Thermal Physics and Kinetic Theory — WJEC A-Level Study Guide
Exam Board: WJEC | Level: A-Level
Thermal Physics and Kinetic Theory bridges the macroscopic world of pressure, volume, and temperature with the microscopic reality of molecules in constant random motion — and WJEC tests both levels with rigour. Mastering this topic means owning the kinetic theory pressure derivation, applying the First Law of Thermodynamics with correct sign conventions, and never, ever substituting temperature in Celsius. Get this right and you're looking at a reliable source of high-mark questions across every paper.

## Overview
Thermal Physics and Kinetic Theory (WJEC specification reference 1.4) is one of the most mathematically demanding yet conceptually elegant topics in A-Level Physics. It operates on two levels simultaneously: the **macroscopic** level, where we describe gases using measurable quantities like pressure, volume, and temperature; and the **microscopic** level, where we model gases as vast collections of randomly moving molecules and derive macroscopic behaviour from first principles.
This dual perspective is precisely what makes the topic so rewarding — and so frequently examined. WJEC questions on this topic range from short 'State' questions worth 1 mark (testing precise definitions) to extended 'Show that' derivations worth 4–5 marks (testing your ability to construct a logical argument from Newton's Second Law through to the kinetic theory pressure equation). The First Law of Thermodynamics adds a further layer, requiring candidates to handle sign conventions with care and apply energy conservation to thermodynamic processes.
This topic connects directly to **mechanics** (Newton's Second Law underpins the kinetic theory derivation), **waves and oscillations** (internal energy and temperature link to thermal equilibrium concepts), and **electricity** (energy transfer and work done appear in both contexts). Examiners frequently set synoptic questions that require you to draw on these connections.
## Key Concepts
### Concept 1: Temperature, Internal Energy, and the Ideal Gas
Temperature is a measure of the **average kinetic energy** of the particles in a substance. This is the microscopic definition — and it is the one examiners expect at A-Level. The macroscopic experience of 'hotness' is simply our perception of this average molecular kinetic energy.
