Overview
Quantitative Chemistry (Topic 4.3) is where chemistry meets mathematics. It's the study of exactly how much of a substance is involved in a chemical reaction. In industry, chemists don't just mix things together and hope for the best; they need to calculate precisely how much raw material to buy and how much product they can expect to sell. This topic gives you the tools to make those calculations.
This topic is fundamentally important because it underpins almost everything else in chemistry. Whether you're calculating the energy change in an exothermic reaction, determining the rate of a reaction, or figuring out the concentration of an acid in a titration, you'll be using the skills you learn here.
Examiners love quantitative chemistry because it allows them to test multiple skills at once: your understanding of chemical concepts, your ability to balance equations, and your mathematical prowess. You can expect a mix of short, one-mark recall questions (like "What is the value of the Avogadro constant?") and extended, multi-step calculation questions worth up to 6 marks.
Key Concepts
Concept 1: Relative Formula Mass (Mr)
The relative formula mass (M_r) is the sum of the relative atomic masses (A_r) of all the atoms shown in a chemical formula. You don't need to memorize A_r values; they are always provided on your periodic table.
To calculate M_r, you simply identify how many of each type of atom are in the formula, multiply that number by the atom's A_r, and add them all together.
Why does this matter? Because atoms are too small to weigh individually, we need a way to compare the masses of different molecules. The M_r gives us a proportional scale.
