
Overview
Welcome to Topic C4: Predicting and Identifying Reactions and Products. This topic is the detective work of Chemistry. It bridges the theoretical understanding of atomic structure with the practical reality of the laboratory. By understanding the underlying trends of the Periodic Table, you can predict how entirely new elements will behave. Furthermore, you will learn the specific chemical tests required to identify unknown gases, cations (positive ions), and anions (negative ions).
This topic is crucial because it frequently appears in extended response questions (6-markers) where examiners expect you to link macroscopic observations (like a colour change) to microscopic properties (like electron shielding). It also connects deeply with quantitative chemistry and chemical bonding. Typical exam questions will ask you to interpret experimental results, justify reactivity trends, or write balanced ionic equations for precipitation reactions.

Key Concepts
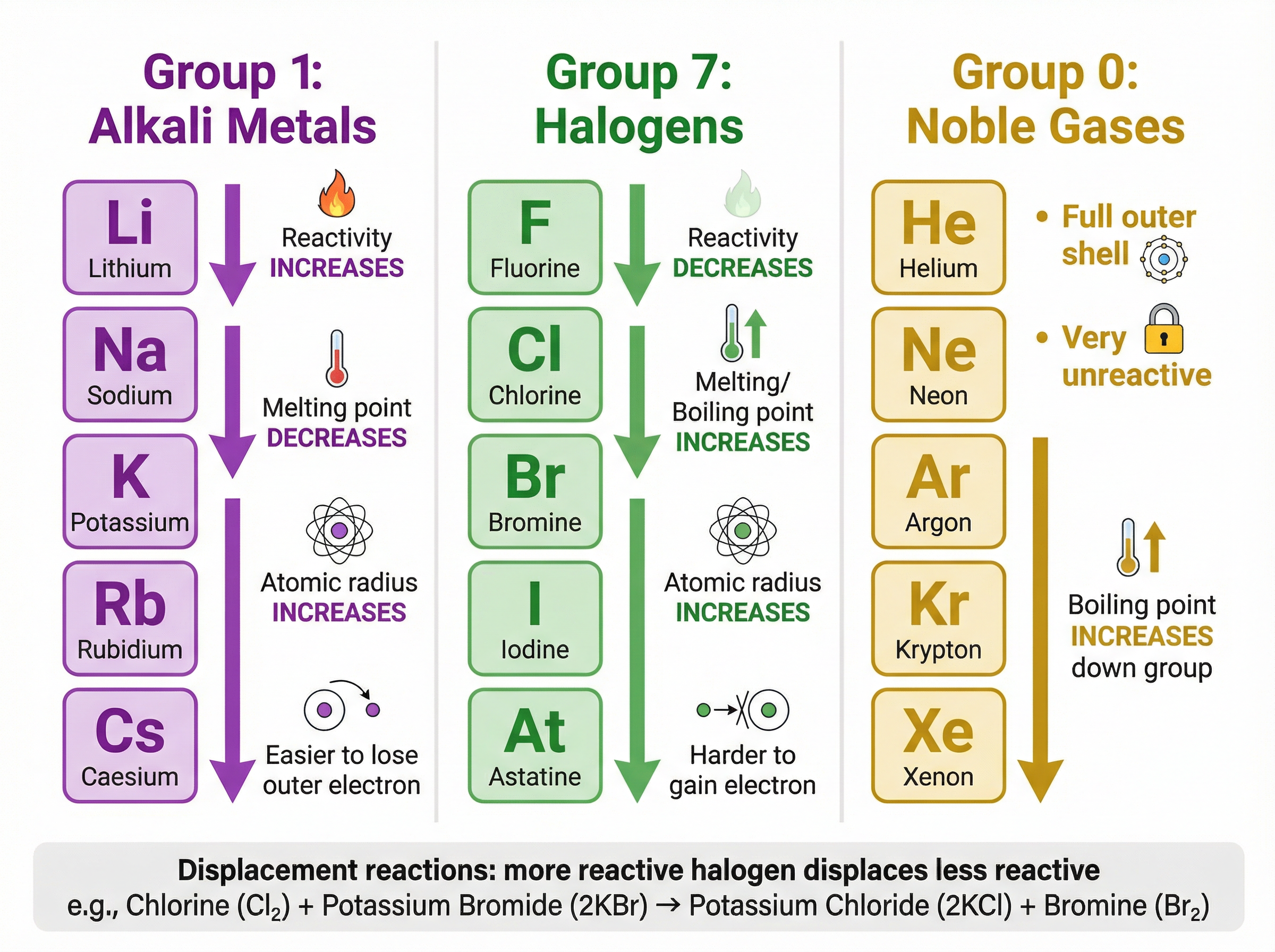
Concept 1: Group 1 - The Alkali Metals
The alkali metals (Lithium, Sodium, Potassium, Rubidium, Caesium) occupy the first column of the Periodic Table. They all have one electron in their outermost shell, which defines their chemical properties. They react by losing this single electron to form a 1+ ion.
