Study Notes
Overview
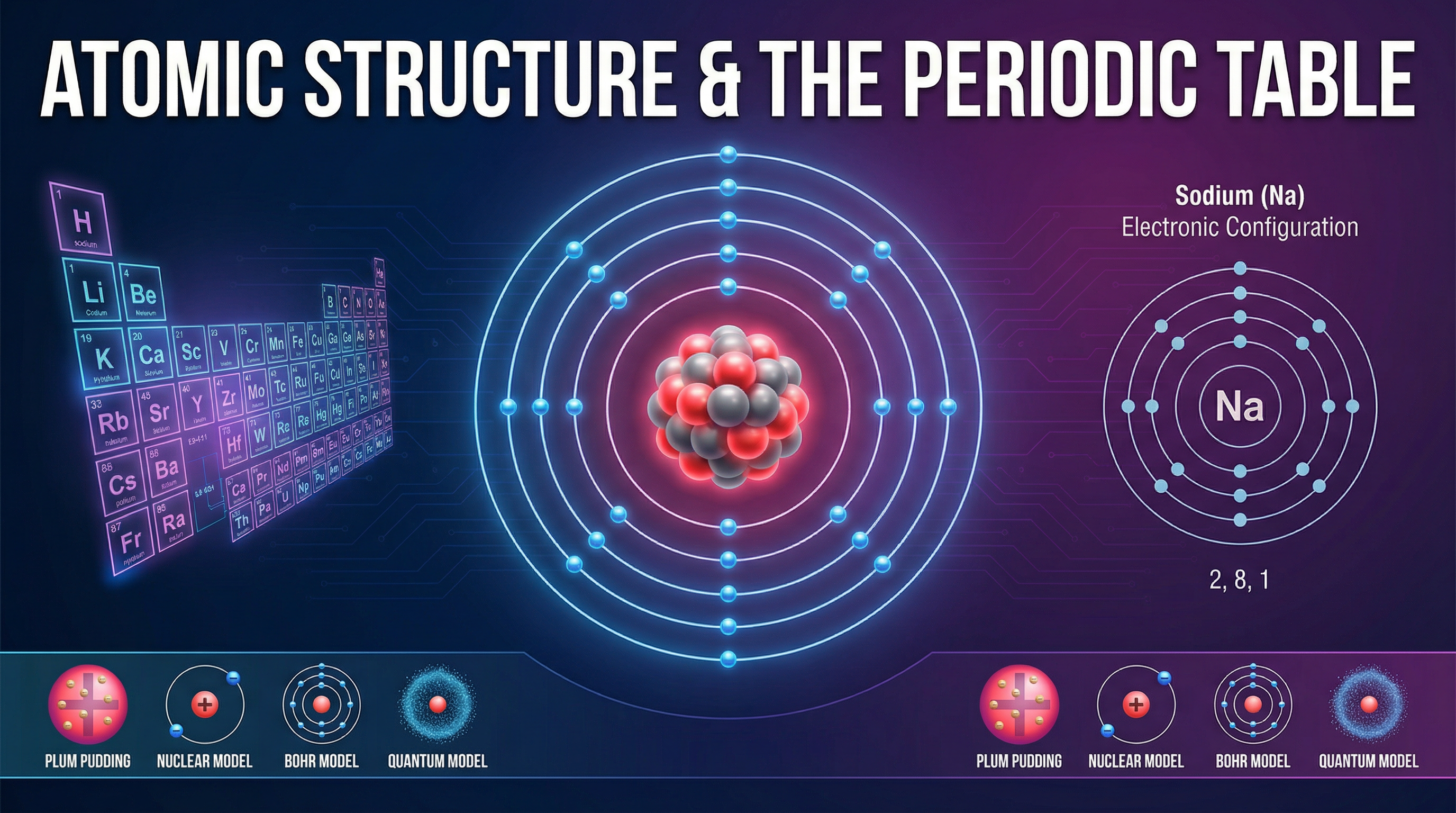
Welcome to Topic 4.1: Atomic Structure and the Periodic Table. This topic is the absolute foundation of your GCSE Chemistry journey. It explores the fundamental building blocks of all matter—atoms—and how they are organised. Understanding the subatomic particles (protons, neutrons, and electrons) and their arrangement is crucial, as it directly explains why elements behave the way they do.
