Study Notes

Overview
Welcome to Atomic Structure, the foundational topic of your A-Level Chemistry journey. This section moves beyond the simple models of GCSE to the more sophisticated quantum mechanical model of the atom. Understanding how electrons, protons, and neutrons behave is crucial as it underpins everything else you'll study, from bonding and periodicity to reaction mechanisms. WJEC examiners frequently test this topic through calculation questions (mass spectrometry), explanation questions (ionisation energy trends), and application questions (electronic configurations of ions). Expect to see multi-step problems that require you to link different concepts together.
Key Concepts
1. The Quantum Mechanical Model of the Atom
At A-Level, we abandon the idea of electrons orbiting the nucleus in fixed shells. Instead, we describe their location using orbitals: regions of space where there is a high probability (usually 95%) of finding an electron. These orbitals have specific shapes and energy levels.
- Principal Quantum Shells (n): These are the main energy levels, numbered 1, 2, 3, etc., increasing in energy and distance from the nucleus as the number increases.
- Subshells: Within each principal shell (from n=2 upwards), there are subshells with slightly different energies. These are labelled s, p, d, and f.
- Orbitals: Each subshell contains one or more orbitals.
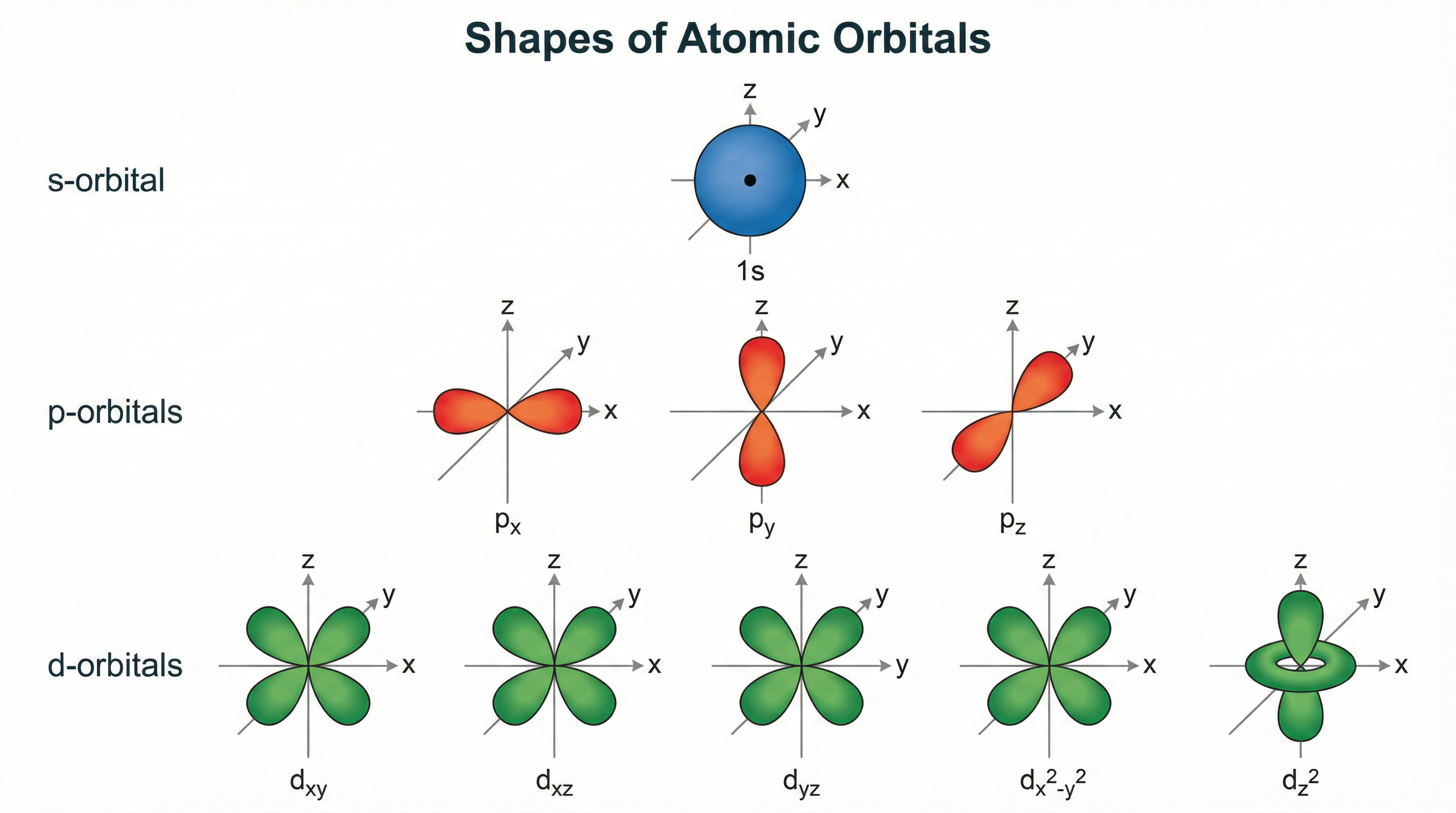
- s-subshells have 1 spherical orbital.
