics that bridges the gap between the atomic nucleus and the fundamental constituents of matter. It's a topic where precise knowledge and mathematical skill are equally rewarded by examiners. You'll explore the random and spontaneous nature of radioactive decay, learning to model it with exponential equations. Then, you'll delve into the Standard Model, a 'particle zoo' where you'll meet quarks, leptons, and the bosons that mediate their interactions. A typical exam question might ask you to calculate the half-life of a radioactive isotope from graphical data, or to verify whether a proposed particle interaction is possible by applying conservation laws. Mastering this topic not only secures a significant portion of marks but also provides a deep understanding of the principles that underpin much of modern technology, from medical imaging to power generation.
## Key Concepts
### 1. Radioactive Decay
An unstable nucleus can spontaneously and randomly emit radiation to become more stable. This process is not affected by external conditions like temperature or pressure. There are three main types of decay candidates must be familiar with.

* **Alpha (α) Decay**: An alpha particle, which is a helium nucleus (²⁴He), is emitted. This is common in heavy nuclei. The parent nucleus's mass number decreases by 4 and its atomic number decreases by 2. Alpha particles are highly ionising but have a short range (a few cm in air) and are stopped by paper.
* **Beta-Minus (β⁻) Decay**: A neutron in the nucleus transforms into a proton, and a high-speed electron (the β⁻ particle) and an **electron antineutrino (ν̅e)** are emitted. The mass number is unchanged, but the atomic number increases by 1. Beta particles are less ionising than alpha but have a longer range (stopped by a few mm of aluminium). **Credit is only given if the antineutrino is included to conserve lepton number.**
* **Gamma (γ) Decay**: An excited nucleus releases excess energy as a high-energy photon (a gamma ray). There is no change to the mass or atomic number of the nucleus. Gamma rays are the least ionising but most penetrating, requiring thick lead or concrete to absorb them.
### 2. The Mathematics of Decay
The rate of decay, or **Activity (A)**, is measured in Becquerels (Bq), where 1 Bq = 1 decay per second. It follows an exponential decay law.
**Formula**: A = A₀e^(-λt) (Given on formula sheet)
* **A**: Activity at time t
* **A₀**: Initial activity
* **λ**: The decay constant (s⁻¹), representing the probability of a nucleus decaying per unit time.
* **t**: Time (s)
