Overview
Organic chemistry is the study of carbon-based compounds, which form the foundation of all living things and the fossil fuels we rely on daily. Carbon is unique because it can form four strong covalent bonds, allowing it to create long chains and rings—the backbone of millions of different molecules.
In your GCSE Chemistry exam, this topic is heavily tested. Examiners will expect you to recognise patterns within families of compounds called homologous series. You must be able to draw and name the first four members of the alkanes, alkenes, alcohols, and carboxylic acids. Furthermore, this topic links closely with your understanding of covalent bonding, fractional distillation, and the environmental impact of combustion. Questions frequently assess your ability to balance equations, compare reaction conditions (such as fermentation versus hydration), and explain the mechanisms of cracking and polymerisation.

Key Concepts
Concept 1: Homologous Series and Alkanes
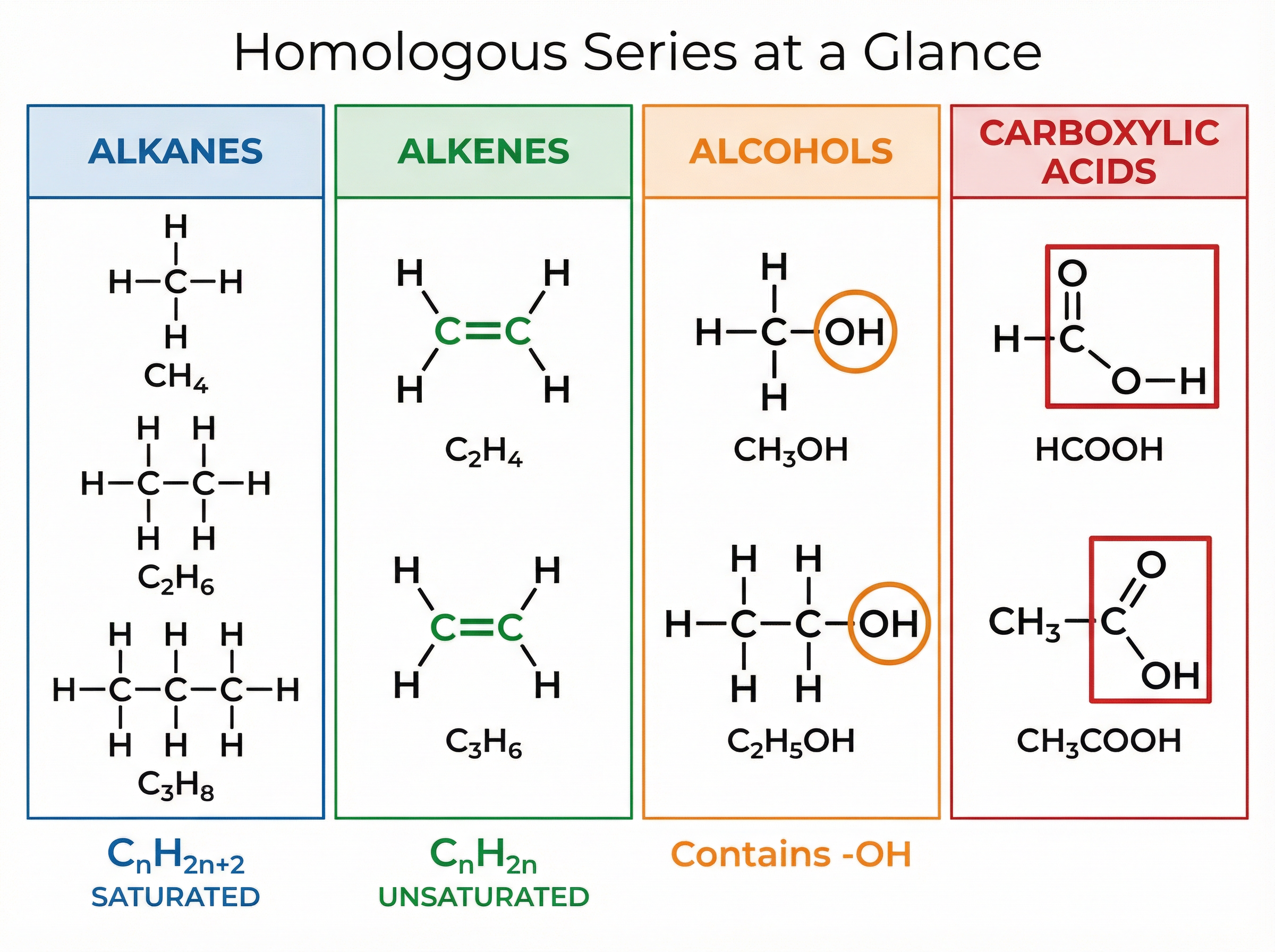
A homologous series is a family of organic compounds that share the same general formula, possess similar chemical properties, and show a gradual trend in physical properties (such as boiling point) as the carbon chain gets longer.