- p-subshells have 3 dumbbell-shaped orbitals (px, py, pz).
- d-subshells have 5 more complex-shaped orbitals.
- f-subshells have 7 even more complex orbitals.
Each orbital can hold a maximum of two electrons, which must have opposite spins (the Pauli Exclusion Principle).
2. Electronic Configuration
This is the notation used to describe the arrangement of electrons in an atom. There are three rules to follow:
- Aufbau Principle: Electrons fill the lowest energy orbitals first. The order of filling is: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p... Note that the 4s subshell has a lower energy than the 3d subshell, so it fills first.
- Hund's Rule: When filling orbitals of equal energy (degenerate orbitals, like the three p-orbitals), electrons fill each orbital singly before any pairing occurs. This minimises electron-electron repulsion.
- Pauli Exclusion Principle: An orbital can hold a maximum of two electrons, and they must have opposite spins.
Example: The electronic configuration of Silicon (14 electrons) is 1s² 2s² 2p⁶ 3s² 3p². For the 3p² electrons, one will be in the 3px orbital and one in the 3py orbital.
Exceptions: For WJEC, you must know two key exceptions: Chromium (Cr) and Copper (Cu). To achieve greater stability, they promote a 4s electron to the 3d subshell.
- Chromium (Cr): Expected: [Ar] 4s² 3d⁴. Actual: [Ar] 4s¹ 3d⁵ (a half-filled d-subshell is stable).
- Copper (Cu): Expected: [Ar] 4s² 3d⁹. Actual: [Ar] 4s¹ 3d¹⁰ (a full d-subshell is stable).
When forming ions, transition metals lose their 4s electrons first before the 3d electrons.
3. Ionisation Energy
First Ionisation Energy (IE) is the energy required to remove one mole of electrons from one mole of gaseous atoms to form one mole of gaseous 1+ ions. The key factors affecting IE are:
- Nuclear Charge: More protons in the nucleus lead to a stronger attraction for the outer electrons, increasing IE.
- Atomic Radius: A larger distance between the nucleus and the outer electron weakens the attraction, decreasing IE.