The most important trend is that reactivity increases as you go down the group.
Why does this happen? As you descend the group, the atoms have more electron shells. This means the outermost electron is further away from the positive attraction of the nucleus. Additionally, the inner electron shells "shield" the outer electron from the nuclear charge. Because the electrostatic attraction is weaker, less energy is required to overcome it and lose the outer electron.
Example: When potassium reacts with water, it is vigorous enough to ignite the hydrogen gas produced, burning with a characteristic lilac flame.
2K(s) + 2H_2O(l) \rightarrow 2KOH(aq) + H_2(g)
Concept 2: Group 7 - The Halogens
The halogens (Fluorine, Chlorine, Bromine, Iodine, Astatine) are non-metals in Group 7. They have seven electrons in their outer shell and react by gaining one electron to form a 1- ion (a halide ion).
Conversely to Group 1, reactivity decreases as you go down Group 7.
Why? To react, a halogen atom must attract an incoming electron to complete its outer shell. As you go down the group, the outer shell is further from the nucleus and shielded by more inner shells. Therefore, the electrostatic attraction from the nucleus to the incoming electron is weaker, making it harder to gain an electron.
Example: A more reactive halogen will displace a less reactive halogen from an aqueous solution of its salt. If you add chlorine water to potassium bromide solution, the chlorine displaces the bromine, turning the solution orange.
