Study Notes
Overview
Foundations in Chemistry is exactly what the title suggests—the bedrock upon which all advanced chemical knowledge is built. Whether you are calculating the mass of a product in an industrial process or predicting the pH of a solution, the concepts in this module are your essential toolkit. This topic bridges the gap between GCSE and A-Level, taking familiar ideas like atomic structure and moles and adding rigorous mathematical precision.
In the exam, this module is often tested through multi-step calculation questions, definitions of key terms, and the application of redox principles to unfamiliar reactions. Examiners look for precision: correct significant figures, clear working out, and the accurate use of terminology like "relative isotopic mass" versus "relative atomic mass." Understanding these foundations is not just about passing Module 2; it is about equipping yourself with the language and logic needed for organic, physical, and inorganic chemistry.
Key Concepts
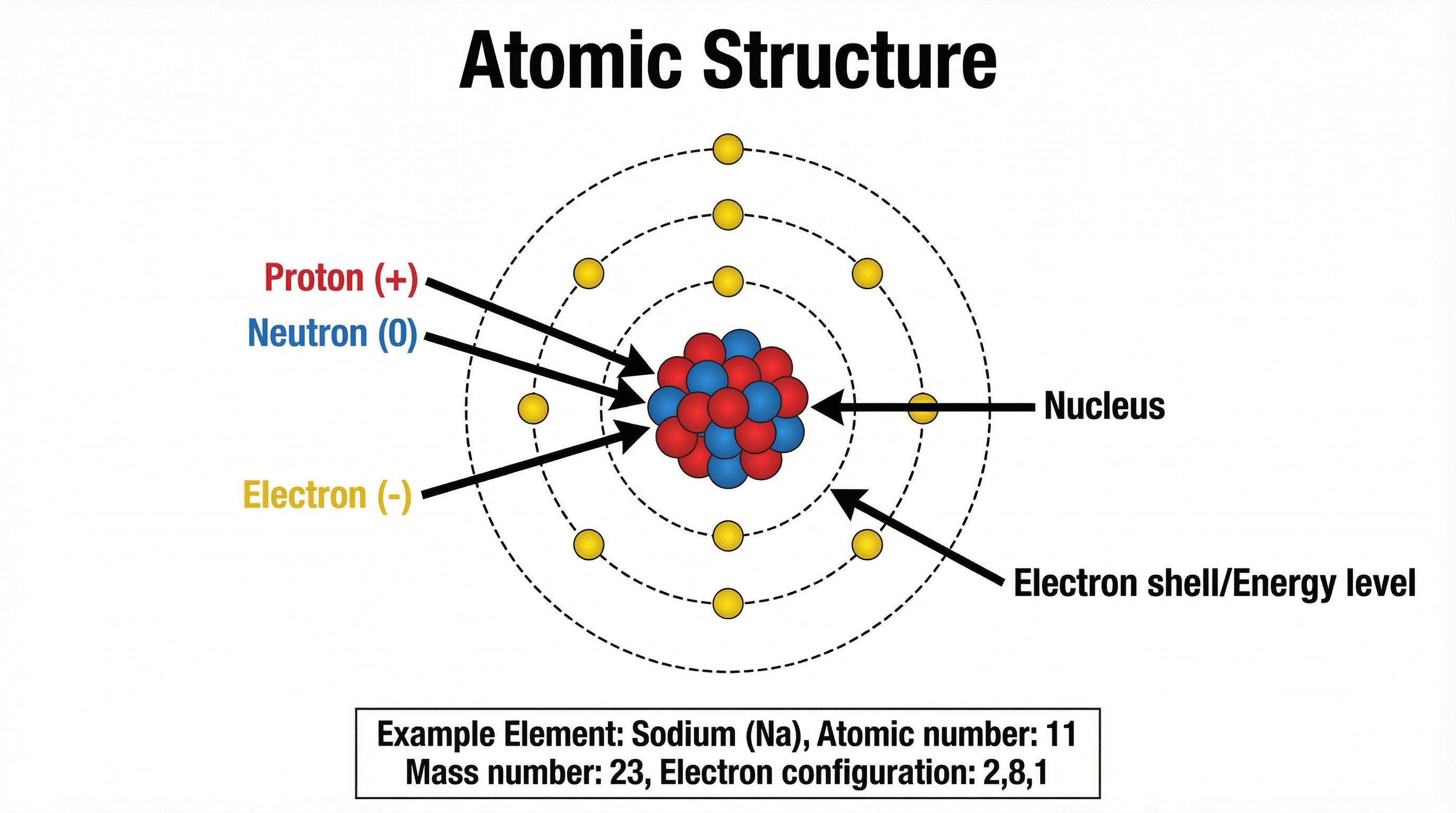
Concept 1: Atomic Structure and Isotopes
At the centre of every atom lies the nucleus, containing protons and neutrons, while electrons orbit in energy levels (shells). The atomic number (Z) defines the element by its proton count, while the mass number (A) is the sum of protons and neutrons.
