Particle Model of Matter — WJEC GCSE Study Guide
Exam Board: WJEC | Level: GCSE
This guide provides a comprehensive, exam-focused breakdown of the WJEC GCSE Physics topic: Particle Model of Matter (1.2). It covers the states of matter, internal energy, density, and gas pressure, complete with multi-modal resources to secure top marks.
## Overview
The Particle Model of Matter is a cornerstone of physics, explaining the properties of solids, liquids, and gases by considering them as collections of tiny, moving particles. For your WJEC GCSE exam, a deep understanding of this topic is crucial for explaining macroscopic phenomena like changes of state, density, and gas pressure. Examiners will expect you to apply kinetic theory, distinguish between internal energy components, and analyse data from the required practical on density. This guide will equip you with the precise language, calculation skills, and exam technique needed to tackle questions on this topic with confidence. Typical exam questions range from short-answer definitions to extended 6-mark responses requiring detailed explanations of physical processes.

## Key Concepts
### Concept 1: States of Matter
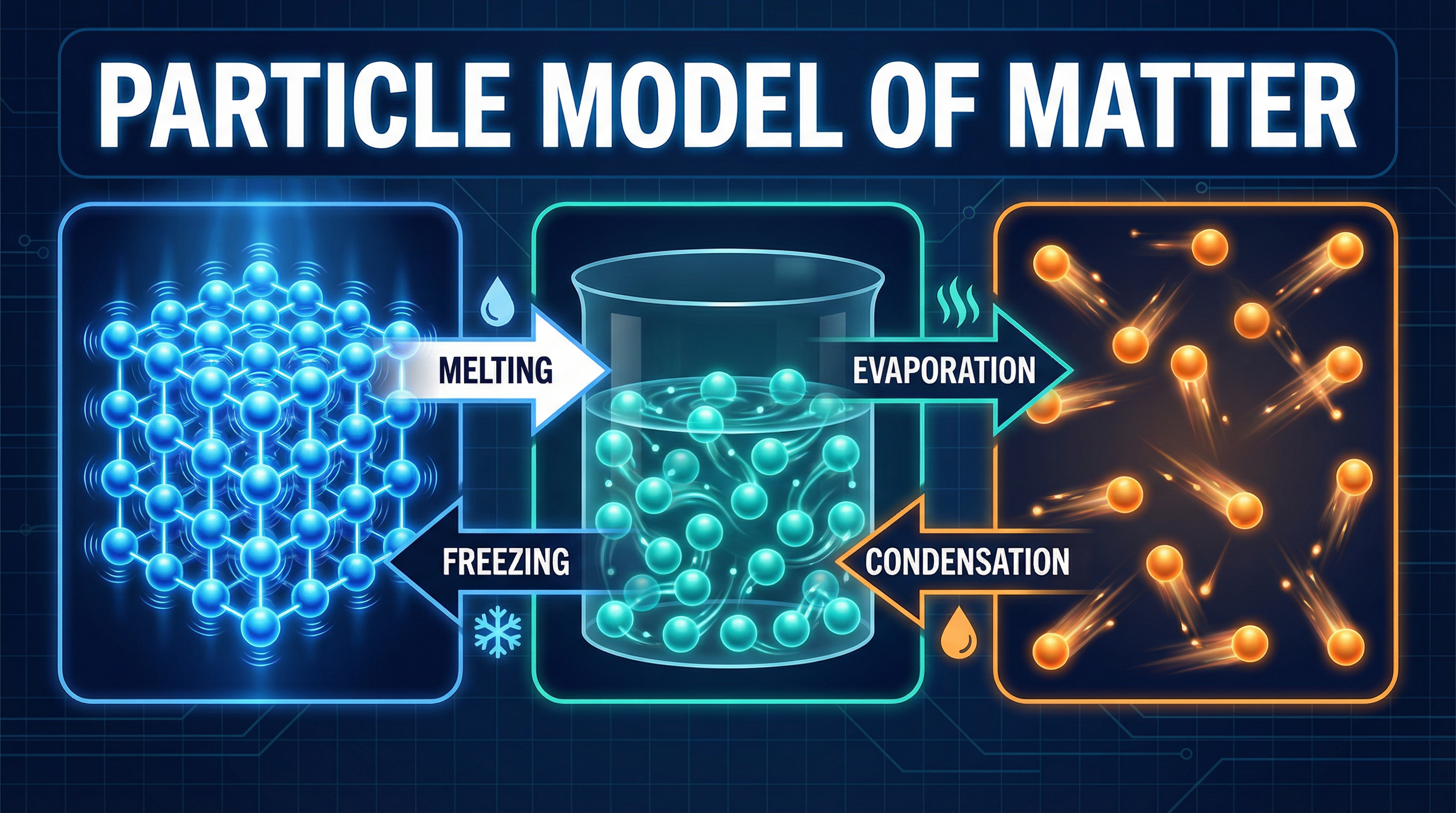
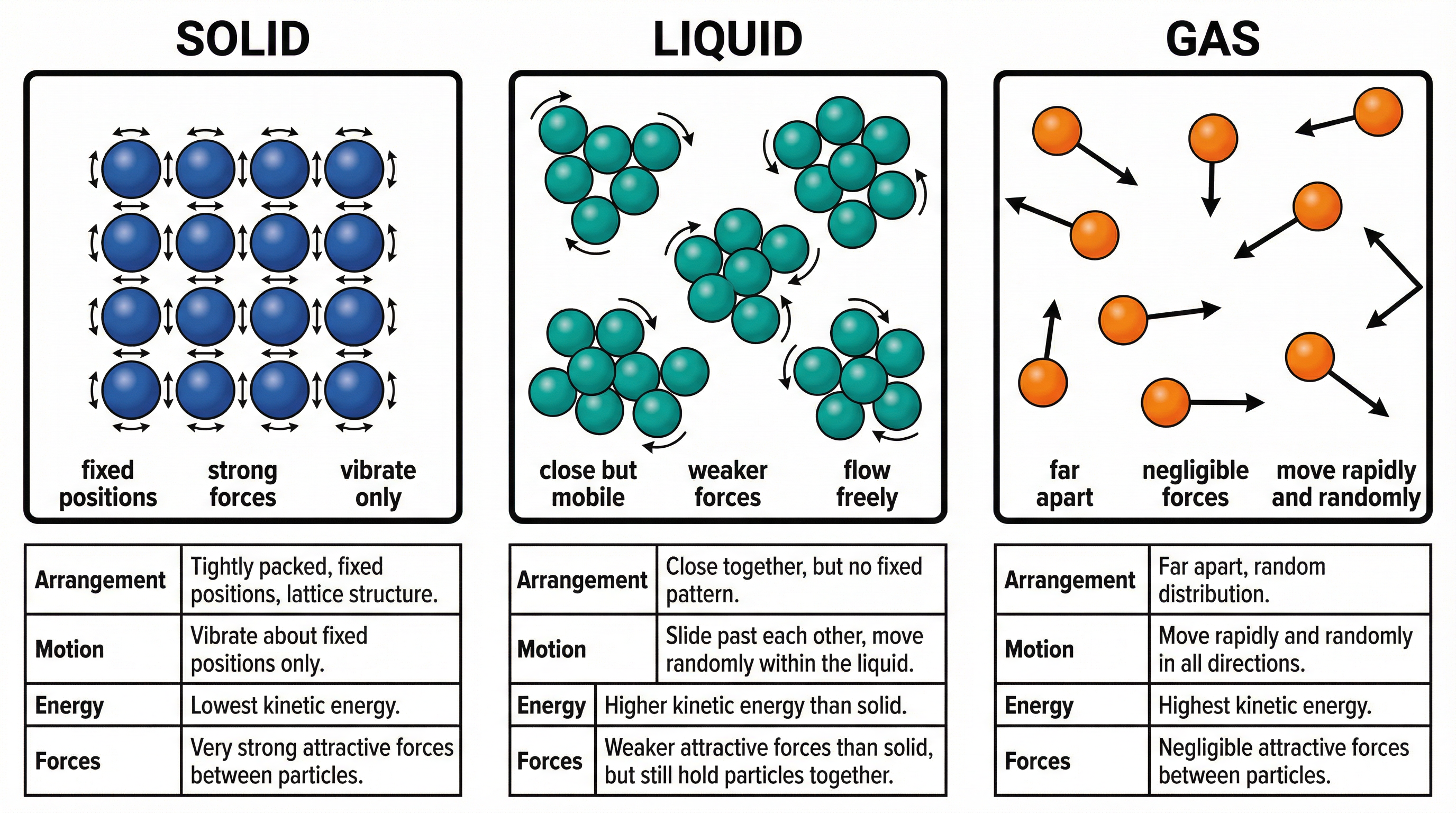
All matter is composed of particles (atoms, ions, or molecules). The arrangement and energy of these particles determine the state of the matter.
