Study Notes

Overview
Welcome to the definitive guide for Alkanes, Topic 3.2 in the AQA A-Level Chemistry specification. Alkanes are the simplest family of hydrocarbons, but they form the bedrock of organic chemistry and are a recurring feature in exams. This topic explores their structure, physical properties, and key reactions. A thorough understanding is crucial as it provides the foundational knowledge for more complex organic topics like alkenes and alcohols. Examiners frequently test this area through a mix of short-recall questions (e.g., stating conditions for cracking) and longer, multi-mark mechanism questions (e.g., free-radical substitution). Mastering the precise language required, such as explaining boiling point trends in terms of intermolecular forces, is essential for achieving full credit.
Key Concepts
Concept 1: Structure and Bonding in Alkanes
Alkanes are saturated hydrocarbons, meaning they contain only carbon and hydrogen atoms joined by single covalent bonds. Their general formula is CnH2n+2. Each carbon atom in an alkane forms four single covalent bonds in a tetrahedral arrangement, with bond angles of 109.5°. This tetrahedral shape is due to the repulsion between the four bonding pairs of electrons around the central carbon atom. The C-C and C-H bonds are sigma (σ) bonds, formed by the direct, head-on overlap of atomic orbitals, which allows for free rotation around the bond axis. This free rotation is why longer-chain alkanes are not rigid, straight molecules but flexible chains.
Concept 2: Intermolecular Forces and Physical Properties
The physical properties of alkanes, such as their boiling points, are determined by the weak van der Waals forces (also known as London dispersion forces) between molecules. These are the only intermolecular forces present because the C-H bond has very low polarity.
- Boiling Point Trends: As the carbon chain length increases, the number of electrons in the molecule increases. This leads to stronger temporary and induced dipoles, resulting in stronger van der Waals forces between molecules. Consequently, more energy is required to overcome these forces, leading to an increase in boiling point. For full marks, candidates must explicitly link stronger forces to the need for more energy.