Example: Calculate the M_r of sulfuric acid, H_2SO_4.
- Hydrogen (A_r = 1): 2 atoms \times 1 = 2
- Sulfur (A_r = 32): 1 atom \times 32 = 32
- Oxygen (A_r = 16): 4 atoms \times 16 = 64
- Total M_r = 2 + 32 + 64 = 98
Concept 2: The Mole and Avogadro's Constant
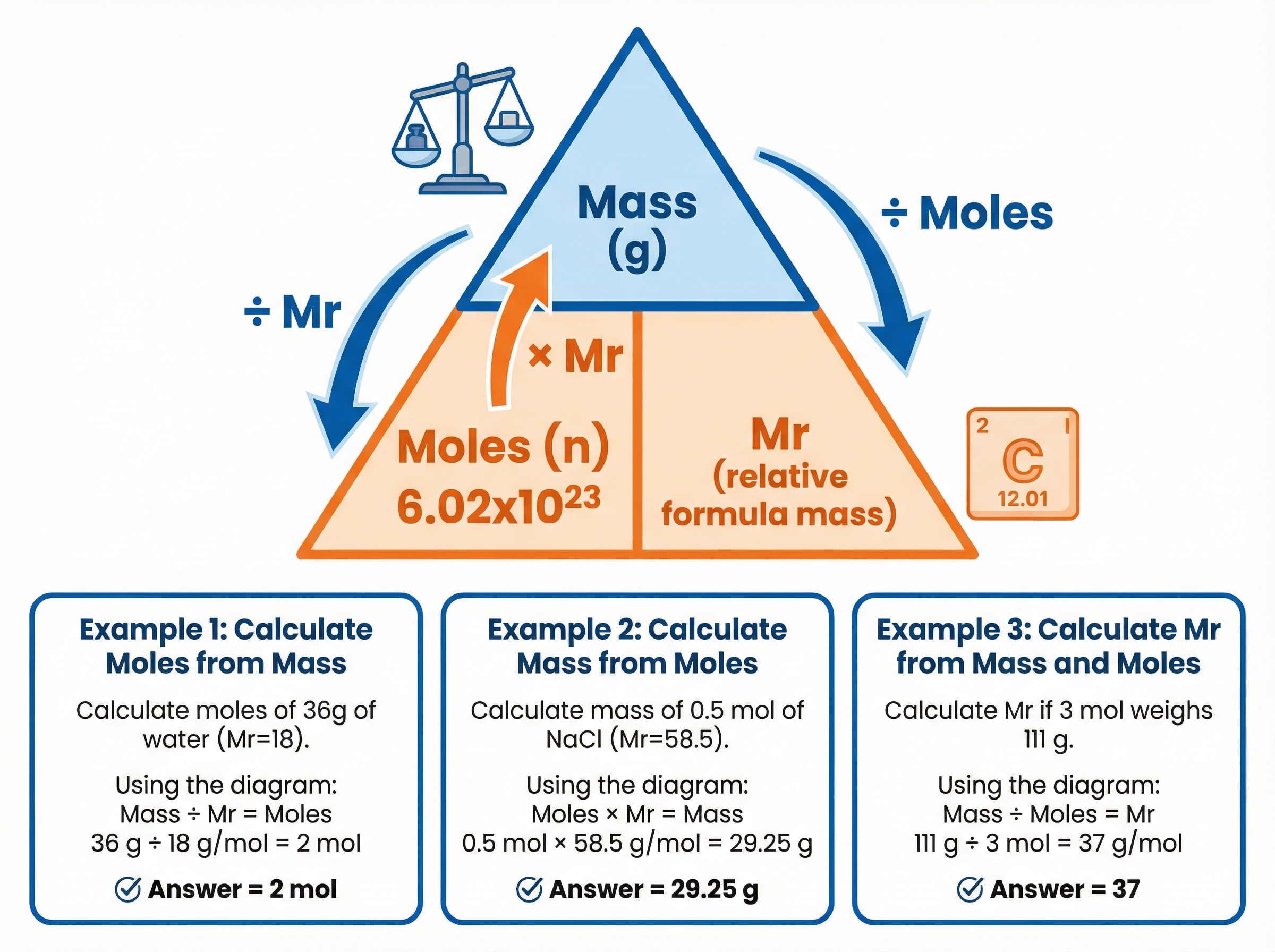
Chemical reactions happen between individual atoms and molecules, but we can't count them out one by one. Instead, we use a counting unit called the mole. Just as a "dozen" means 12, a "mole" means 6.02 \times 10^{23} particles. This massive number is called the Avogadro constant.
Here is the magic of the mole: One mole of any substance has a mass in grams exactly equal to its relative formula mass (M_r). So, 1 mole of carbon (A_r = 12) weighs exactly 12g. 1 mole of water (M_r = 18) weighs exactly 18g.
This gives us our most important equation:
Moles = \frac{Mass}{M_r}
Example: How many moles are in 66g of carbon dioxide (CO_2)?
- M_r of CO_2 = 12 + (16 \times 2) = 44
- Moles = \frac{66}{44} = 1.5 moles
Concept 3: Conservation of Mass and Balanced Equations
The Law of Conservation of Mass states that no atoms are lost or made during a chemical reaction. Therefore, the total mass of the products must equal the total mass of the reactants.
This is why we must balance chemical equations. The large numbers placed in front of formulas (coefficients) tell us the molar ratio of the reaction.
For example, in the equation N_2 + 3H_2 \rightarrow 2NH_3, the ratio is 1 mole of nitrogen reacting with 3 moles of hydrogen to produce 2 moles of ammonia.
Example: If 28g of nitrogen reacts completely with 6g of hydrogen, what mass of ammonia is produced?