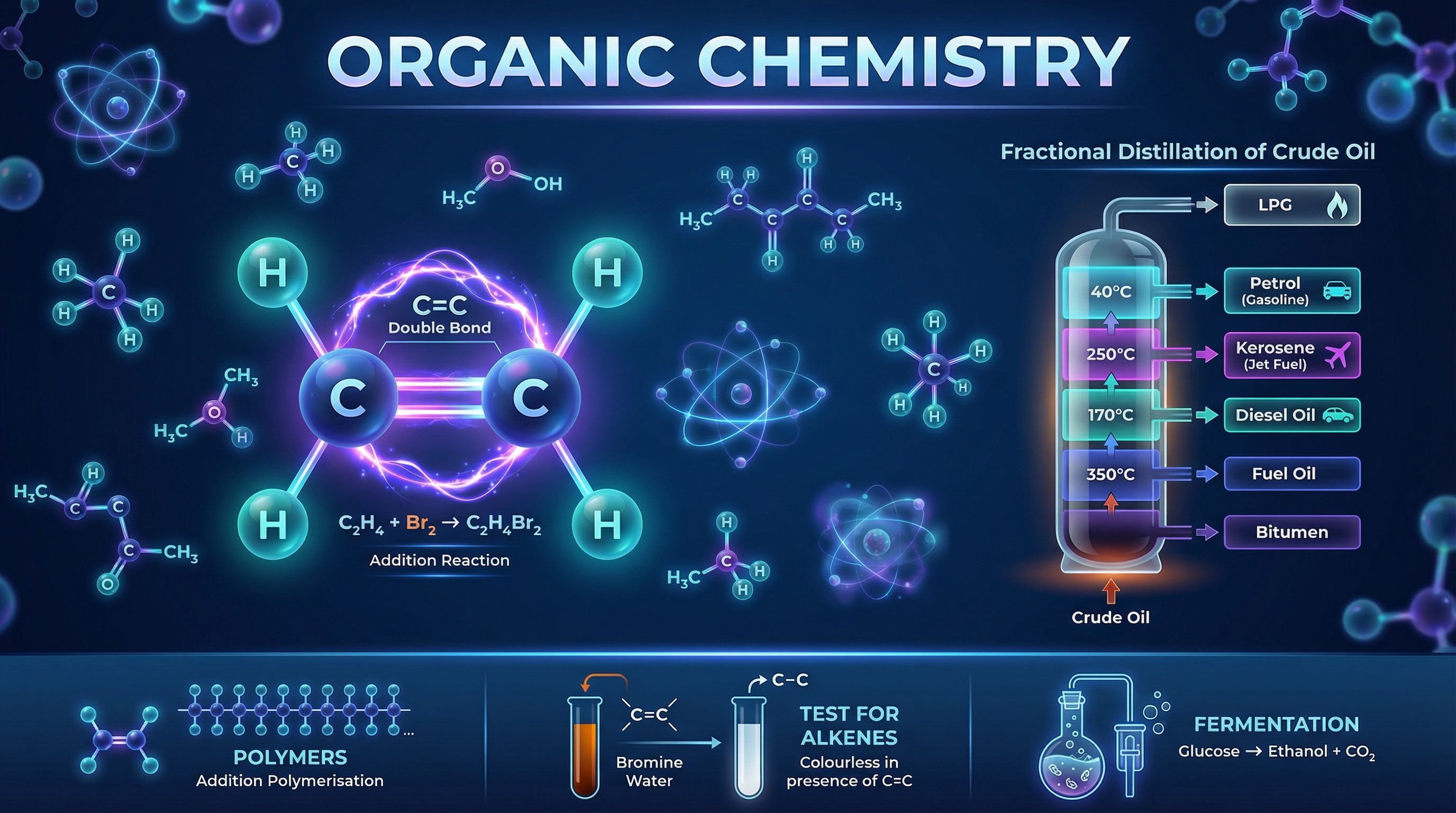
The simplest homologous series is the alkanes. Alkanes are described as saturated hydrocarbons. "Hydrocarbon" means they contain only hydrogen and carbon atoms. "Saturated" means they contain only single carbon-carbon bonds—they have the maximum possible number of hydrogen atoms attached. Their general formula is C_nH_{2n+2}.