The **internal energy** of a substance is defined as the sum of the random distribution of kinetic and potential energies of all the molecules it contains. For a real substance, both kinetic energy (from molecular motion) and potential energy (from intermolecular forces) contribute. However, for an **ideal gas**, the intermolecular forces are assumed to be negligible, which means the potential energy contribution is zero. Therefore, the internal energy of an ideal gas is purely kinetic.
> **Examiner note**: Candidates must explicitly state that potential energy is zero *because* intermolecular forces are negligible. Simply stating 'PE = 0' without justification will not earn the mark.
Temperature must always be expressed in **Kelvin** for gas law calculations. The conversion is: $T = \theta + 273.15$, where $\theta$ is temperature in degrees Celsius. **Absolute zero** (0 K) is the temperature at which molecular motion theoretically ceases and internal energy is at its minimum. It is the foundation of the Kelvin scale.
**The assumptions of an ideal gas** are critical knowledge:
- Molecules are point masses (negligible volume compared to the container)
- Collisions between molecules and with container walls are **perfectly elastic**
- The duration of collisions is negligible compared to the time between collisions
- There are no intermolecular forces except during collisions
- The molecules move in random directions with a range of speeds
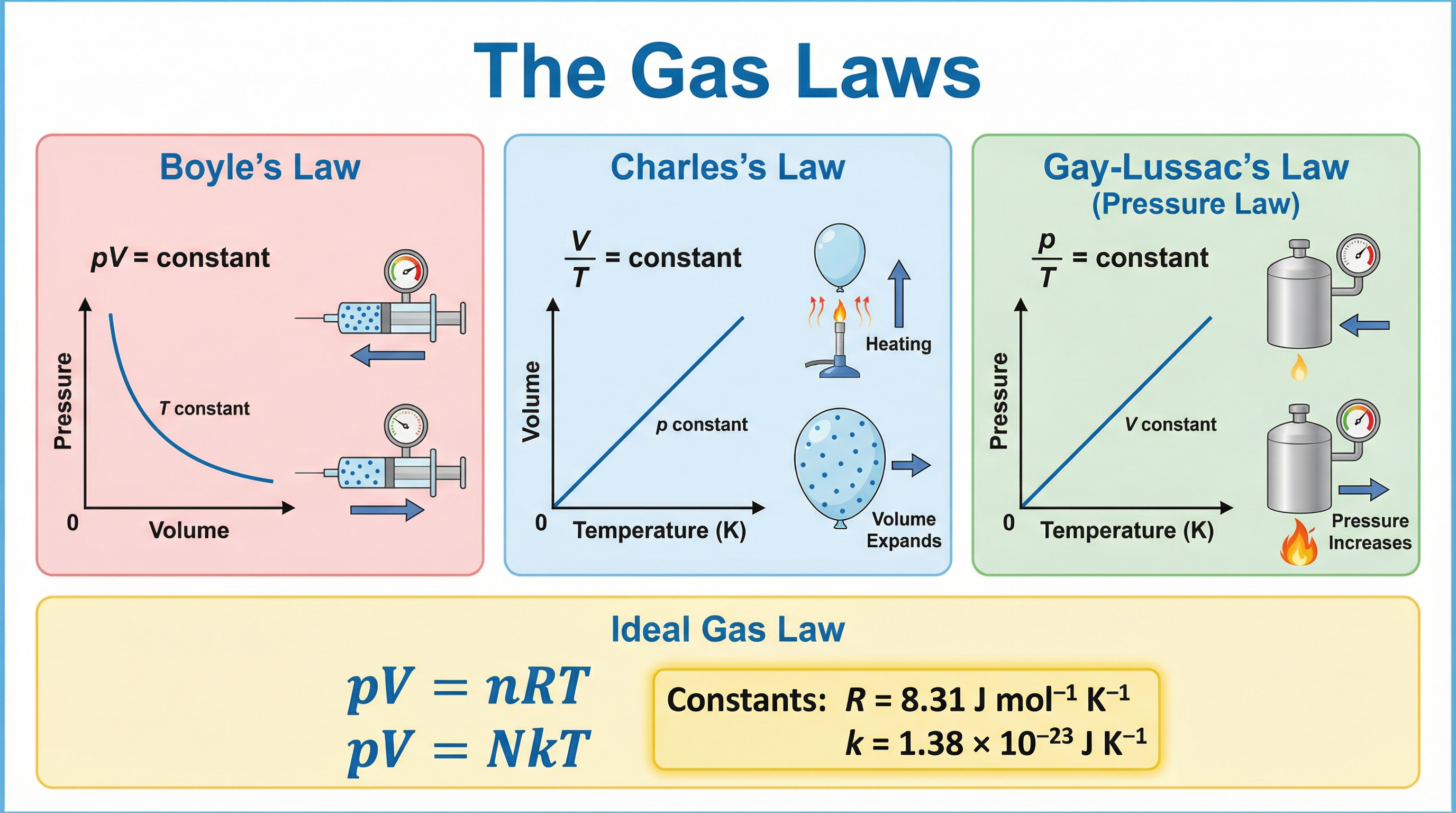
### Concept 2: The Empirical Gas Laws
The three empirical gas laws describe how the macroscopic properties of a fixed mass of gas are related when one variable is held constant.
| Law | Constant | Relationship | Equation |
|---|---|---|---|
| **Boyle's Law** | Temperature (T) | $p \propto \frac{1}{V}$ | $pV = \text{constant}$ |
| **Charles's Law** | Pressure (p) | $V \propto T$ | $\frac{V}{T} = \text{constant}$ |
| **Gay-Lussac's Law** | Volume (V) | $p \propto T$ | $\frac{p}{T} = \text{constant}$ |
Combining all three gives the **combined gas law**: $\frac{p_1 V_1}{T_1} = \frac{p_2 V_2}{T_2}$, which is enormously useful for problems where a gas changes state.
Introducing the amount of gas yields the **ideal gas equation** in two equivalent forms:
$$pV = nRT \quad \text{(using moles)}$$
$$pV = NkT \quad \text{(using number of molecules)}$$
Where $R = 8.31 \text{ J mol}^{-1} \text{K}^{-1}$ (molar gas constant) and $k = 1.38 \times 10^{-23} \text{ J K}^{-1}$ (Boltzmann constant). Note that $R = N_A k$, where $N_A = 6.02 \times 10^{23} \text{ mol}^{-1}$ is the Avogadro constant.
### Concept 3: The Kinetic Theory Pressure Derivation
This is the most frequently examined derivation in this topic. WJEC regularly sets it as a 4–5 mark 'Show that' question. You must know every step.