- Total mass of reactants = 28g + 6g = 34g
- Therefore, total mass of products = 34g
Concept 4: Reacting Mass Calculations
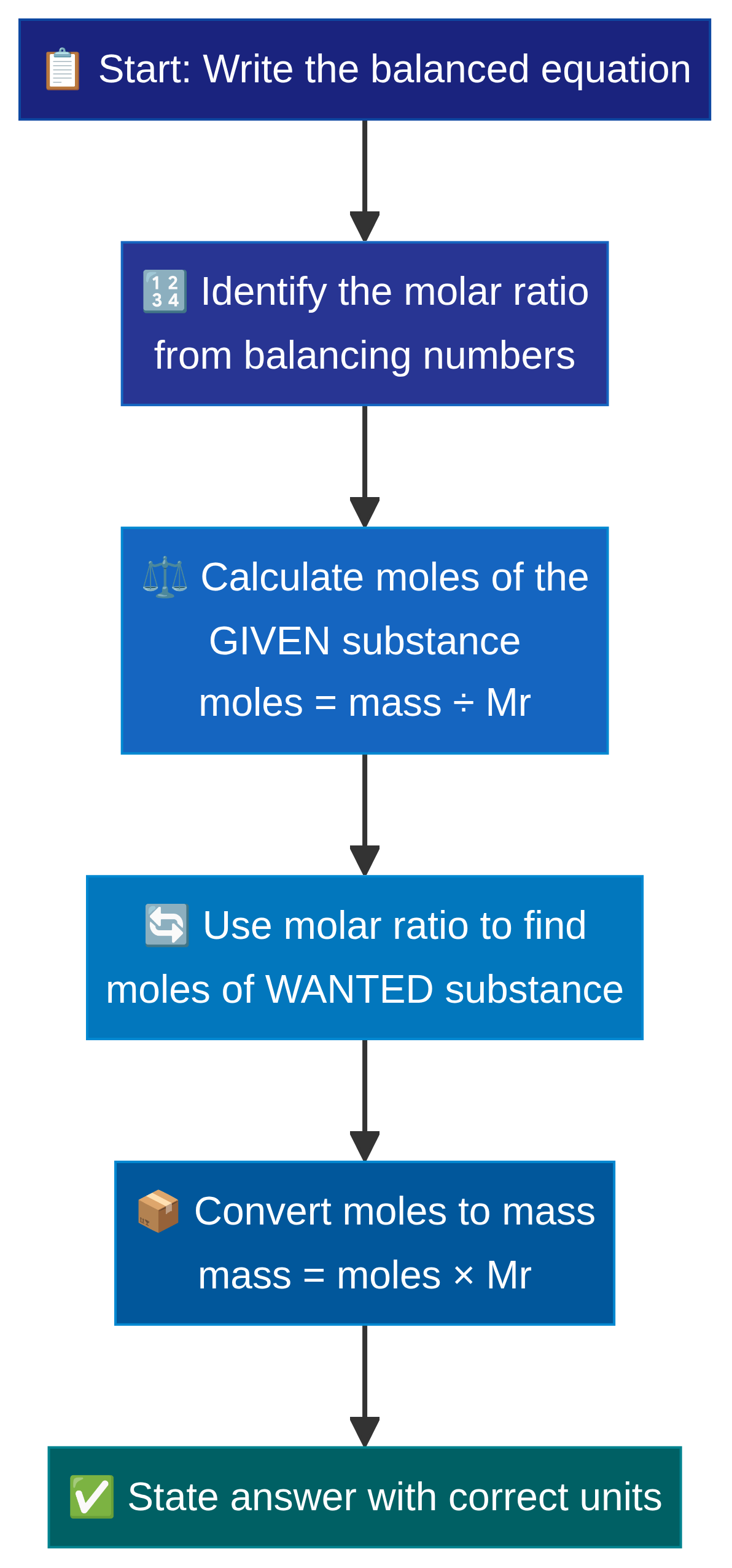
This is where we combine the mole concept with balanced equations to predict how much product we can make from a given mass of reactant.
Examiners award method marks for these multi-step calculations, so always lay out your working clearly. The standard method involves three steps:
- Calculate the moles of the substance you have the mass for.
- Use the balanced equation ratio to find the moles of the substance you want to know about.
- Calculate the mass of that substance using its M_r.
Example: What mass of calcium oxide (CaO) is produced when 50g of calcium carbonate (CaCO_3) is thermally decomposed?
Equation: CaCO_3 \rightarrow CaO + CO_2
- Step 1 (Moles of known): M_r of CaCO_3 = 100. Moles = \frac{50}{100} = 0.5 mol
- Step 2 (Ratio): The ratio of CaCO_3 to CaO is 1:1. So, moles of CaO = 0.5 mol
- Step 3 (Mass of unknown): M_r of CaO = 56. Mass = 0.5 \times 56 = 28g
Concept 5: Limiting Reactants (Higher Tier)
In most chemical reactions, one reactant is entirely used up, while the other is in excess. The reactant that is completely consumed is called the limiting reactant because it limits the amount of product that can be formed.