As the carbon chain increases in length, the intermolecular forces between the molecules become stronger. This explains why boiling points and viscosity (thickness) increase with chain length, while flammability decreases.
Example: If an alkane has 5 carbon atoms (n=5), the number of hydrogen atoms is (2 \times 5) + 2 = 12. The formula is C_5H_{12} (pentane).
Concept 2: Fractional Distillation and Cracking
Crude oil is a complex mixture of hydrocarbons, mostly alkanes. Because different chain lengths have different boiling points, we can separate them using fractional distillation. The crude oil is heated until it vaporises and enters a fractionating column, which is hot at the bottom and cooler at the top. Long-chain hydrocarbons condense at the bottom, while short-chain hydrocarbons condense at the top.
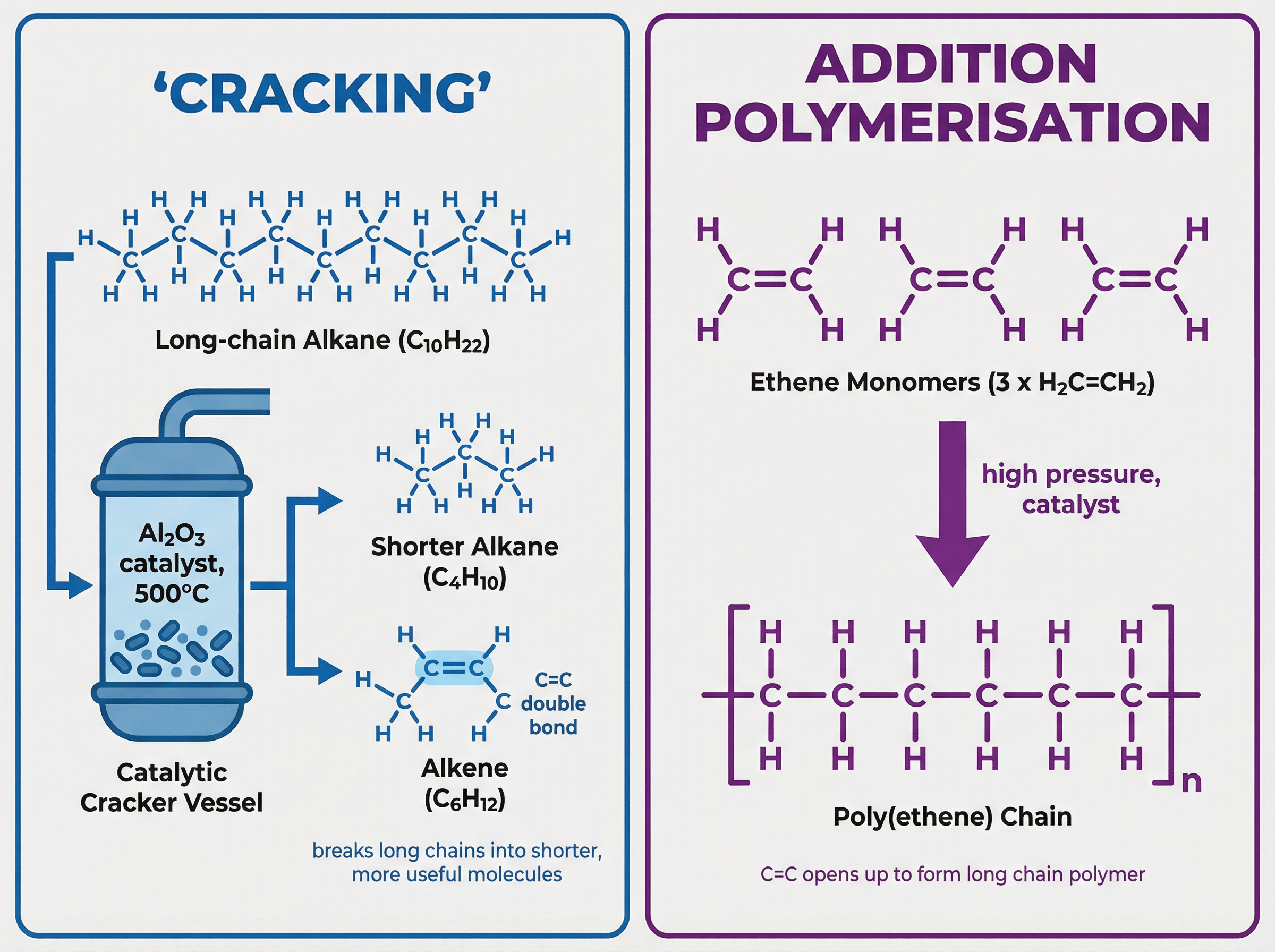
However, there is a higher demand for short-chain hydrocarbons (like petrol) than for long-chain ones (like bitumen). To solve this, industrial chemists use a process called cracking. Cracking is a thermal decomposition reaction that breaks down large, unuseful alkane molecules into smaller, more useful alkanes and alkenes.
This requires high temperatures (around 500°C) and a catalyst (such as aluminium oxide), or mixing with steam at very high temperatures.
Concept 3: Alkenes and Addition Reactions
Alkenes are another homologous series of hydrocarbons, but they are unsaturated. This means they contain at least one carbon-carbon double bond (C=C). Their general formula is C_nH_{2n}.
The double bond makes alkenes much more reactive than alkanes. They undergo addition reactions, where the double bond opens up to allow new atoms to bond to the carbon atoms.
The most common test for an alkene is the bromine water test. When orange bromine water is added to an alkene, it undergoes an addition reaction and turns colourless. If added to an alkane, it remains orange because alkanes lack the reactive double bond.
Concept 4: Alcohols and Carboxylic Acids
