Study Notes

Overview
Organic chemistry is the study of carbon compounds, a vast and vital field that underpins biology, medicine, and materials science. For your exam, this topic is a high-yield area, often accounting for 15-20% of the total marks. You will be expected to not only recall facts but to apply your knowledge to solve problems, such as deducing structures from analytical data or proposing synthetic routes for new molecules.
This guide covers the fundamental principles of organic chemistry, including nomenclature, isomerism, and the characteristic reactions of key functional groups like alkanes, alkenes, and alcohols. Success here requires a shift in thinking: rather than just memorising equations, you must understand the mechanisms—the step-by-step movement of electrons that drives reactions. This deep understanding will allow you to tackle even the most novel application questions the examiners throw at you.
Key Concepts
Concept 1: Homologous Series and Functional Groups
Organic compounds are grouped into families called homologous series. Members of a series share the same general formula and functional group, which dictates their chemical properties. They differ only by a -CH₂- unit in the carbon chain.
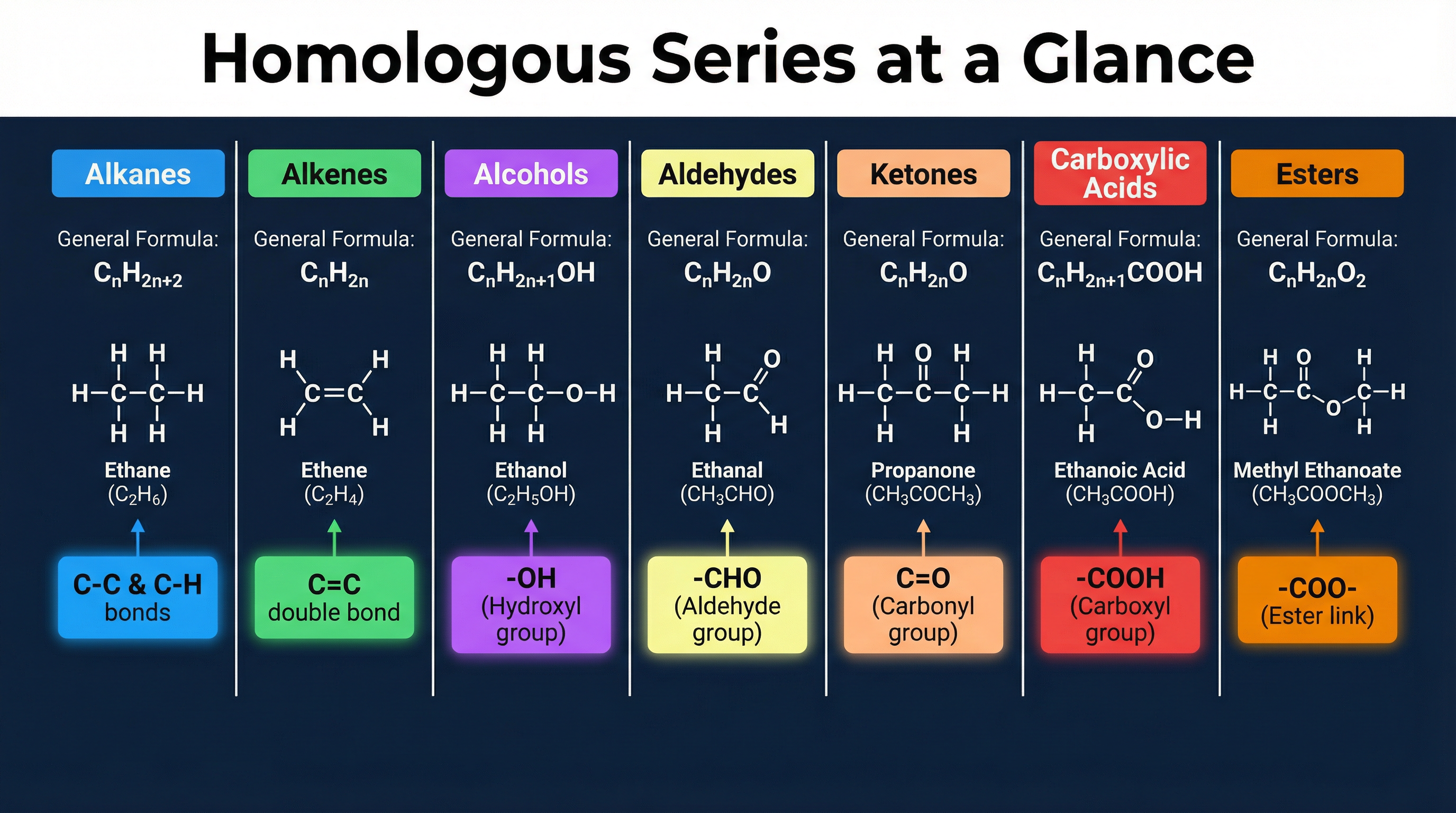
- Functional Group: The specific atom or group of atoms responsible for the characteristic chemical reactions of a compound (e.g., the -OH group in alcohols).
