Overview

Welcome to the foundational language of Chemistry! This topic is about how we communicate chemical ideas clearly and safely. You will learn how to write chemical formulae, balance word and symbol equations, and interpret the hazard symbols found on chemical containers.
Why is this important? Examiners consider these to be core skills. You cannot access the higher marks in quantitative chemistry (like moles and reacting masses) if you cannot first write a balanced symbol equation. Furthermore, questions on hazard symbols and risk assessments frequently appear in the practical-based questions of your exam papers.
This topic connects to almost everything else in the specification. Whether you are studying rates of reaction, electrolysis, or organic chemistry, you will be expected to write balanced equations with correct state symbols.
Listen to the summary podcast here:

Key Concepts
Concept 1: Chemical Formulae
A chemical formula shows the elements present in a compound and the relative number of atoms of each element. For example, the formula for water is H₂O. This tells us that one molecule of water contains two hydrogen atoms and one oxygen atom. The small subscript number applies only to the element immediately before it. If there is no number, it means there is one atom of that element.
Examiner Tip: Never change the small subscript numbers in a chemical formula when balancing an equation. Changing H₂O to H₂O₂ changes water into hydrogen peroxide—a completely different chemical!
Concept 2: Balancing Equations
The Law of Conservation of Mass states that no atoms are lost or made during a chemical reaction. Therefore, the mass of the products equals the mass of the reactants. This means that chemical equations must be balanced—there must be the same number of atoms of each element on both sides of the equation.
To balance an equation, you add large numbers (coefficients) in front of the chemical formulae.
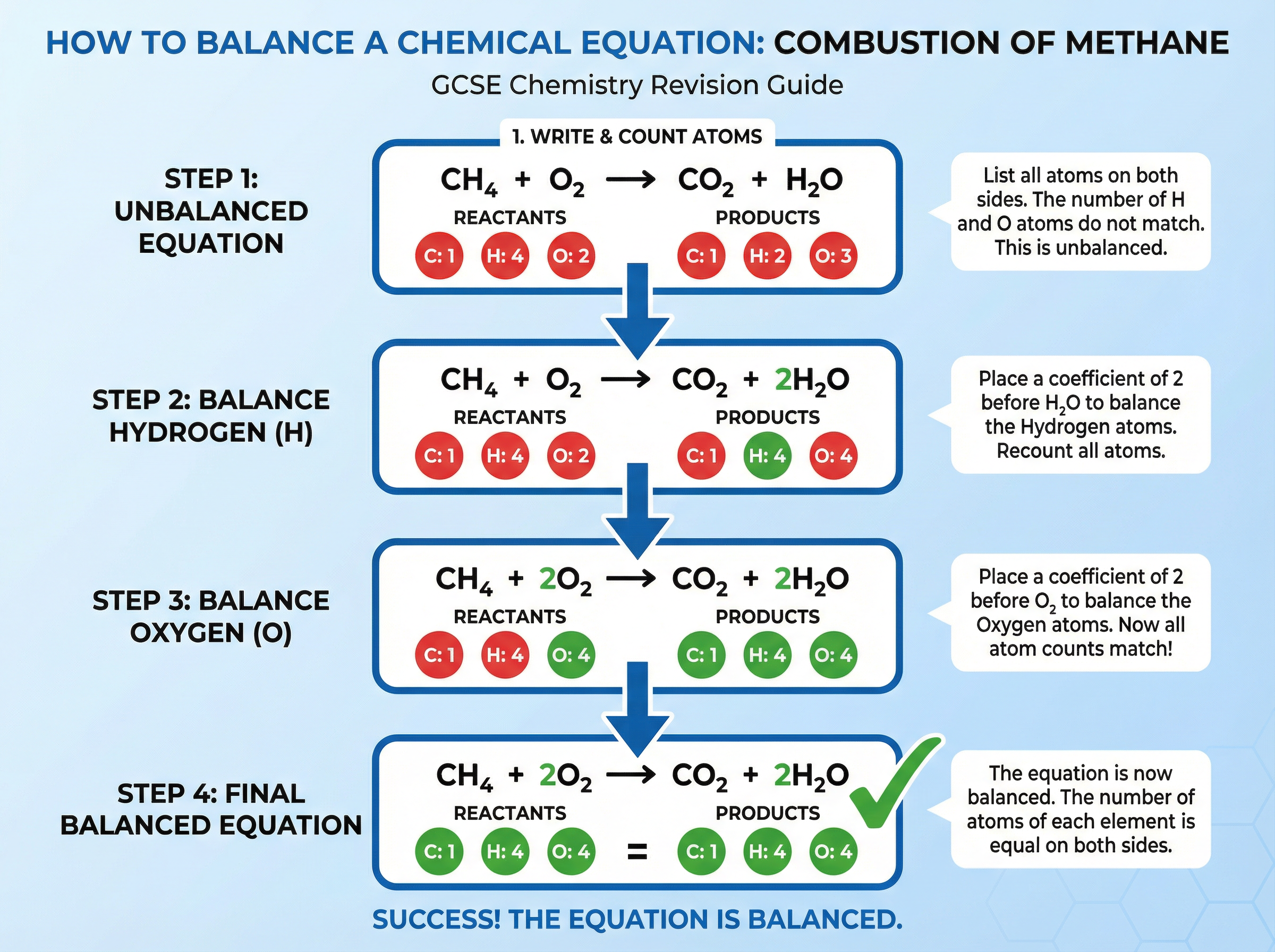
Example: Balancing the combustion of methane.
Unbalanced: CH₄ + O₂ → CO₂ + H₂O
Left side: 1 C, 4 H, 2 O