Isotopes are atoms of the same element with the same number of protons but different numbers of neutrons. Crucially, isotopes have identical chemical properties because they have the same electron configuration, which determines how they react. However, they have different physical properties (like mass and density) due to the mass difference.
Concept 2: Relative Masses
Chemists deal with atoms so small that weighing them individually is impossible. Instead, we use the carbon-12 scale. The standard is that one atom of carbon-12 has a mass of exactly 12 atomic mass units.
- Relative Atomic Mass (A_r): The weighted mean mass of an atom of an element compared to 1/12th of the mass of an atom of carbon-12.
- Relative Isotopic Mass: The mass of an isotope of an element compared to 1/12th of the mass of an atom of carbon-12.
The "weighted mean" accounts for the percentage abundance of each isotope naturally found in a sample.
Concept 3: The Mole and Avogadro Constant
The mole (symbol: mol) is the chemist's dozen. It is the unit for amount of substance. One mole contains exactly 6.02 \times 10^{23} particles (atoms, molecules, ions, or electrons). This number is the Avogadro constant (N_A).
Why this specific number? Because 6.02 \times 10^{23} atoms of carbon-12 weigh exactly 12 grams. This allows us to scale up from the atomic world to the laboratory world.
Concept 4: Acids, Bases, and Neutralisation
- Acid: A species that releases H^+ ions in aqueous solution (a proton donor).
- Base: A species that accepts H^+ ions (a proton acceptor).
- Alkali: A soluble base that releases OH^- ions in water.
Strong acids (like HCl, HNO_3, H_2SO_4) fully dissociate in water. Weak acids (like CH_3COOH) only partially dissociate. This difference is vital for pH calculations and understanding equilibrium.
Concept 5: Redox Reactions
Redox stands for Reduction-Oxidation. These two processes happen simultaneously.
- Oxidation is the loss of electrons (or increase in oxidation number).
- Reduction is the gain of electrons (or decrease in oxidation number).
Oxidation numbers are a way of tracking electrons. Elements in their standard state have an oxidation number of 0. In compounds, oxygen is usually -2, hydrogen is +1, and fluorine is -1. The sum of oxidation numbers in a neutral compound is 0.
Mathematical/Scientific Relationships
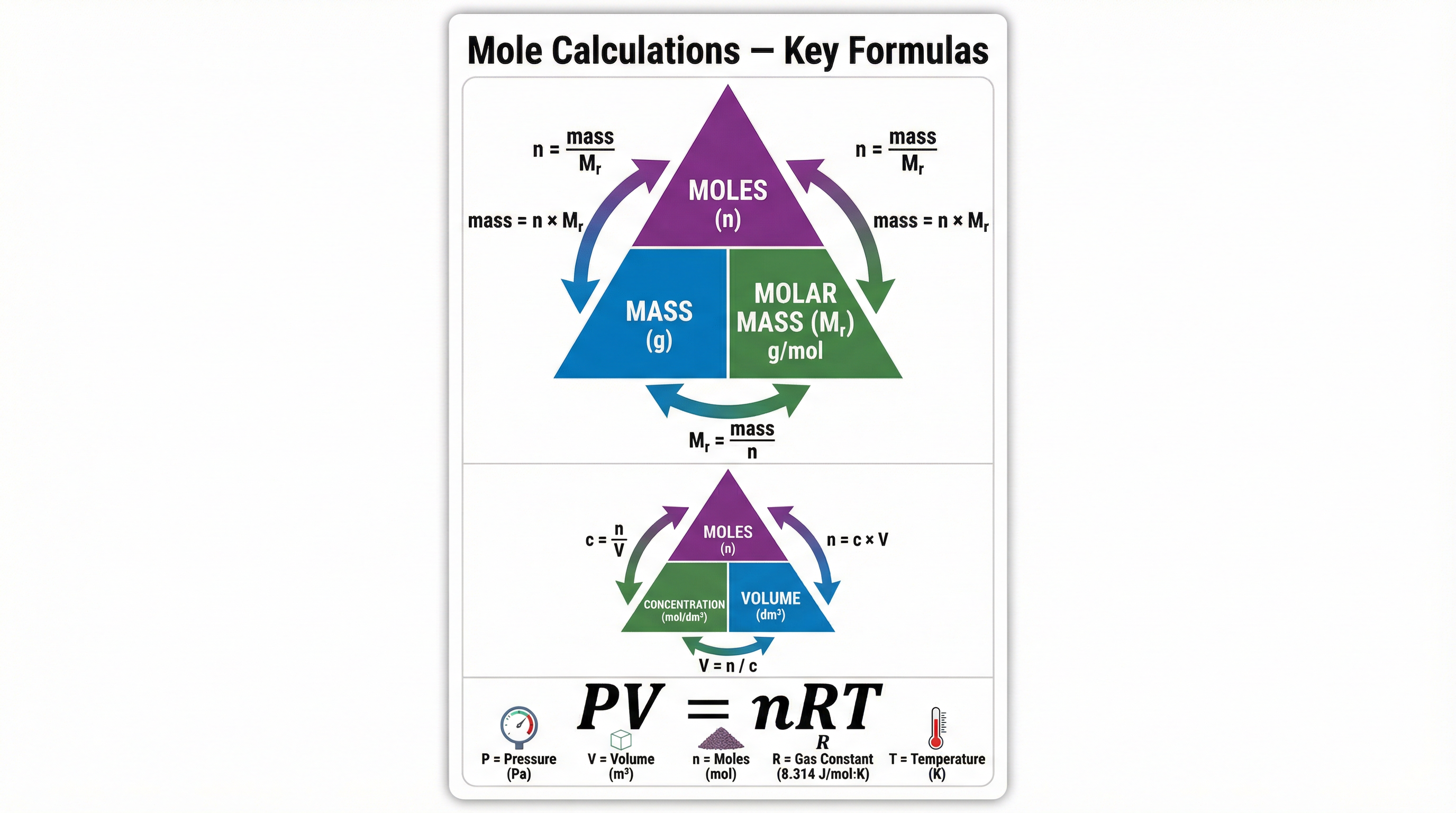
1. Moles, Mass, and Molar Mass
n = \frac{m}{M}
Where:
- n = amount of substance (mol)
- m = mass (g)
- M = molar mass (g mol^{-1})
2. Moles, Concentration, and Volume
n = c \times V
Where:
