Study Notes
Overview
Atomic structure and the periodic table form the absolute bedrock of GCSE and A-Level Chemistry. This topic explores the fundamental building blocks of all matter—atoms—and how their internal structure dictates chemical behaviour. Understanding the arrangement of protons, neutrons, and electrons is crucial, as it directly explains why elements react the way they do and why the periodic table is structured as it is.
Examiners frequently test this topic through a mix of short-answer recall questions, calculation-based questions on relative atomic mass, and extended 4-6 mark questions requiring you to describe historical models or explain trends. Furthermore, mastering atomic structure provides the essential synoptic links needed for subsequent topics, including bonding, quantitative chemistry, and rates of reaction.
Listen to our comprehensive revision podcast to reinforce these concepts:
Key Concepts
Concept 1: Subatomic Particles and Atomic Structure
Atoms consist of a central, incredibly dense nucleus containing protons and neutrons, surrounded by electrons orbiting in specific energy levels (shells). The defining characteristic of any element is its atomic number (proton number), which dictates the number of protons in its nucleus. In a neutral atom, the number of negatively charged electrons perfectly balances the number of positively charged protons, resulting in no overall electrical charge.
Examiner Tip: Candidates frequently lose marks by confusing atomic number and mass number. Remember that the mass number is always the larger number, representing the total sum of protons and neutrons. The mass of an electron is considered negligible (approximately 1/2000th the mass of a proton), which is why electrons are not included in the mass number calculation.
Concept 2: Isotopes and Relative Atomic Mass (Higher Tier)
Isotopes are atoms of the same element that possess the same number of protons but a different number of neutrons. Because chemical properties are determined entirely by the electron configuration, isotopes of an element react in exactly the same way. However, their physical properties, such as density or radioactivity, may differ due to the varying mass.