- General Formula: An algebraic formula that describes any member of a family (e.g., CₙH₂ₙ₊₂ for alkanes).
Why this matters: If you know how ethanol reacts, you can predict how propanol or butanol will react because they are in the same series. This pattern recognition is crucial for the exam.
Concept 2: Isomerism
Isomers are molecules with the same molecular formula but different arrangements of atoms. There are two main types you need to master:
- Structural Isomerism: Atoms are bonded in a different order.
- Chain isomerism: Different carbon skeletons (e.g., butane vs. methylpropane).
- Position isomerism: Functional group is in a different place (e.g., propan-1-ol vs. propan-2-ol).
- Functional group isomerism: Different functional groups entirely (e.g., aldehydes vs. ketones).
- Stereoisomerism: Atoms have the same connectivity but different spatial arrangement.
- E/Z Isomerism: Occurs in alkenes due to restricted rotation around the C=C double bond.
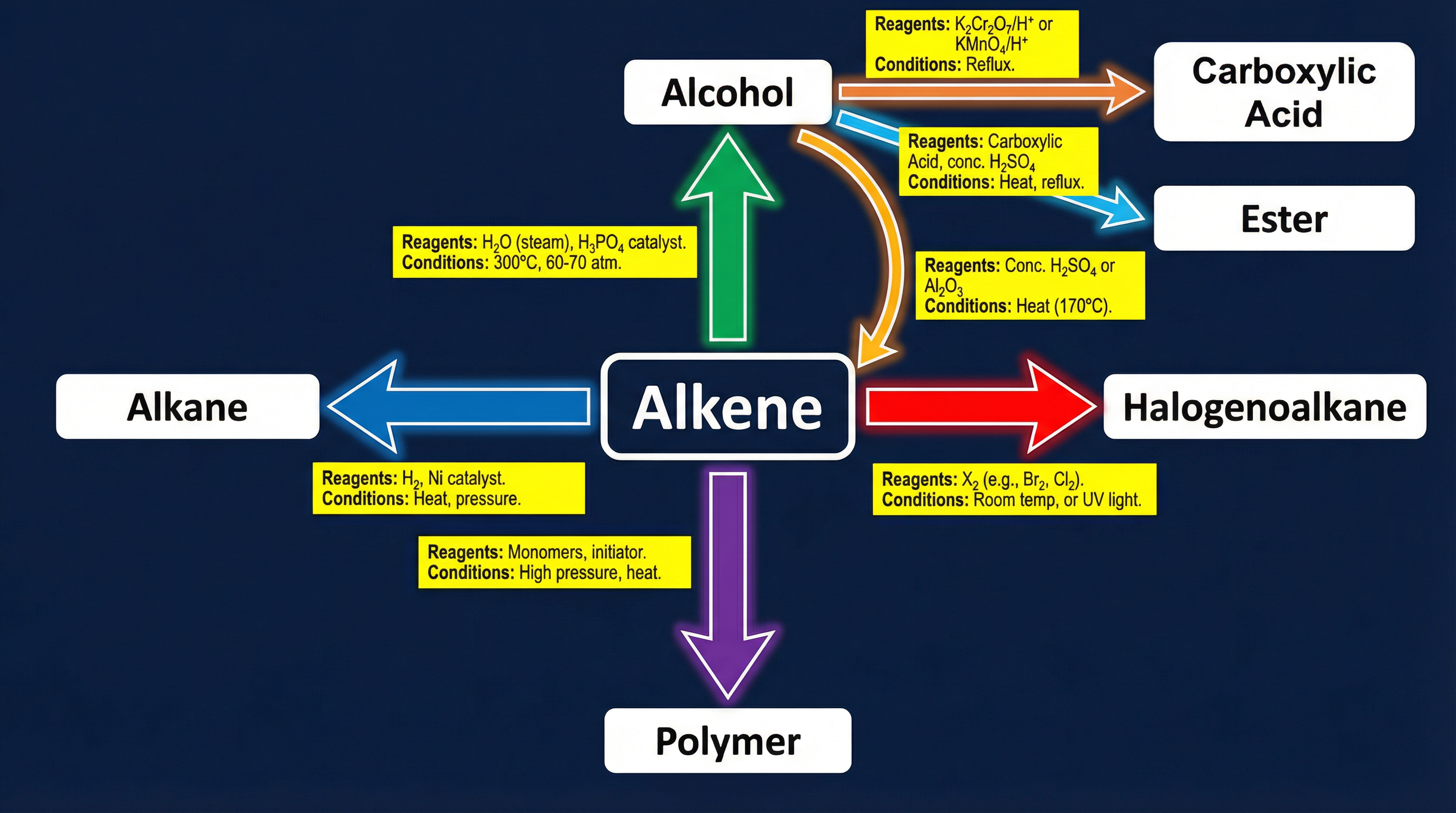
Concept 3: Reaction Mechanisms
Examiners love mechanisms because they test your understanding of how reactions happen. You must be able to draw these precisely using curly arrows to show the movement of electron pairs.
- Free Radical Substitution: Typical of alkanes reacting with halogens (UV light required).
- Electrophilic Addition: Typical of alkenes, where the high electron density of the double bond attacks an electrophile.
- Nucleophilic Substitution: Typical of halogenoalkanes, where a nucleophile replaces the halogen atom.
Mathematical/Scientific Relationships
General Formulas
- Alkanes: C_nH_{2n+2}
- Alkenes: C_nH_{2n}
- Alcohols: C_nH_{2n+1}OH