- **Solids**: Particles are held in fixed, regular positions (a lattice structure). They have strong intermolecular forces holding them together. The particles possess kinetic energy, but it is only sufficient for them to vibrate about their fixed points. This is why solids have a definite shape and volume.
- **Liquids**: Particles are still in close contact but are free to move past one another. The intermolecular forces are weaker than in solids. Their kinetic energy is higher, allowing them to flow and take the shape of their container. They have a definite volume but not a definite shape.
- **Gases**: Particles are far apart and move randomly and rapidly in all directions. The intermolecular forces are negligible. They have the highest kinetic energy of the three states. This is why gases have no definite shape or volume and will fill any container they are in. They can also be easily compressed.
### Concept 2: Internal Energy and Changes of State
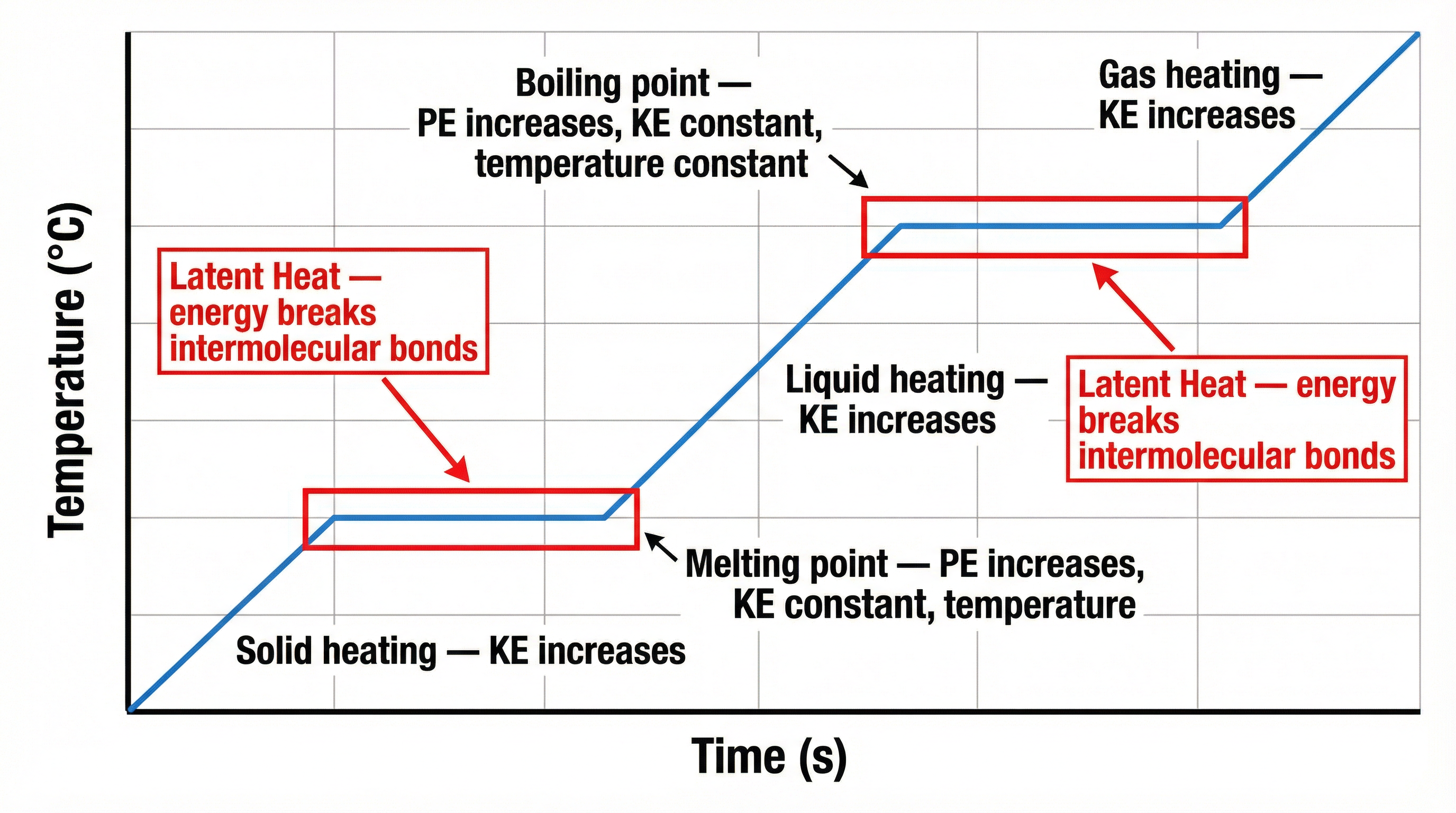
Internal energy is a critical concept. Examiners award a mark for this precise definition:
> **Internal Energy** is the total energy stored by the particles that make up a system. It is the sum of the total kinetic energy and the total potential energy of the particles.