This topic is heavily synoptic. The concepts you learn here will be applied repeatedly when you study bonding, quantitative chemistry, and chemical changes. Examiners frequently test your ability to link atomic structure to the reactivity trends seen in the periodic table. Expect a mix of calculation questions (determining subatomic particles), short-answer recall questions (definitions of isotopes), and longer explanatory questions (comparing the properties of different groups).
Listen to the companion podcast to reinforce these concepts:
Key Concepts
Concept 1: Subatomic Particles and the Nucleus
Atoms are the smallest part of an element that can exist independently. However, they are composed of even smaller subatomic particles: protons, neutrons, and electrons. The protons and neutrons are tightly packed in a central, dense nucleus, while electrons orbit this nucleus in distinct energy levels (shells).
Understanding the relative masses and charges of these particles is essential. Protons have a relative mass of 1 and a relative charge of +1. Neutrons also have a relative mass of 1 but carry no charge (0). Electrons have a negligible mass (often approximated as 1/2000 or 0) and a relative charge of -1. Because atoms contain equal numbers of protons and electrons, they have no overall electrical charge.
Example: A sodium atom has 11 protons and 11 electrons. The 11 positive charges cancel out the 11 negative charges, making the atom neutral.
Concept 2: Atomic Number, Mass Number, and Isotopes
The identity of an element is determined solely by its atomic number (the number of protons). If you change the number of protons, you change the element. The mass number represents the total number of protons and neutrons combined.
Isotopes are a crucial concept that examiners love to test. Isotopes are defined as atoms of the same element that have the same number of protons but different numbers of neutrons. This means they have the same atomic number but different mass numbers. Because they have the same number of electrons, isotopes of an element react in exactly the same way chemically.
Example: Carbon-12 has 6 protons and 6 neutrons. Carbon-14 has 6 protons and 8 neutrons. Both are carbon, but their mass numbers differ.