- n = amount of substance (mol)
- c = concentration (mol dm^{-3})
- V = volume (dm^3)
Note: If volume is in cm^3, divide by 1000 to convert to dm^3.
3. Ideal Gas Equation
pV = nRT
Where:
- p = pressure (Pa)
- V = volume (m^3) (Watch out! This is m^3, not dm^3)
- n = amount of substance (mol)
- R = ideal gas constant (8.314 J mol^{-1} K^{-1})
- T = temperature (K) (Add 273 to ^\circC)
4. Percentage Yield
\text{Percentage Yield} = \frac{\text{Actual Yield}}{\text{Theoretical Yield}} \times 100
5. Atom Economy
\text{Atom Economy} = \frac{\text{Molar mass of desired product}}{\text{Sum of molar masses of all products}} \times 100
Practical Applications
Titrations: A key practical skill involving the precise measurement of volumes to determine the concentration of an unknown solution. You must be able to rinse burettes correctly (with the solution they will contain), read the meniscus at eye level, and perform concordant titres (within 0.10 cm^3).
Standard Solutions: Preparing a solution of known concentration. This involves weighing a primary standard accurately, dissolving it in distilled water, transferring to a volumetric flask, and making up to the mark with the bottom of the meniscus sitting on the line.
Gas Collection: Measuring the volume of gas produced in a reaction to determine molar mass or reaction stoichiometry. Common errors include gas escaping before the bung is replaced or carbon dioxide dissolving in water."
Worked Examples
3 detailed examples with solutions and examiner commentary
Practice Questions
Test your understanding — click to reveal model answers
Define the term 'relative isotopic mass'. [2 marks]
Hint: Think about the comparison to carbon-12. Is it a weighted mean or a specific mass?
Sodium reacts with water: 2Na(s) + 2H_2O(l) \rightarrow 2NaOH(aq) + H_2(g). Calculate the mass of sodium required to produce 0.500 dm^3 of hydrogen gas at RTP (24.0 dm^3 mol^{-1}). [3 marks]
Hint: Find moles of hydrogen first using gas volume, then use the 2:1 ratio.
A sample of hydrated calcium chloride, CaCl_2 \cdot xH_2O, has a molar mass of 147.1 g mol^{-1}. Calculate the value of x. (A_r: Ca=40.1, Cl=35.5, H=1.0, O=16.0) [3 marks]
Hint: Calculate the mass of the anhydrous part ($CaCl_2$) first, then see what mass is left for the water.
Explain, in terms of electrons, what occurs during oxidation and reduction. [2 marks]
Hint: Remember OIL RIG.
Identify the oxidising agent in the reaction: Cl_2 + 2NaBr \rightarrow 2NaCl + Br_2. Explain your answer using oxidation numbers. [3 marks]
Hint: The oxidising agent gets reduced (gains electrons).
Question 6