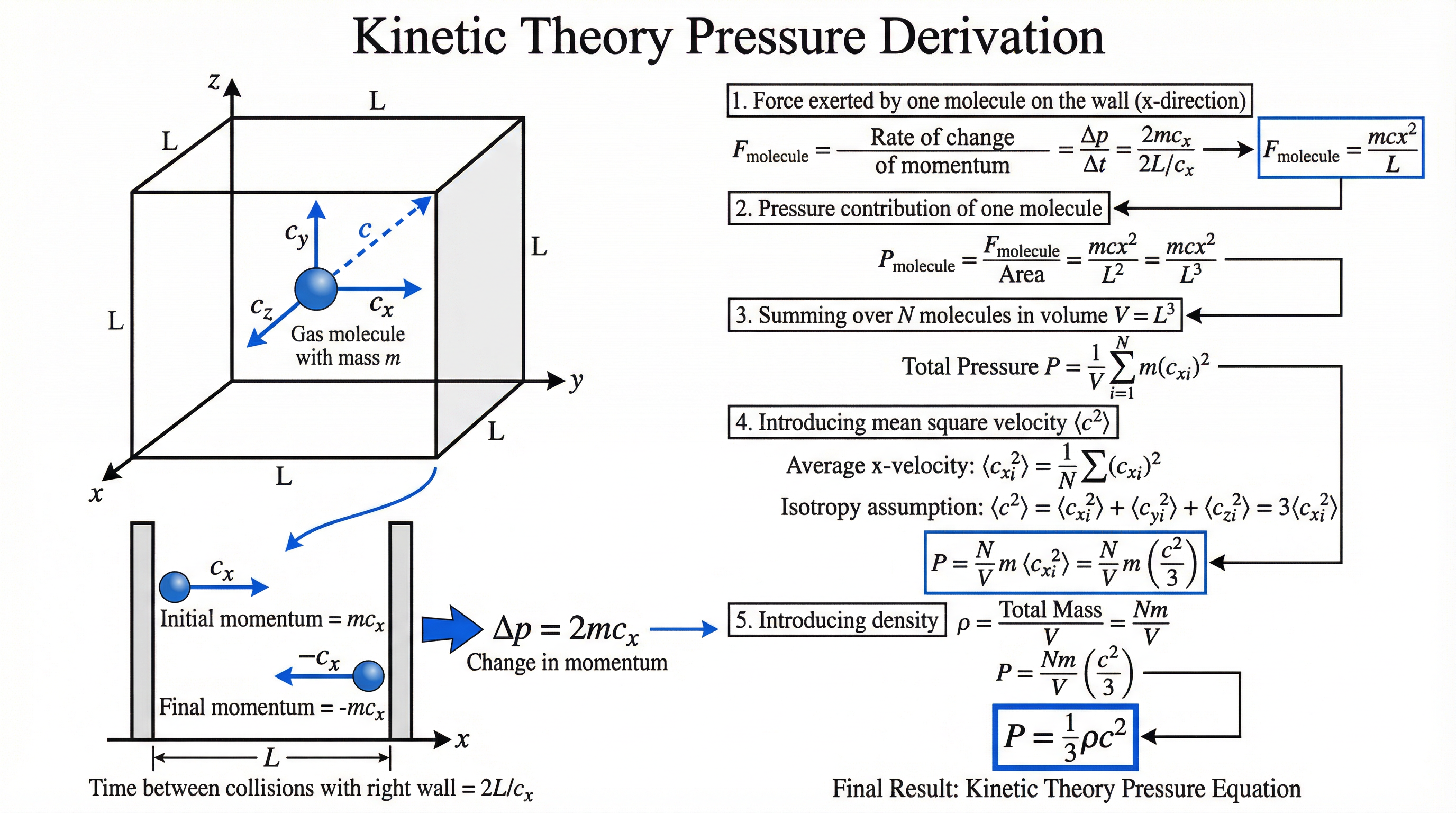
Consider a single molecule of mass $m$ moving with x-component of velocity $c_x$ inside a cubic box of side length $L$.
**Step 1 — Change in momentum:** When the molecule collides elastically with a wall perpendicular to the x-axis, its x-velocity reverses. Change in momentum = $2mc_x$.
**Step 2 — Time between collisions:** The molecule must travel a distance $2L$ before hitting the same wall again. Time = $\frac{2L}{c_x}$.