- Carboxylic Acids: C_nH_{2n+1}COOH
Atom Economy
In organic synthesis, we want to maximise the amount of useful product.
\text{Atom Economy} = \frac{\text{Molecular Mass of Desired Product}}{\text{Sum of Molecular Masses of All Reactants}} \times 100%
High atom economy means less waste, which is a key principle of Green Chemistry.
Practical Applications
Organic chemistry isn't just theory; it's the engine of the modern world.
- Fractional Distillation: Separating crude oil into useful fractions like petrol, kerosene, and diesel.
- Polymerisation: Turning small alkene monomers into durable plastics for packaging, clothing, and construction.
- Biofuels: Fermenting sugars with yeast to produce ethanol, a renewable fuel source.
"
Worked Examples
3 detailed examples with solutions and examiner commentary
Practice Questions
Test your understanding — click to reveal model answers
Name the following compound: CH3-CH(CH3)-CH2-CH=CH2
Hint: Number the chain from the end that gives the double bond the lowest number.
Describe a chemical test to distinguish between butane and but-2-ene. State the reagent and the observation for each.
Hint: Think about the test for unsaturation.
Outline the mechanism for the reaction of ethane with chlorine in the presence of UV light to form chloroethane.
Hint: Remember the three stages: I, P, T.
Explain why tertiary alcohols cannot be oxidised by acidified potassium dichromate(VI).
Hint: Look at the carbon atom attached to the -OH group.
Calculate the atom economy for the production of ethene from the dehydration of ethanol: C2H5OH -> C2H4 + H2O.
Hint: Desired product is ethene. Use atomic masses: C=12, H=1, O=16.