To find the limiting reactant, you cannot just look at which reactant has the smallest mass. You must calculate the moles of each reactant and compare them to the molar ratio required by the balanced equation.
Example: 4.8g of magnesium (Mg) reacts with 7.3g of hydrogen chloride (HCl). Which is the limiting reactant?
Equation: Mg + 2HCl \rightarrow MgCl_2 + H_2
- Moles of Mg = \frac{4.8}{24} = 0.2 mol
- Moles of HCl = \frac{7.3}{36.5} = 0.2 mol
- The equation requires 2 moles of HCl for every 1 mole of Mg (a 1:2 ratio).
- To react with 0.2 mol of Mg, we would need 0.4 mol of HCl. We only have 0.2 mol of HCl.
- Therefore, HCl is the limiting reactant.
Concept 6: Concentration of Solutions
Concentration measures how much solute is dissolved in a given volume of solvent. It can be measured in two units: grams per decimetre cubed (g/dm^3) or moles per decimetre cubed (mol/dm^3).
The formulas are:
- Concentration (g/dm^3) = \frac{Mass (g)}{Volume (dm^3)}
- Concentration (mol/dm^3) = \frac{Moles}{Volume (dm^3)}
CRITICAL EXAM TIP: Volumes are often given in cm^3 in exams. You must convert cm^3 to dm^3 by dividing by 1000 before calculating concentration.
Concept 7: Percentage Yield and Atom Economy

In real life, you never get 100% of the product you calculate theoretically. Percentage yield compares the actual mass of product obtained with the maximum theoretical mass.
Percentage Yield = \frac{Actual Yield}{Theoretical Yield} \times 100
Yields are less than 100% because:
- The reaction may be reversible and not go to completion.
- Some product may be lost when separated from the reaction mixture.
- Some reactants may react in different, unexpected ways (side reactions).
Atom economy is a measure of the amount of starting materials that end up as useful products. It's crucial for sustainable development and economic reasons in industry.
Atom Economy = \frac{M_r \text{ of desired product}}{\text{Total } M_r \text{ of all reactants}} \times 100
Reactions with high atom economy are preferred because they produce less waste and use raw materials more efficiently.
Concept 8: Volumes of Gases (Higher Tier)
At room temperature and pressure (RTP - usually 20^\circ C and 1 atmosphere), one mole of any gas occupies exactly 24 dm^3 (24,000 cm^3). This is called the molar gas volume.
Volume of gas (dm^3) = Moles \times 24
This means that equal volumes of different gases at the same temperature and pressure contain the same number of molecules.
Podcast Revision
Listen to this 12-minute revision podcast covering all the key concepts, common pitfalls, and examiner tips for Quantitative Chemistry.

Mathematical/Scientific Relationships
Here are the essential formulas you need for this topic.
Must Memorise:
- Moles = \frac{Mass}{M_r}
- Concentration (g/dm^3) = \frac{Mass (g)}{Volume (dm^3)}
- Concentration (mol/dm^3) = \frac{Moles}{Volume (dm^3)}
- Percentage Yield = \frac{Actual Yield}{Theoretical Yield} \times 100
- Atom Economy = \frac{M_r \text{ of desired product}}{\text{Total } M_r \text{ of all reactants}} \times 100
- Volume of gas (dm^3) = Moles \times 24 (Higher Tier)
Key Unit Conversions:
- cm^3 \rightarrow dm^3: Divide by 1000
- dm^3 \rightarrow cm^3: Multiply by 1000
- mol/dm^3 \rightarrow g/dm^3: Multiply by the M_r of the solute
- g/dm^3 \rightarrow mol/dm^3: Divide by the M_r of the solute
Practical Applications
While there isn't a specific required practical solely dedicated to reacting masses, these skills are heavily assessed in the Titration required practical.
In a titration, you use a solution of known concentration to find the concentration of an unknown solution. The calculation involves finding the moles of the known substance using n = c \times V, using the balanced equation to find the molar ratio, and then calculating the concentration of the unknown using c = \frac{n}{V}. Examiners frequently combine practical technique questions with a 4-5 mark calculation.