Right side: 1 C, 2 H, 3 O
First, balance the hydrogen by putting a 2 in front of the water:
CH₄ + O₂ → CO₂ + 2H₂O
Now we have 4 H on both sides. But we have 2 O from CO₂ and 2 O from 2H₂O, making 4 O on the right.
Next, balance the oxygen by putting a 2 in front of the O₂:
CH₄ + 2O₂ → CO₂ + 2H₂O
Left side: 1 C, 4 H, 4 O
Right side: 1 C, 4 H, 4 O. The equation is balanced.
Concept 3: State Symbols
State symbols are letters in brackets placed after a chemical formula to indicate its physical state at room temperature or under the conditions of the reaction.
- (s) = solid
- (l) = liquid
- (g) = gas
- (aq) = aqueous (dissolved in water)
Examiners frequently award a specific mark for including correct state symbols.
Concept 4: Ionic Equations
Ionic equations show only the species that are reacting. The ions that do not change during the reaction are called 'spectator ions' and are left out of the final equation.
For example, in a neutralisation reaction between hydrochloric acid and sodium hydroxide:
Full equation: HCl(aq) + NaOH(aq) → NaCl(aq) + H₂O(l)
Ionic equation: H⁺(aq) + OH⁻(aq) → H₂O(l)
The Na⁺ and Cl⁻ ions are spectator ions because they remain dissolved in the solution before and after the reaction.
Concept 5: Hazard Symbols and Risk Assessment
Hazard symbols are universally recognised pictograms used to warn about the dangers of specific chemicals.

When evaluating risks in practical procedures, you must identify the hazard, state the risk, and suggest a suitable precaution. For example, if using concentrated sulfuric acid, the hazard is that it is corrosive. The risk is that it could cause severe skin burns or eye damage. The precaution is to wear chemical-resistant gloves and safety goggles.
Practical Applications
These concepts are applied every time you step into a laboratory. Before any required practical, a risk assessment must be carried out using hazard symbols. Balancing equations is used in industrial chemistry to calculate exactly how much of each reactant is needed to produce a specific amount of product, minimising waste and maximising profit.