Because elements exist as mixtures of isotopes in nature, chemists use Relative Atomic Mass (Ar). This is a weighted average that accounts for both the mass of each isotope and its relative abundance.
Example: Chlorine exists as 75% Chlorine-35 and 25% Chlorine-37.
Relative Atomic Mass = ((75 × 35) + (25 × 37)) / 100 = 35.5
Concept 3: The Historical Development of the Atomic Model
The atomic model has evolved significantly as new scientific evidence emerged. Examiners love to test your understanding of this progression, particularly the transition from the plum pudding model to the nuclear model.
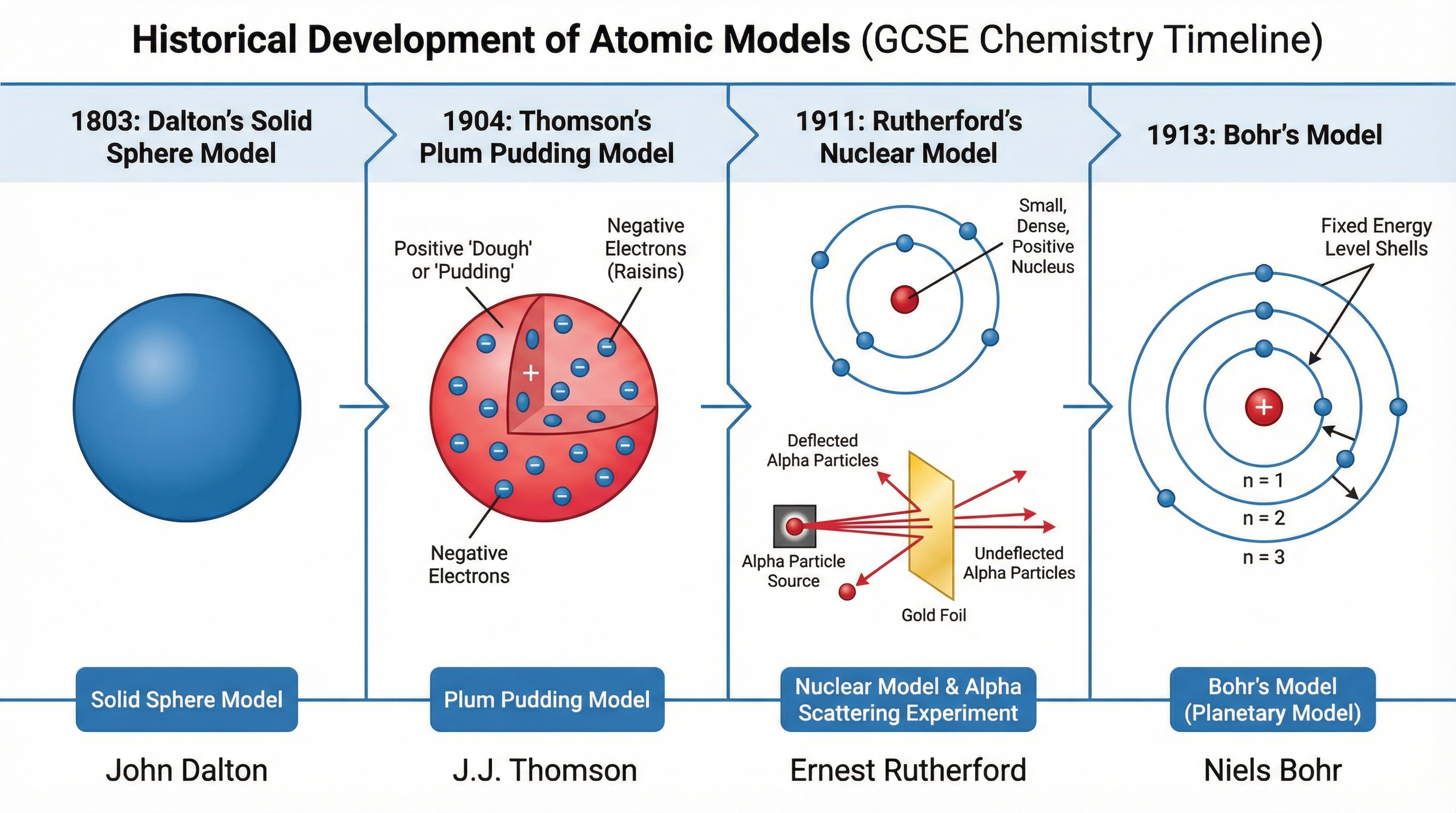
- Dalton's Solid Sphere (1803): Atoms were considered indivisible spheres.
- Thomson's Plum Pudding (1904): Following the discovery of the electron, the atom was modelled as a ball of positive charge with negative electrons embedded within it.
- Rutherford's Nuclear Model (1911): The alpha particle scattering experiment proved the plum pudding model wrong. Most alpha particles passed straight through gold foil (showing atoms are mostly empty space), while a few were deflected or bounced back (proving the existence of a small, dense, positively charged nucleus).
- Bohr's Model (1913): Bohr adapted the nuclear model by suggesting electrons orbit the nucleus at specific distances in fixed energy levels.
Concept 4: Electron Configuration and the Periodic Table
The arrangement of electrons in an atom determines its position in the periodic table and its chemical properties. Electrons fill energy levels starting from the lowest energy level (closest to the nucleus) outwards. The first shell holds a maximum of 2 electrons, while the second and third shells hold up to 8 electrons each.
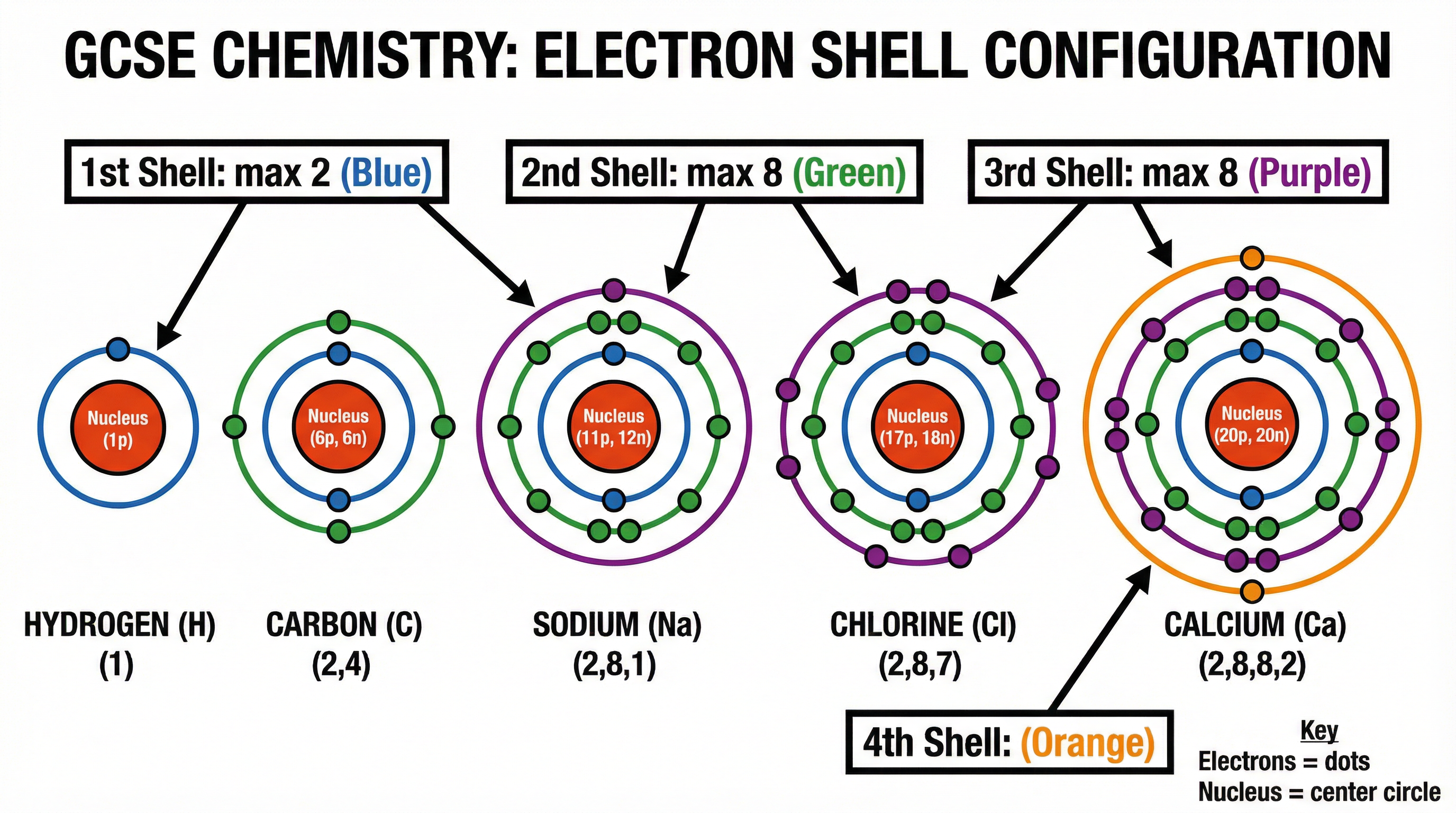
The periodic table is a masterpiece of scientific organisation. The group number (vertical columns) indicates the number of electrons in the outermost shell, which explains why elements in the same group have similar chemical properties. The period number (horizontal rows) indicates the total number of electron shells the atom possesses.
Mathematical/Scientific Relationships
- Number of Neutrons = Mass Number - Atomic Number