- **Kinetic Energy (KE)**: Related to the motion of the particles. **Temperature** is a measure of the average kinetic energy of the particles. When you heat a substance and its temperature rises, the particles gain kinetic energy and move faster.
- **Potential Energy (PE)**: Related to the position of the particles and the forces between them. When a substance changes state, the energy supplied is used to overcome these forces, increasing the potential energy, not the kinetic energy.
This is why temperature remains **constant** during a change of state (melting, boiling). The energy being supplied (latent heat) is changing the potential energy of the particles, not their kinetic energy.
### Concept 3: Density
Density is a measure of how much "stuff" (mass) is packed into a given space (volume). A dense material has a lot of mass in a small volume.
- **Calculation**: The formula for density is a cornerstone of this topic.
- **ρ = m / V**
- Where: ρ (rho) is density (kg/m³), m is mass (kg), and V is volume (m³).
- **States**: Generally, solids are the densest, followed by liquids, and then gases. This is because the particles in a solid are packed most tightly, while in a gas they are very far apart.
### Concept 4: Gas Pressure (Higher Tier)
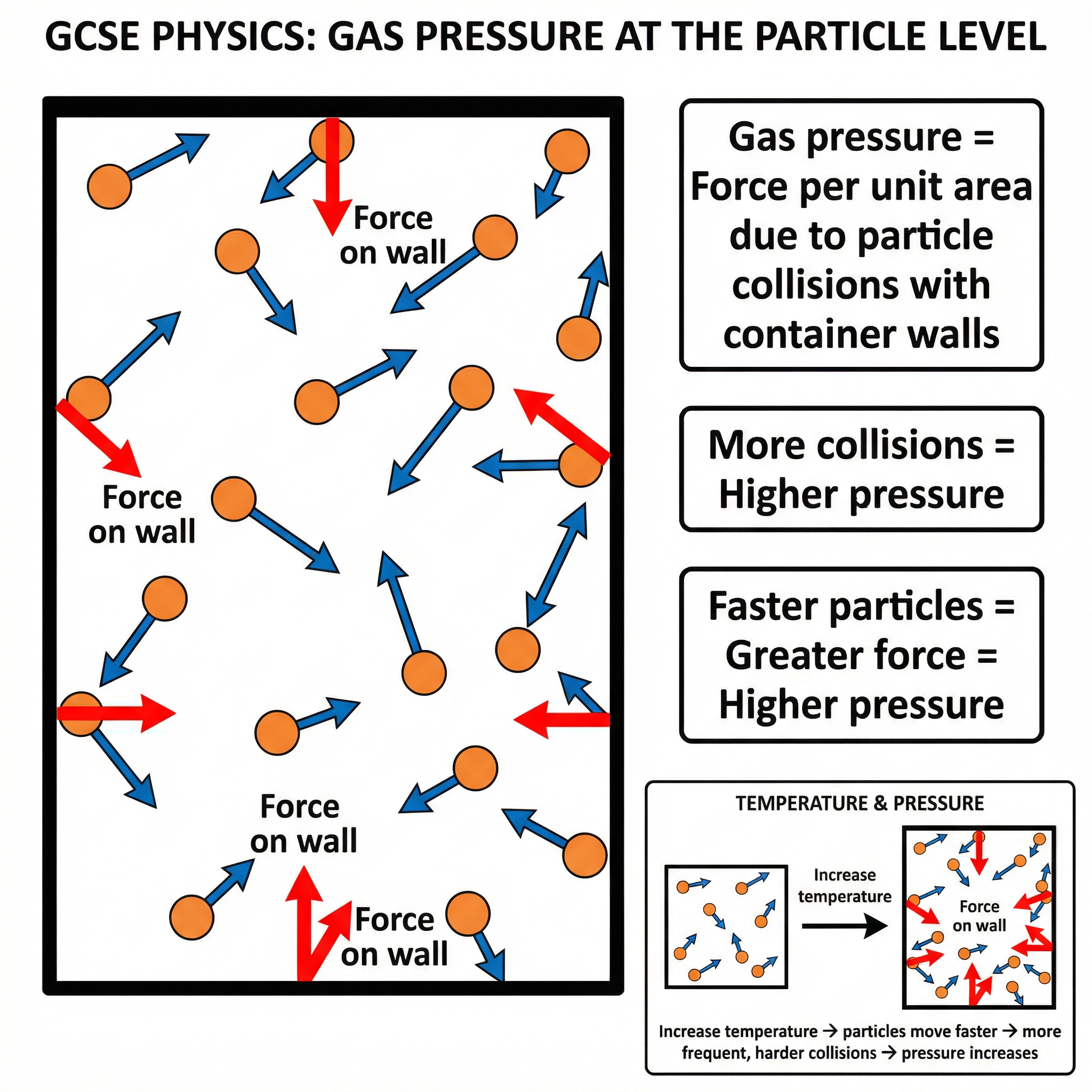
Gas pressure is caused by the collisions of gas particles with the walls of their container. Each collision exerts a small force. The sum of these forces over an area results in pressure.
- **Factors Affecting Pressure**:
- **Temperature**: Increasing the temperature of a gas increases the kinetic energy of its particles. They move faster, colliding with the walls more frequently and with greater force. This increases the pressure.
- **Volume**: Decreasing the volume of a container forces the particles closer together. They will collide with the walls more frequently, increasing the pressure.
## Mathematical/Scientific Relationships
- **Density**: **ρ = m / V** (Must memorise)