**Half-Life (T½)** is the time taken for the number of undecayed nuclei (and therefore the activity) to halve.
**Formula**: λ = ln(2) / T½ (Must memorise)
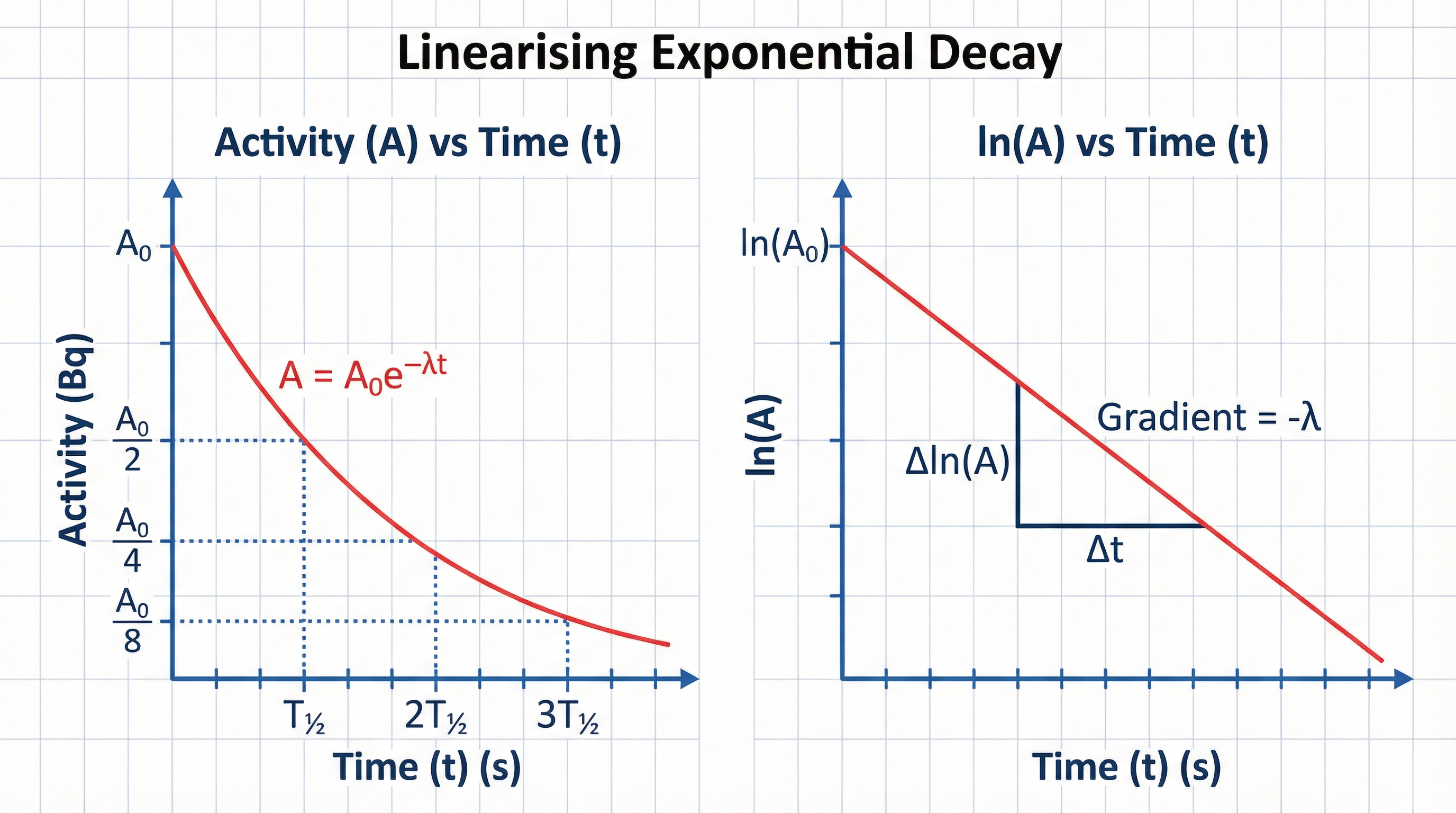
Examiners frequently test the skill of linearising decay data. By taking the natural logarithm of the decay equation, we get:
ln(A) = ln(A₀) - λt
This is in the form y = c + mx. A plot of ln(A) against t gives a straight line with a **gradient of -λ** and a y-intercept of ln(A₀). A mark is consistently awarded for stating that the gradient is equal to the negative decay constant.
### 3. The Standard Model of Particle Physics
This model classifies all known fundamental particles. It is essential to distinguish between the different families.

* **Fermions**: The matter particles. They are split into Quarks and Leptons.
* **Quarks**: Experience the strong nuclear force. They are fundamental particles that combine to form **hadrons**. You need to know the **up (u)** quark (charge +2/3) and the **down (d)** quark (charge -1/3).
* **Leptons**: Do not experience the strong nuclear force. They are fundamental particles. Examples include the **electron (e⁻)** and its corresponding **neutrino (νe)**.
### 4. Hadrons: Baryons and Mesons
Hadrons are composite particles made of quarks.
* **Baryons**: Made of **three quarks**. Protons (uud) and neutrons (udd) are baryons. All baryons are assigned a **Baryon Number (B) of +1**. Antibaryons have B = -1.
* **Mesons**: Made of a **quark-antiquark pair**. Pions (e.g., π⁺ is ud̅) are mesons. All mesons have a Baryon Number of 0.
### 5. Conservation Laws
For any particle interaction to be possible, three quantities must be conserved:
1. **Charge (Q)**
2. **Baryon Number (B)**
3. **Lepton Number (L)** (Leptons have L=+1, antileptons have L=-1)
**Exam Technique**: Always set up a table to check the conservation of Q, B, and L before and after the interaction. This structured approach is highly credited by examiners.
## Practical Applications
* **Carbon Dating**: The decay of Carbon-14 (a beta emitter with a half-life of ~5730 years) is used to date organic remains.
* **Medical Tracers**: Technetium-99m is a gamma emitter used in medical imaging. Its short half-life (6 hours) and the fact it only emits gamma rays mean the patient dose is minimised.
* **Smoke Detectors**: An alpha source (Americium-241) ionises the air in a chamber, allowing a current to flow. Smoke particles absorb the alpha particles, the current drops, and the alarm sounds.
## Podcast Episode
For an in-depth audio walkthrough of this topic, including exam tips and a quick-fire quiz, listen to our dedicated podcast episode.