- Effect of Branching: Branched-chain isomers have lower boiling points than their straight-chain counterparts. The branches prevent the molecules from packing closely together, reducing the surface area of contact between them. This weakens the van der Waals forces, meaning less energy is needed to separate the molecules.
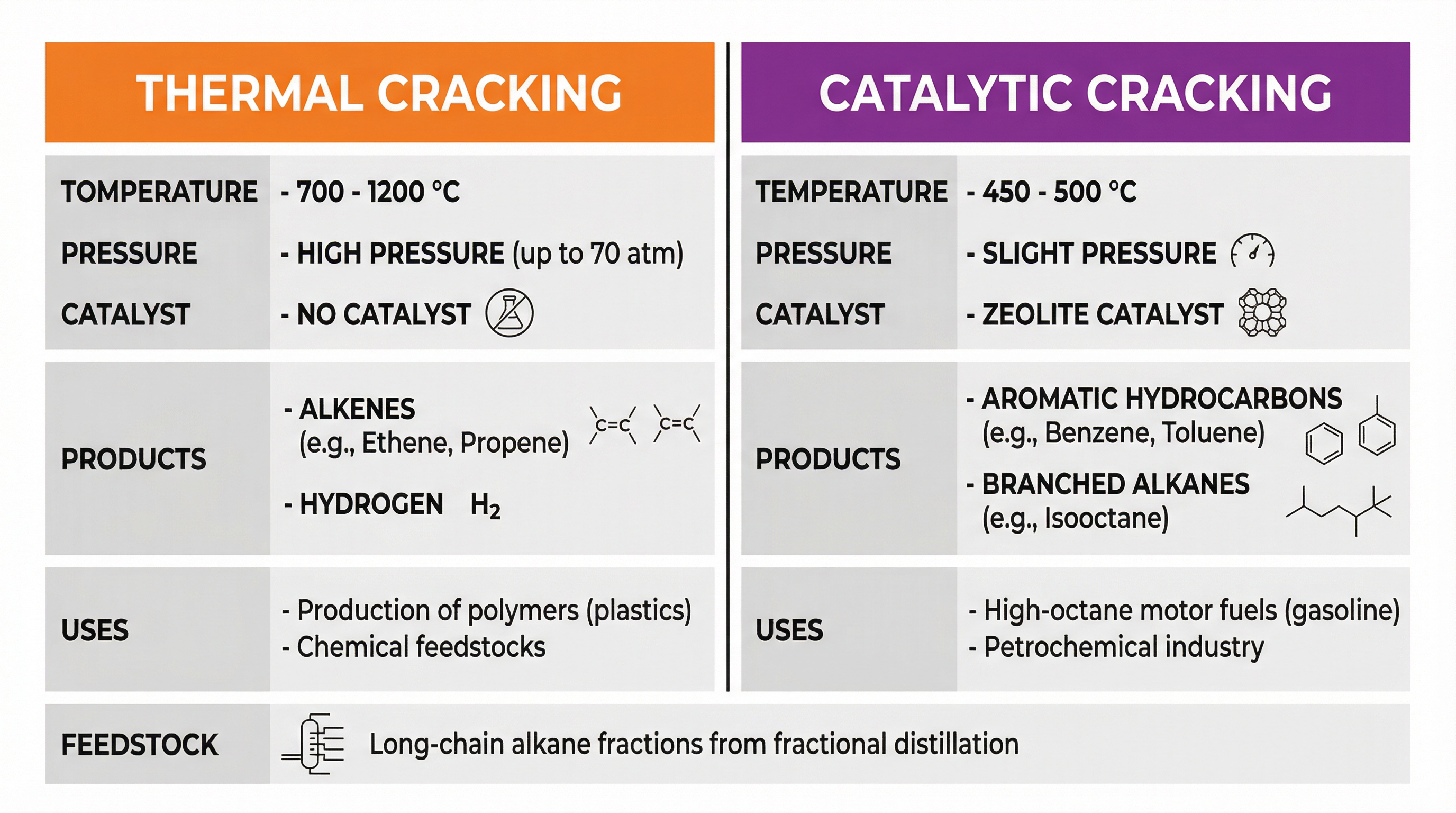
Concept 3: Cracking of Alkanes
Cracking is the process of breaking down large, less useful long-chain alkanes into smaller, more valuable molecules like short-chain alkanes, alkenes, and hydrogen. This is a vital industrial process for producing petrol and the alkenes needed for the polymer industry.
- Thermal Cracking: This process uses high temperatures (700-1200°C) and high pressures (up to 70 atm) without a catalyst. It primarily produces a high percentage of alkenes (e.g., ethene for making poly(ethene)) and hydrogen.
- Catalytic Cracking: This process uses a lower temperature (450-500°C), a slight pressure, and a zeolite catalyst. Zeolites are crystalline aluminosilicates with a porous structure, providing a large surface area for the reaction. Catalytic cracking is mainly used to produce aromatic hydrocarbons and branched alkanes, which are components of high-octane motor fuels.
Concept 4: Combustion
Alkanes are widely used as fuels because they release a large amount of energy upon combustion.
- Complete Combustion: In a plentiful supply of oxygen, alkanes burn to produce carbon dioxide and water. Example (Propane): C₃H₈(g) + 5O₂(g) → 3CO₂(g) + 4H₂O(l)
- Incomplete Combustion: In a limited supply of oxygen, alkanes undergo incomplete combustion, producing carbon monoxide (CO) and/or soot (C), along with water. Carbon monoxide is highly toxic as it binds irreversibly to haemoglobin in red blood cells, preventing the transport of oxygen.
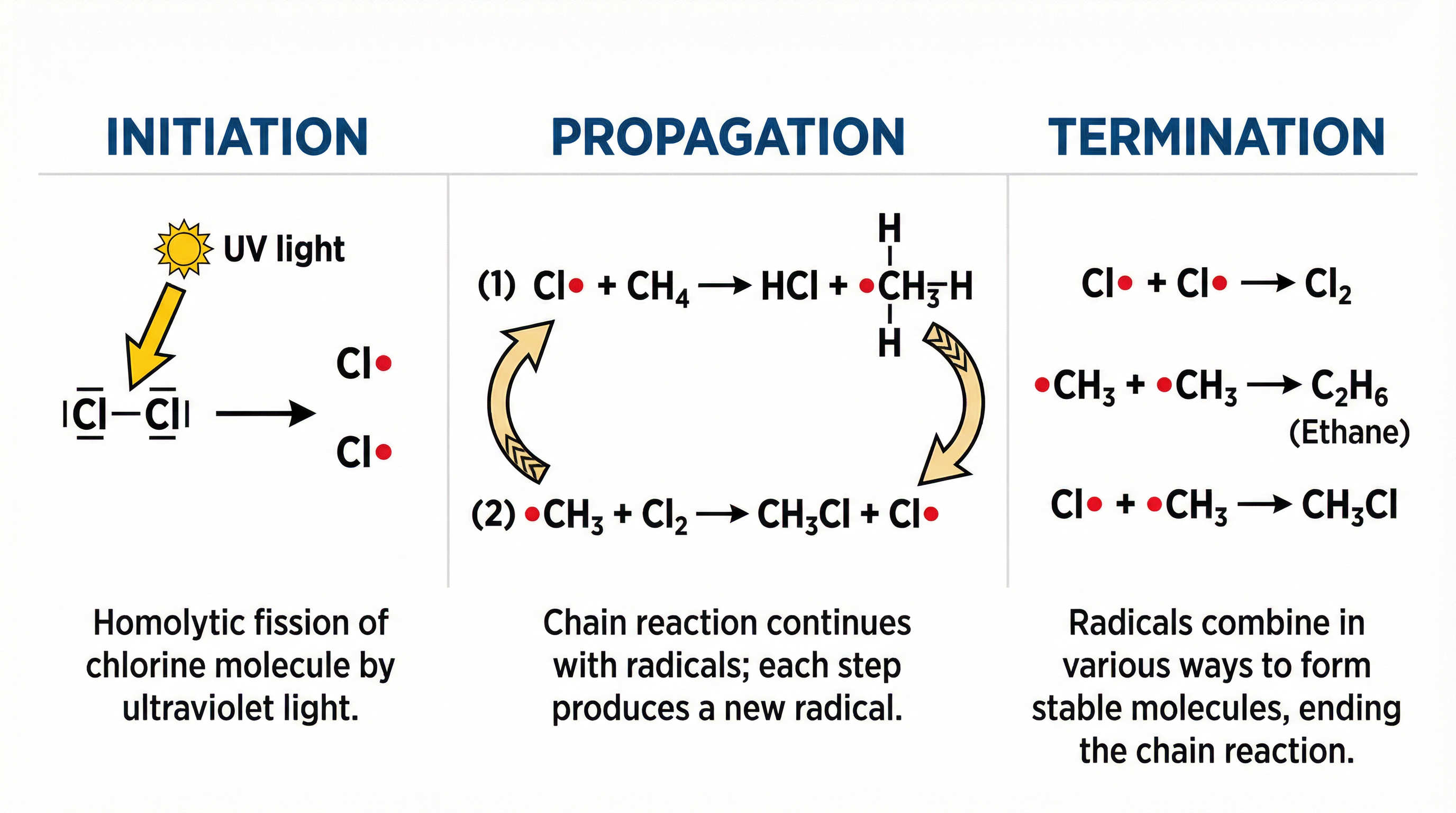
Concept 5: Free-Radical Substitution
Alkanes react with halogens (e.g., chlorine or bromine) in the presence of ultraviolet (UV) light. This is a substitution reaction where a hydrogen atom is replaced by a halogen atom. The reaction proceeds via a free-radical mechanism, which occurs in three distinct stages.
Mathematical/Scientific Relationships
- General Formula for Alkanes: CnH2n+2 (Must memorise)
- Complete Combustion Equation: Alkane + Oxygen → Carbon Dioxide + Water (Must be able to balance for any given alkane)
- Incomplete Combustion Equation: Alkane + Oxygen → Carbon Monoxide + Water (Must be able to balance)