**Step 3 — Force (Newton's Second Law):** By Newton's Second Law, force = rate of change of momentum:
$$F = \frac{\Delta p}{\Delta t} = \frac{2mc_x}{2L/c_x} = \frac{mc_x^2}{L}$$
**Step 4 — Pressure from one molecule:** Pressure = Force / Area = $\frac{mc_x^2/L}{L^2} = \frac{mc_x^2}{L^3} = \frac{mc_x^2}{V}$
**Step 5 — Sum over N molecules:** Total pressure = $\frac{Nm\overline{c_x^2}}{V}$
**Step 6 — Isotropy assumption:** Since molecular motion is random and isotropic, $\overline{c_x^2} = \overline{c_y^2} = \overline{c_z^2} = \frac{1}{3}\overline{c^2}$
Therefore: $p = \frac{Nm\overline{c^2}}{3V}$
**Step 7 — Introduce density:** Since $\rho = \frac{Nm}{V}$:
$$\boxed{p = \frac{1}{3}\rho\overline{c^2}}$$
### Concept 4: Root Mean Square Speed and Temperature
By combining $pV = NkT$ with $p = \frac{Nm\overline{c^2}}{3V}$, we can derive the relationship between molecular kinetic energy and temperature:
$$\frac{1}{2}m\overline{c^2} = \frac{3}{2}kT$$
This is a profound result: the **average translational kinetic energy** of a gas molecule depends only on temperature. It is directly proportional to absolute temperature.
The **root mean square speed** is defined as: $c_{rms} = \sqrt{\overline{c^2}}$
This is NOT the same as the mean speed $\bar{c}$. The order of operations is critical: **Square** each speed, find the **Mean**, then take the **Root** — hence 'root mean square'.
From the above: $c_{rms} = \sqrt{\frac{3kT}{m}} = \sqrt{\frac{3RT}{M}}$, where $M$ is the molar mass.
### Concept 5: The First Law of Thermodynamics
The First Law is a statement of conservation of energy for a thermodynamic system:
$$\boxed{Q = \Delta U + W}$$
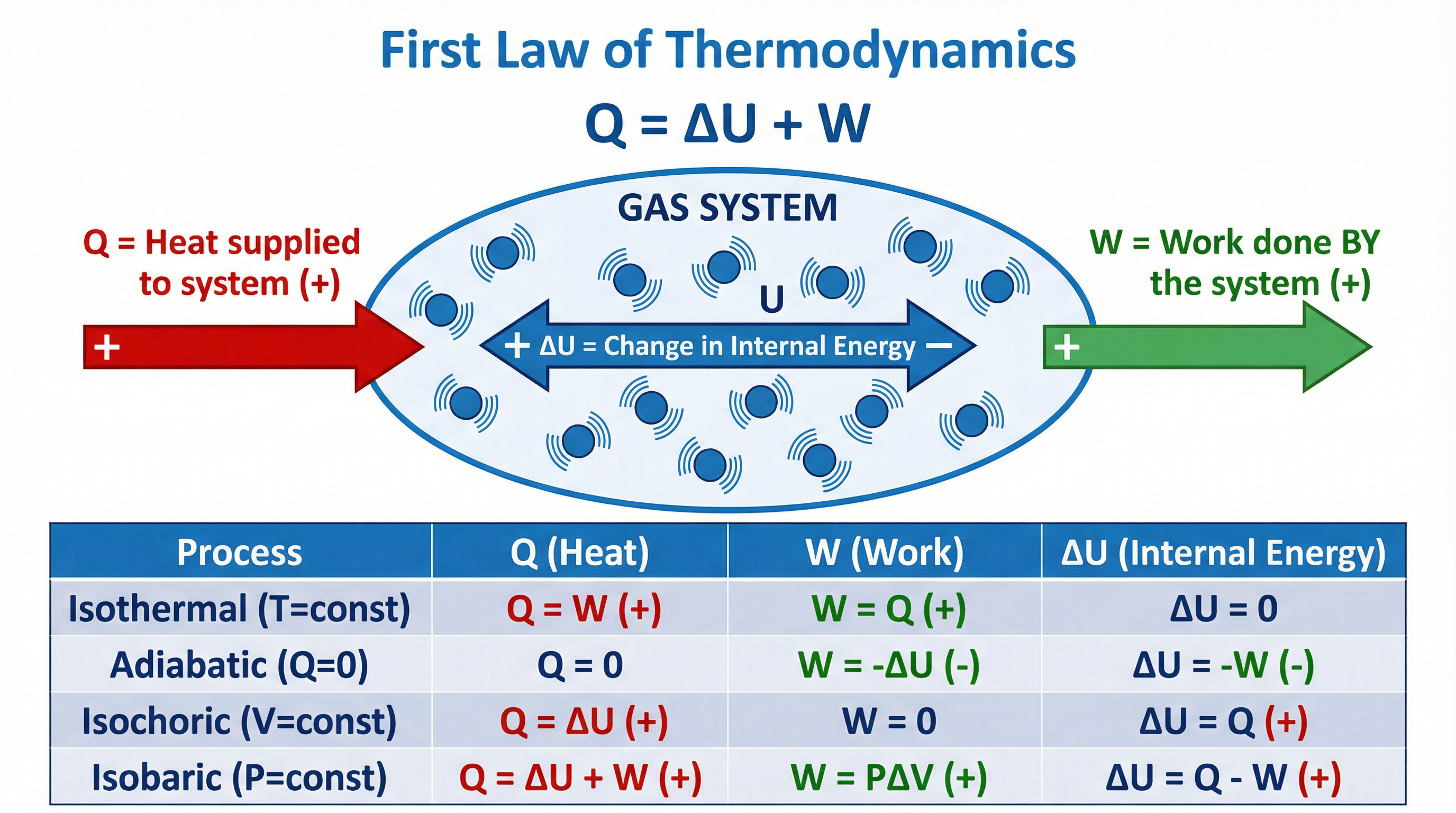
Where:
- $Q$ = heat supplied **to** the gas (positive when heat flows in)
- $\Delta U$ = increase in internal energy of the gas
- $W$ = work done **by** the gas (positive when gas expands)
The sign convention is critical:
| Situation | Sign of Q | Sign of W | Effect on ΔU |
|---|---|---|---|
| Heat supplied to gas | + | — | ΔU increases |
| Gas expands (does work) | — | + | ΔU decreases |
| Gas compressed (work done on it) | — | − | ΔU increases |
For a gas expanding against a constant pressure: $W = p\Delta V$