- ρ: density (kg/m³ or g/cm³)
- m: mass (kg or g)
- V: volume (m³ or cm³)
- **Specific Latent Heat**: **E = mL** (Given on formula sheet)
- E: energy for a change of state (J)
- m: mass (kg)
- L: specific latent heat (J/kg) (of fusion or vaporisation)
- **Gas Pressure & Volume (Boyle's Law)**: **pV = constant** (Given on formula sheet - Higher Tier only)
- p: pressure (Pa or N/m²)
- V: volume (m³)
- This relationship holds true only if the **temperature is constant**.
## Required Practical: Determining Density
This is a frequently tested practical. You must know the methods for both regular and irregular objects.
**Apparatus**:
- Regular object (e.g., a metal cube)
- Irregular object (e.g., a rock)
- 30 cm ruler or vernier callipers
- Digital balance
- Eureka (displacement) can
- Measuring cylinder
- Water
**Method for a Regular Solid (e.g., a cube)**:
1. Measure the mass of the cube using the digital balance. Record in kg.
2. Measure the length, width, and height of the cube using the ruler or vernier callipers. Record in m.
3. Calculate the volume using V = l × w × h.
4. Calculate the density using ρ = m / V.
**Method for an Irregular Solid (e.g., a rock)**:
1. Measure the mass of the rock using the digital balance. Record in kg.
2. Fill the Eureka can with water until it just starts to flow from the spout.
3. Place an empty measuring cylinder under the spout.
4. Carefully lower the rock into the Eureka can, ensuring it is fully submerged.
5. Collect the displaced water in the measuring cylinder. The volume of this water is equal to the volume of the rock. Record in m³ (remembering 1 ml = 1 cm³).
6. Calculate the density using ρ = m / V.
**Common Errors**:
- Misreading the scale on the ruler or measuring cylinder (parallax error).
- Incorrectly converting units (e.g., cm³ to m³).
- Splashing water when lowering the object into the Eureka can.
- Forgetting to zero the balance before measuring mass.