Cl_2(aq) + 2KBr(aq) \rightarrow 2KCl(aq) + Br_2(aq)
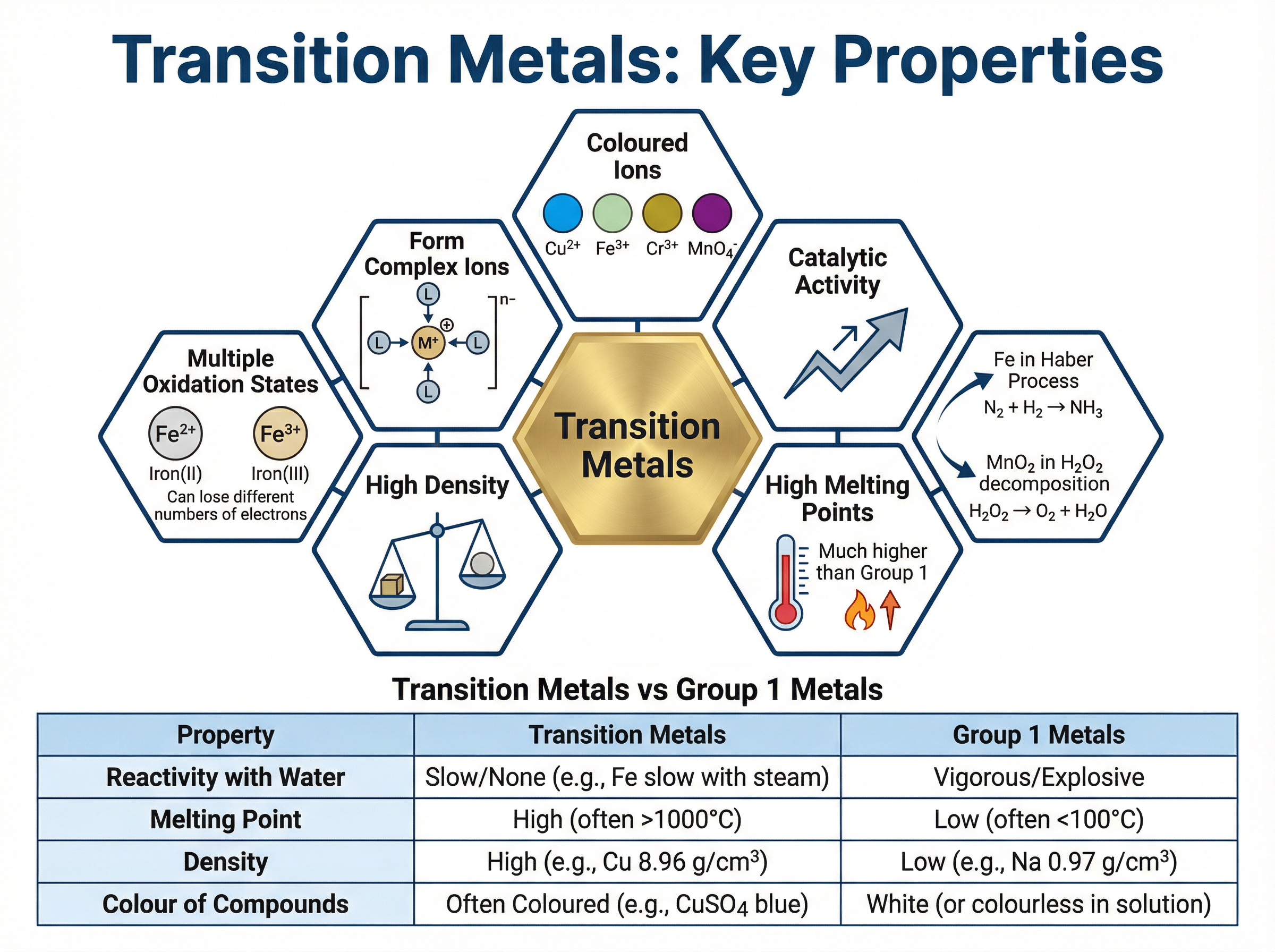
Concept 3: Transition Metals
The transition metals sit in the central block of the Periodic Table. Unlike Group 1 metals, they are typically hard, dense, and have high melting points. They are uniquely characterised by their ability to form ions with different charges (multiple oxidation states), form coloured compounds, and act as effective catalysts.
Example: Iron can form Fe^{2+} (which forms pale green compounds) and Fe^{3+} (which forms yellow-brown compounds). Iron is also the crucial catalyst in the Haber process for manufacturing ammonia.
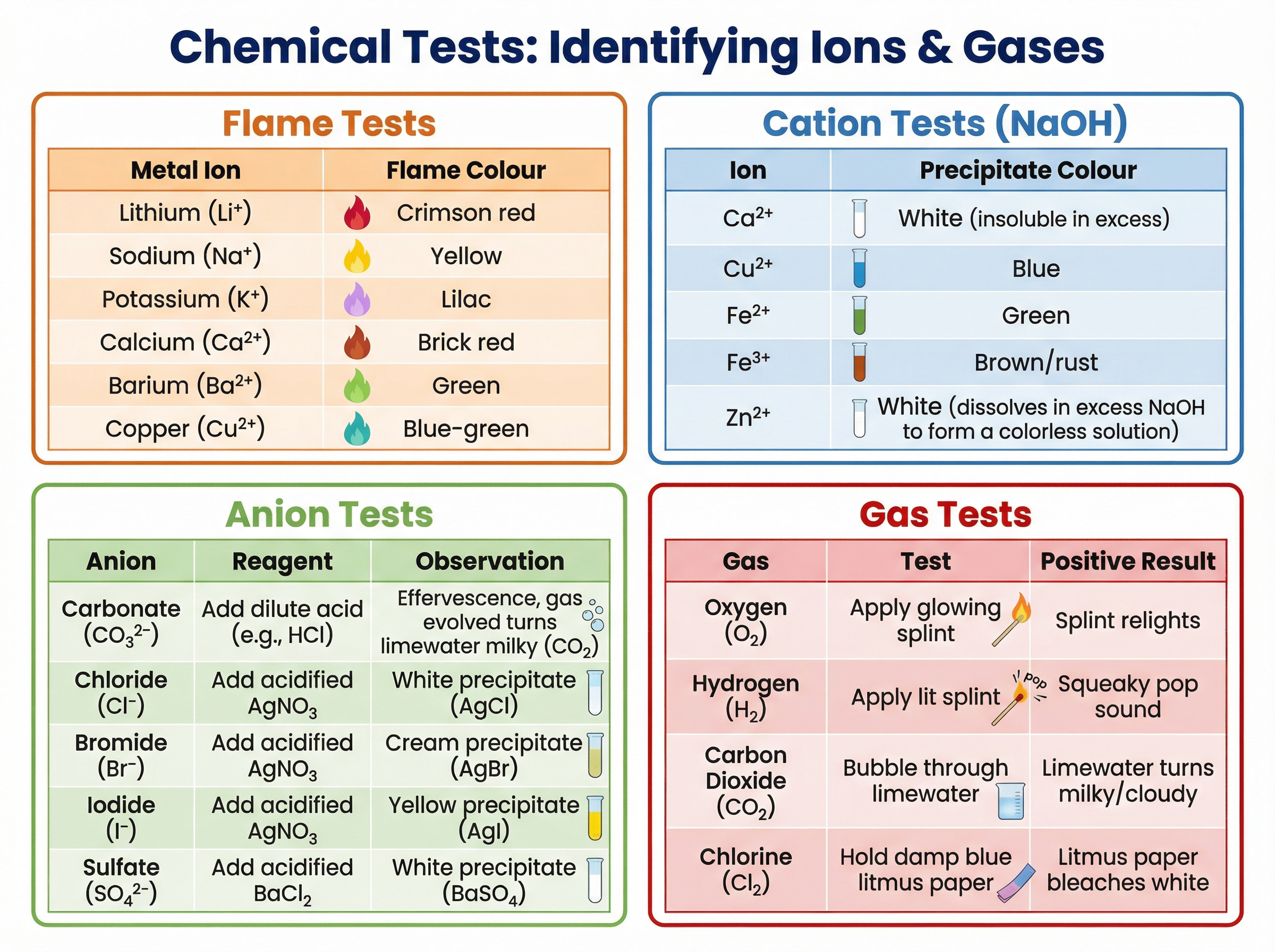
Concept 4: Chemical Tests for Gases and Ions
Analytical chemistry relies on specific tests to identify unknown substances. You must memorise the reagents, procedures, and positive results for these tests.
Gas Tests:
- Oxygen (O_2): Relights a glowing splint.
- Hydrogen (H_2): Burns with a 'squeaky pop' when a lit splint is applied.
- Carbon Dioxide (CO_2): Turns limewater (calcium hydroxide solution) milky/cloudy when bubbled through.
- Chlorine (Cl_2): Bleaches damp blue litmus paper white.
**Cation Tests (Positive Ions):**We use sodium hydroxide (NaOH) solution to identify metal cations based on the colour of the insoluble hydroxide precipitate formed.
- Cu^{2+}: Blue precipitate
- Fe^{2+}: Green precipitate
- Fe^{3+}: Brown/rust precipitate
- Ca^{2+}: White precipitate (insoluble in excess NaOH)