- Shielding: Inner shells of electrons repel the outer electrons, reducing the effective nuclear charge felt by them. More inner shells mean more shielding and lower IE.
Trends in the Periodic Table:
- Across a Period: IE generally increases. Nuclear charge increases and shielding remains approximately constant, pulling the electron shell closer and making electrons harder to remove.
- Down a Group: IE decreases. The number of inner shells increases, leading to greater shielding and a larger atomic radius, which outweighs the increase in nuclear charge.
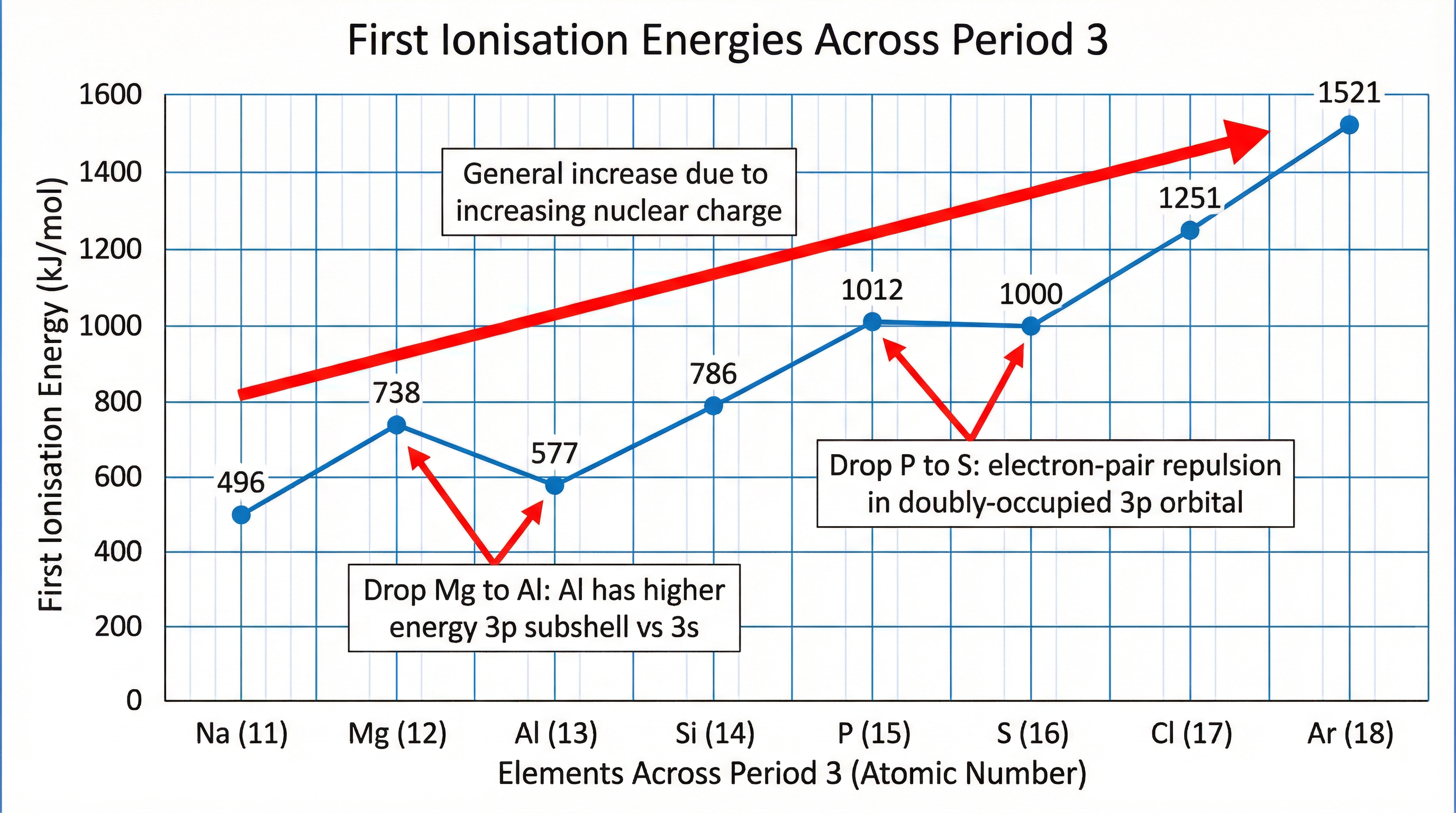
There are two important dips in the trend across Period 3:
- Group 2 to 3 (e.g., Mg to Al): The IE drops because the electron being removed from Aluminium is in a 3p orbital, which is higher in energy and further from the nucleus than the 3s orbital of Magnesium.
- Group 5 to 6 (e.g., P to S): The IE drops because in Sulphur, the electron is being removed from a doubly-occupied 3p orbital. The repulsion between the two paired electrons makes it easier to remove one.
4. Mass Spectrometry
Mass spectrometry is used to determine the relative atomic mass (Ar) of an element. It separates ions based on their mass-to-charge (m/z) ratio. The output is a mass spectrum showing the relative abundance of each isotope.
Calculation: The relative atomic mass is the weighted mean mass of all isotopes.
Ar = (Σ (isotope mass × abundance)) / (Σ abundance)
Example: A sample of Boron is found to contain 19.9% of ¹⁰B and 80.1% of ¹¹B.
Ar = ((10 × 19.9) + (11 × 80.1)) / 100 = (199 + 881.1) / 100 = 10.81
Podcast Episode
Listen to our dedicated podcast episode for a full audio walkthrough of this topic, including exam tips and a quick-fire quiz.
Mathematical/Scientific Relationships
- Relative Atomic Mass Formula:
Ar = (Σ (isotope mass × abundance)) / (Σ abundance)(Must memorise) - Energy of a Photon:
E = hν(where h is Planck's constant and ν is frequency). This is given on the formula sheet but its application to emission spectra is key.
Practical Applications
- Mass Spectrometry: Used in forensic science, drug testing in sport, and carbon dating.