Concept 3: Electronic Structure
Electrons occupy specific energy levels or shells around the nucleus. The arrangement of these electrons follows strict rules: the first shell can hold a maximum of 2 electrons, while the second and third shells can hold up to 8 electrons each. The electronic structure can be written as numbers (e.g., 2,8,1) or drawn as a diagram.

The number of electrons in the outermost shell is particularly important because it determines the element's group in the periodic table and its chemical properties. Elements in the same group have the same number of outer electrons, which is why they react similarly.
Concept 4: The Periodic Table and Group Trends
The modern periodic table arranges elements in order of increasing atomic number. Elements with similar properties are placed in vertical columns called groups.
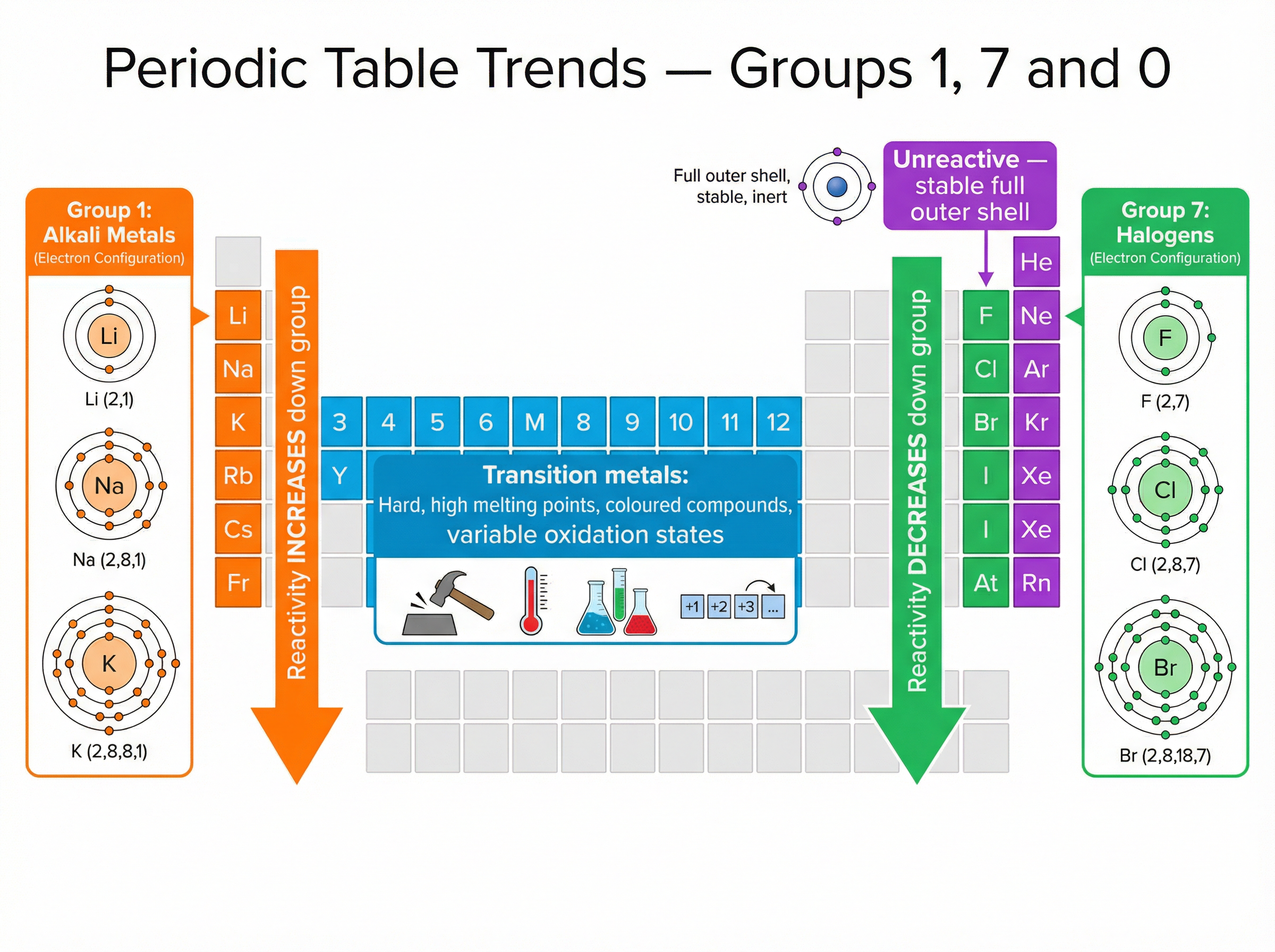
Group 1 (Alkali Metals): These have one outer electron. They react by losing this electron to form a +1 ion. Reactivity increases down the group because the outer electron gets further from the nucleus, increasing shielding and making the electron easier to lose.
Group 7 (Halogens): These have seven outer electrons. They react by gaining one electron to form a -1 ion. Reactivity decreases down the group because the outer shell is further from the nucleus, making it harder to attract an incoming electron.
Group 0 (Noble Gases): These elements have a full outer shell (8 electrons, or 2 for helium). Because their electron arrangement is already stable, they are highly unreactive (inert).
Concept 5: The Development of the Atomic Model
Scientific models evolve as new evidence is discovered. The model of the atom has changed significantly over time.
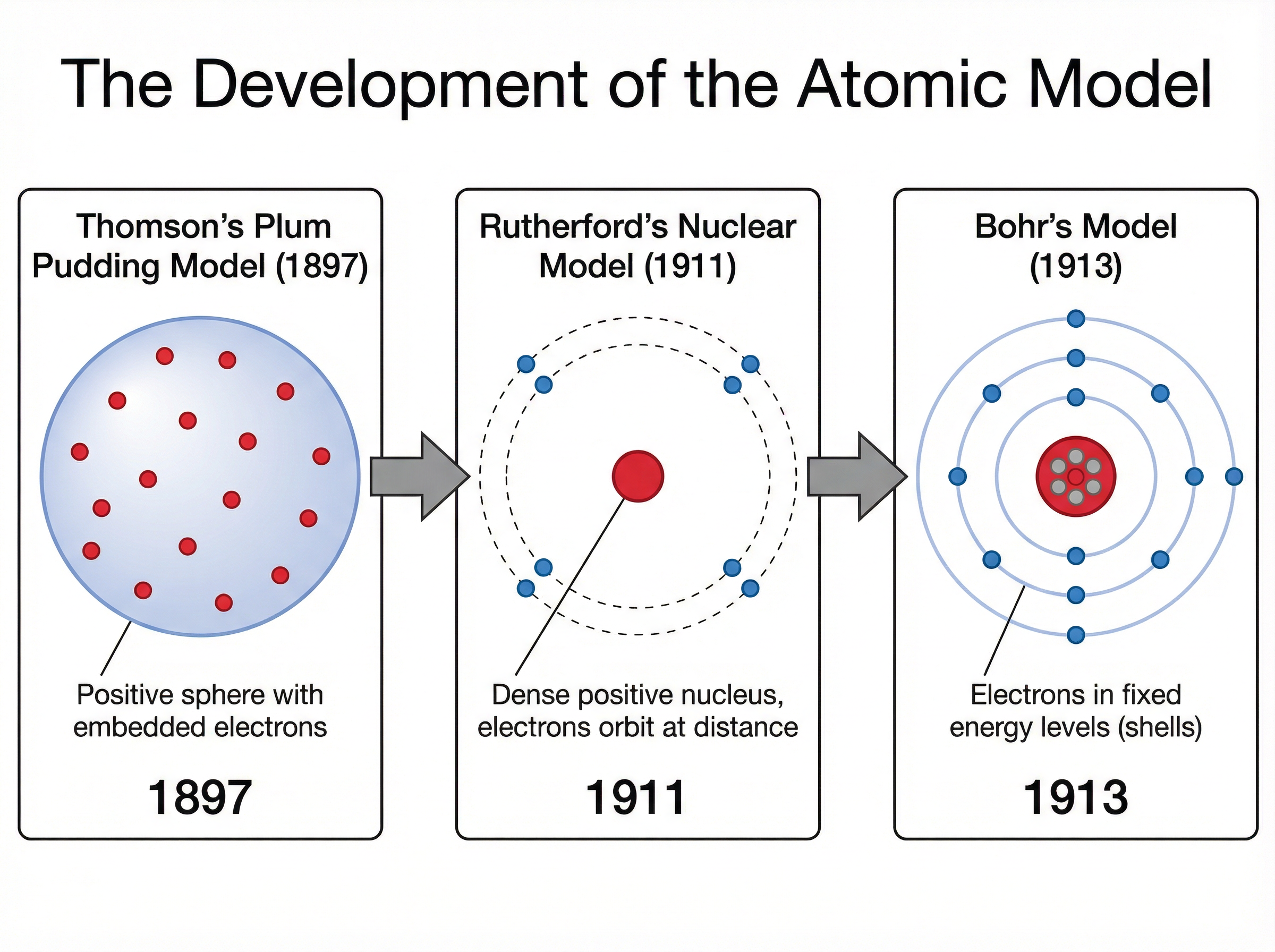
- Plum Pudding Model (Thomson): Suggested the atom was a ball of positive charge with negative electrons embedded in it.
- Nuclear Model (Rutherford): The alpha particle scattering experiment showed that most of the atom is empty space, with a tiny, dense, positively charged nucleus at the centre.