- Relative Atomic Mass (Ar) = Σ(Isotope Mass × Percentage Abundance) / 100
- Maximum Electrons per Shell: Shell 1 = 2, Shell 2 = 8, Shell 3 = 8 (at GCSE level)
Practical Applications
Understanding atomic structure is not just theoretical. Isotopes like Carbon-14 are used in radiocarbon dating to determine the age of archaeological artefacts. In medicine, radioactive isotopes are used as tracers for diagnostic imaging and in radiotherapy to target cancer cells. The predictable electron configurations of elements allow materials scientists to design new alloys and semiconductors for modern electronics.
Visual Resources
2 diagrams and illustrations
Interactive Diagrams
2 interactive diagrams to visualise key concepts
Concept map showing the structure of an atom and the role of each subatomic particle.
Logical flow of Rutherford's alpha particle scattering experiment observations and conclusions.
Worked Examples
3 detailed examples with solutions and examiner commentary
Practice Questions
Test your understanding — click to reveal model answers
An atom of sodium has an atomic number of 11 and a mass number of 23. State the number of protons, neutrons, and electrons in this neutral atom. [3 marks]
Hint: Remember the PEN rule and how to calculate neutrons from the mass number.
Explain how the results of the alpha particle scattering experiment led to the nuclear model of the atom. [4 marks]
Hint: Link specific observations (what happened to the alpha particles) to specific conclusions (what this means about the atom).
Copper exists as two isotopes: Copper-63 (abundance 69.2%) and Copper-65 (abundance 30.8%). Calculate the relative atomic mass of copper. [3 marks]
Hint: Use the formula: (mass1 × abundance1) + (mass2 × abundance2) / 100.
Explain why isotopes of the same element have identical chemical properties but can have different physical properties. [3 marks]
Hint: Think about which subatomic particles dictate chemical reactions versus which dictate mass.
An element has the electron configuration 2, 8, 3. Deduce its group and period on the periodic table, and explain your reasoning. [4 marks]
Hint: Look at the number of shells and the number of electrons in the outermost shell.