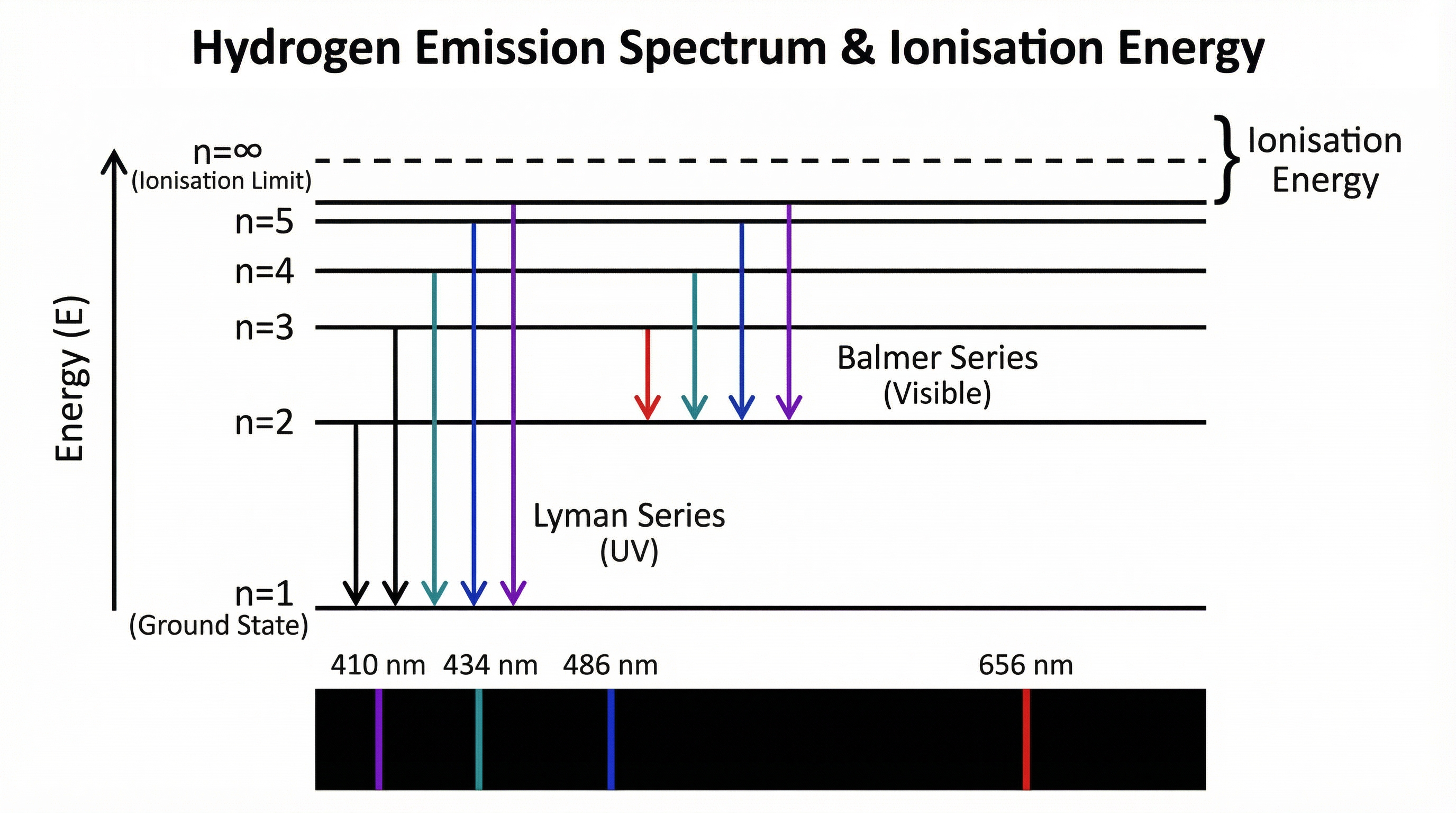
- Atomic Emission Spectra: The unique line spectra of elements are used in astronomy to determine the composition of stars and in fireworks to create different colours.
Visual Resources
3 diagrams and illustrations
Interactive Diagrams
2 interactive diagrams to visualise key concepts
Flowchart showing the processes of excitation and ionisation.
Concept map showing the general increase and key drops in first ionisation energy across Period 3.
Worked Examples
3 detailed examples with solutions and examiner commentary
Practice Questions
Test your understanding — click to reveal model answers
The element with the atomic number 24 has an anomalous electronic configuration. State and explain this configuration. (3 marks)
Hint: Think about the stability of d-subshells. Which element is Z=24?
Explain why the second ionisation energy of sodium is significantly higher than its first ionisation energy. (3 marks)
Hint: Consider the electronic configurations of Na and Na⁺. Where is the second electron being removed from?
A sample of magnesium contains three isotopes: ²⁴Mg (78.6%), ²⁵Mg (10.1%), and ²⁶Mg (11.3%). Calculate the relative atomic mass of magnesium to three significant figures. (3 marks)
Hint: Use the standard formula for calculating relative atomic mass from percentage abundances.
Describe the shape of a p-orbital and state how many p-orbitals are found in a p-subshell. (2 marks)
Hint: What does a p-orbital look like? How many orientations can it have?
The successive ionisation energies of an element X are shown below (in kJ/mol): 738, 1451, 7733, 10543. In which group of the periodic table is element X found? Explain your reasoning. (3 marks)
Hint: Look for a large jump in the ionisation energies. What does this jump signify?