- Bohr Model: Niels Bohr adapted the nuclear model by suggesting that electrons orbit the nucleus at specific distances in distinct energy levels (shells).
- Discovery of the Neutron (Chadwick): James Chadwick later provided evidence for the existence of neutrons within the nucleus.
Mathematical/Scientific Relationships
- Number of Neutrons = Mass Number - Atomic NumberUse this formula to calculate neutrons. The mass number is always the larger number provided on the periodic table.
Practical Applications
Understanding isotopes is crucial in medicine and archaeology. For instance, the isotope Carbon-14 is radioactive and decays at a known rate, allowing scientists to determine the age of ancient organic materials through carbon dating. In medicine, specific isotopes are used as tracers in diagnostic imaging to detect diseases like cancer.
Visual Resources
3 diagrams and illustrations
Interactive Diagrams
2 interactive diagrams to visualise key concepts
Subatomic particles and their properties within the atom.
The historical evolution of the atomic model based on new scientific evidence.
Worked Examples
3 detailed examples with solutions and examiner commentary
Practice Questions
Test your understanding — click to reveal model answers
Chlorine has two common isotopes: Chlorine-35 and Chlorine-37. Explain why these two isotopes have the same chemical properties. (2 marks)
Hint: Think about which subatomic particle is involved in chemical reactions.
Compare the chemical and physical properties of transition elements with Group 1 elements. (6 marks)
Hint: Structure your answer into physical properties (melting point, density, hardness) and chemical properties (reactivity, compounds formed).
Describe how the alpha particle scattering experiment led to the nuclear model of the atom. (4 marks)
Hint: What did Rutherford fire at the gold foil, and what were the three main observations and their conclusions?
An element has the electronic structure 2,8,3. Identify its group and period in the periodic table. (2 marks)
Hint: Look at the number of outer electrons for the group, and the total number of shells for the period.
Explain why noble gases (Group 0) are unreactive. (2 marks)
Hint: Look at their outer electron shell.