**Special thermodynamic processes:**
- **Isothermal** ($T$ constant): $\Delta U = 0$, so $Q = W$
- **Adiabatic** ($Q = 0$): $\Delta U = -W$ (internal energy decreases if gas expands)
- **Isochoric** ($V$ constant): $W = 0$, so $Q = \Delta U$
- **Isobaric** ($p$ constant): $W = p\Delta V$, general First Law applies
## Mathematical Relationships — Formula Summary
| Formula | Meaning | Status |
|---|---|---|
| $T = \theta + 273.15$ | Celsius to Kelvin | **Must memorise** |
| $pV = nRT$ | Ideal gas (moles) | Given on formula sheet |
| $pV = NkT$ | Ideal gas (molecules) | Given on formula sheet |
| $p = \frac{1}{3}\rho\overline{c^2}$ | Kinetic theory pressure | Given on formula sheet |
| $\frac{1}{2}m\overline{c^2} = \frac{3}{2}kT$ | KE–temperature link | Given on formula sheet |
| $Q = \Delta U + W$ | First Law | **Must memorise** |
| $W = p\Delta V$ | Work done by gas | **Must memorise** |
| $c_{rms} = \sqrt{\overline{c^2}}$ | RMS speed definition | **Must memorise** |
| $\frac{p_1V_1}{T_1} = \frac{p_2V_2}{T_2}$ | Combined gas law | **Must memorise** |
**Unit conversions commonly lost:**
- Pressure: 1 kPa = 1000 Pa; 1 atm ≈ 101,325 Pa
- Volume: 1 litre = 1 × 10⁻³ m³; 1 cm³ = 1 × 10⁻⁶ m³
- Temperature: Always convert °C → K before substituting
## Practical Applications
This topic underpins a huge range of real-world technologies. The behaviour of gases in **car engines** (compression and expansion strokes) is a direct application of the First Law. **Refrigerators and heat pumps** operate on thermodynamic cycles. The **inflation of tyres** at different temperatures follows Gay-Lussac's Law — tyre pressure increases on a hot day because the gas inside heats up at constant volume. **Atmospheric science** and weather prediction rely on the ideal gas equation to model air masses.
For the **required practical element**, candidates may be asked to verify Boyle's Law using a Boyle's Law apparatus (a sealed column of gas with a pressure gauge), or to verify Charles's Law using a capillary tube in a water bath. In both cases, examiners test: apparatus identification, method steps, expected graph shape, and sources of error.

*Listen to the full 13-minute study podcast above — covering all core concepts, exam tips, and a quick-fire recall quiz.*