Alcohols contain the functional group -OH (hydroxyl group). The most common is ethanol (C_2H_5OH), which is used in alcoholic drinks and as a solvent. Ethanol can be produced biologically via the fermentation of sugar using yeast, or industrially by the hydration of ethene with steam (an addition reaction).
Carboxylic acids contain the functional group -COOH (carboxyl group). They are weak acids, meaning they only partially ionise in water. Ethanoic acid (CH_3COOH) is the main component of vinegar. They react with alcohols to form esters, which are sweet-smelling compounds used in flavourings and perfumes.
Concept 5: Polymerisation
Polymers are very large molecules made when hundreds or thousands of small molecules, called monomers, join together.
In addition polymerisation, the monomers are alkenes. The carbon-carbon double bond opens up to form a continuous chain. For example, ethene monomers join to form poly(ethene). The polymer contains only single bonds.
In condensation polymerisation, the monomers have two functional groups. When they react, they join together and lose a small molecule, typically water. This is how polyesters and polyamides (like nylon) are formed.
Mathematical/Scientific Relationships
- Alkane General Formula: C_nH_{2n+2}
- Alkene General Formula: C_nH_{2n}
- Complete Combustion: Hydrocarbon + Oxygen \rightarrow Carbon Dioxide + Water
- Incomplete Combustion: Hydrocarbon + Oxygen \rightarrow Carbon Monoxide + Water (occurs when oxygen is limited)
- Fermentation Equation: Glucose \rightarrow Ethanol + Carbon Dioxide (C_6H_{12}O_6 \rightarrow 2C_2H_5OH + 2CO_2)
Practical Applications
Understanding organic chemistry is crucial for the petrochemical industry. The separation of crude oil provides the fuels that run our cars, planes, and ships. Cracking ensures we have enough petrol to meet global demand. Furthermore, the synthesis of polymers from alkenes has revolutionised modern manufacturing, giving us plastics for packaging, medical devices, and clothing. However, this also brings environmental challenges, as many addition polymers are non-biodegradable, requiring us to innovate in recycling and the development of bioplastics.