- Flue Gas Desulfurisation:
- CaO(s) + SO₂(g) → CaSO₃(s) (Must memorise)
- CaCO₃(s) + SO₂(g) + ½O₂(g) → CaSO₄(s) + CO₂(g) (Must memorise)
- Catalytic Converter Reaction: 2NO(g) + 2CO(g) → N₂(g) + 2CO₂(g) (Must memorise)
Practical Applications
Alkanes are fundamental to modern life. Methane is the main component of natural gas, used for heating and electricity generation. Propane and butane are used as LPG (liquefied petroleum gas) for heating and cooking. Longer chain alkanes are found in petrol, diesel, and jet fuel. The cracking of alkanes is essential for producing the high-octane petrol required for modern cars and the alkenes that are the building blocks of the plastics industry. The concept of flue gas desulfurisation is applied in power stations to reduce acid rain by removing sulfur dioxide from their emissions.
Visual Resources
2 diagrams and illustrations
Interactive Diagrams
2 interactive diagrams to visualise key concepts
Flowchart of the Free-Radical Substitution mechanism, showing the cycle of propagation and the termination pathways.
Decision flowchart comparing the conditions, products, and uses of Thermal and Catalytic Cracking.
Worked Examples
3 detailed examples with solutions and examiner commentary
Practice Questions
Test your understanding — click to reveal model answers
State the general formula for an alkane and the shape and bond angle around each carbon atom. (3 marks)
Hint: Think about the number of bonds each carbon forms and how the electron pairs repel each other.
Explain why catalytic cracking is an important industrial process. (4 marks)
Hint: Think about the supply and demand of different hydrocarbon fractions from crude oil.
Further substitution can occur during the chlorination of methane, forming dichloromethane (CH₂Cl₂). Outline the two propagation steps that would lead to the formation of a •CHCl₂ radical. (2 marks)
Hint: The starting organic molecule is now chloromethane (CH₃Cl), not methane.
Write a balanced equation for the incomplete combustion of pentane (C₅H₁₂) to form carbon monoxide and water. (2 marks)
A student states that 'butane has a higher boiling point than 2-methylpropane because it has a longer chain'. Criticise this statement. (3 marks)
Hint: Both molecules are isomers. What is the key difference in their shape and how does that affect intermolecular forces?