- Zn^{2+}: White precipitate (dissolves in excess NaOH to form a colourless solution)
Flame Tests:
- Lithium (Li^+): Crimson red
- Sodium (Na^+): Yellow
- Potassium (K^+): Lilac
- Calcium (Ca^{2+}): Brick red
- Barium (Ba^{2+}): Green
- Copper (Cu^{2+}): Blue-green
Anion Tests (Negative Ions):
- Carbonates (CO_3^{2-}): Add dilute acid. Effervescence occurs. Bubble the gas through limewater; it turns milky.
- Halides (Cl^-, Br^-, I^-): Add dilute nitric acid, then silver nitrate solution (AgNO_3).
- Chloride gives a white precipitate (AgCl).
- Bromide gives a cream precipitate (AgBr).
- Iodide gives a yellow precipitate (AgI).
- Sulfates (SO_4^{2-}): Add dilute hydrochloric acid, then barium chloride solution (BaCl_2). A white precipitate of barium sulfate (BaSO_4) forms.
Mathematical/Scientific Relationships
- Ionic Equations for Precipitation: You must be able to write state symbols for precipitation reactions.
Ag^+(aq) + Cl^-(aq) \rightarrow AgCl(s)
The (aq) indicates aqueous (dissolved in water), and (s) indicates the solid precipitate.
Practical Applications
- Instrumental Analysis: Modern laboratories use machines like Mass Spectrometers and Gas Chromatographs instead of simple chemical tests. These are highly valued because they are highly sensitive (can detect tiny amounts), highly accurate, and very fast.
- Water Treatment: Testing for specific ions like chloride and sulfate is essential in monitoring drinking water quality and environmental pollution.



