Study Notes

Overview
Welcome to the study of Atoms and Isotopes, a cornerstone of your AQA GCSE Physics course. This topic unpacks the fundamental building blocks of matter, exploring the atom's structure and the reasons behind nuclear instability and radioactivity. Understanding this area is crucial, as it forms the bedrock for later topics like Nuclear Fission and Fusion (4.4.3). Examiners frequently test this content through a mix of short-answer definition questions, historical model comparisons, and longer, structured calculation questions, particularly on radioactive decay and half-life. A solid grasp of the concepts here will not only earn you direct marks but also provide the synoptic links needed to excel in more complex problems across the physics specification.
Key Concepts
Concept 1: The Structure of the Atom
At the heart of all matter is the atom, which is itself composed of smaller, subatomic particles. For your exam, you must be confident with the properties and locations of these three particles:
- Protons: Found in the central nucleus, they have a relative positive charge of +1 and a relative mass of 1.
- Neutrons: Also in the nucleus, they have no charge (they are neutral) and a relative mass of 1.
- Electrons: Orbit the nucleus in specific energy levels or 'shells'. They have a relative negative charge of -1 and a negligible mass (approximately 1/1840th of a proton).
The nucleus is incredibly small and dense compared to the rest of the atom. It contains all the protons and neutrons, meaning it holds virtually all the atom's mass and all of its positive charge. The electrons orbit at a significant distance, meaning most of the atom is, in fact, empty space. This is a key conclusion from the Rutherford scattering experiment.
Example: A neutral atom of Lithium-7 has 3 protons and 4 neutrons in its nucleus. To be neutral, it must have an equal number of electrons to protons, so it has 3 electrons orbiting the nucleus.
Concept 2: Atomic Number, Mass Number and Isotopes
Two numbers are used to describe the composition of a nucleus:
- Atomic Number (Z): This is the number of protons in the nucleus. It defines the element. Every atom of Carbon has 6 protons; if it had 7, it would be Nitrogen.
- Mass Number (A): This is the total number of protons and neutrons in the nucleus.
From these, you can always calculate the number of neutrons: Number of Neutrons = Mass Number (A) - Atomic Number (Z).
This leads to the crucial definition of isotopes. Isotopes are atoms of the same element with the same number of protons but a different number of neutrons. Because they have the same number of protons, they have the same atomic number and identical chemical properties. However, their different neutron counts mean they have different mass numbers and can have different physical properties, such as stability.
Example: Carbon-12 (6 protons, 6 neutrons) is a stable isotope. Carbon-14 (6 protons, 8 neutrons) is an unstable, radioactive isotope used in carbon dating. Both are chemically carbon.
Concept 3: The Evolution of the Atomic Model
Your understanding of the atom must be placed in historical context. AQA requires you to compare the Plum Pudding model with the modern Nuclear model.
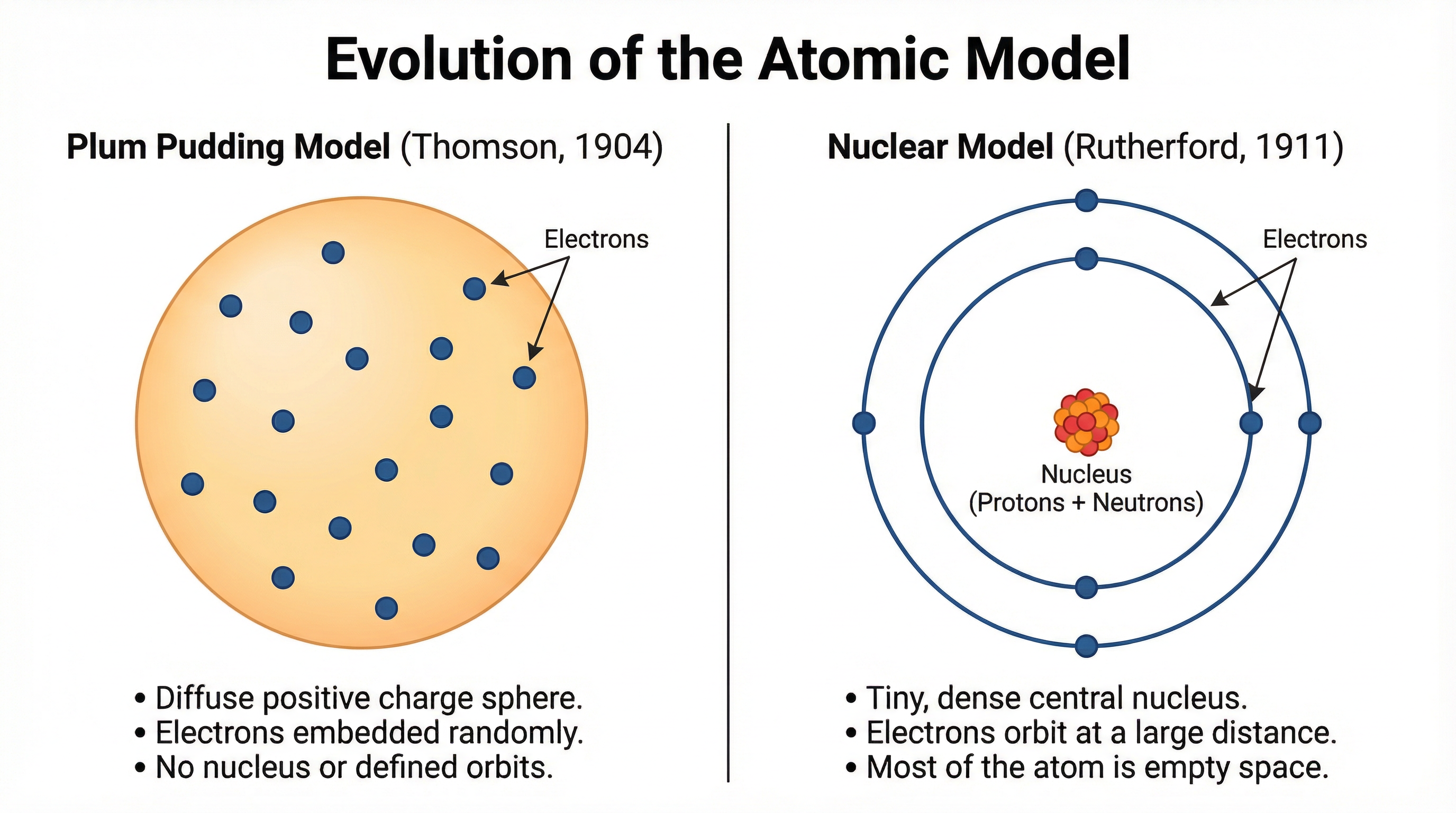
- Plum Pudding Model (J.J. Thomson, 1904): After discovering the electron, Thomson proposed the atom was a sphere of positive charge with negative electrons embedded within it. There was no nucleus.
- Nuclear Model (Ernest Rutherford, 1911): The results of the gold foil experiment disproved the plum pudding model. By firing alpha particles at thin gold foil, Rutherford observed that while most passed through, some were deflected and a very small number were repelled at large angles. This could only be explained if the atom's positive charge and mass were concentrated in a tiny, dense central nucleus, with the electrons orbiting at a distance. This discovery demonstrated that the atom is mostly empty space.
Concept 4: Radioactive Decay
Unstable isotopes undergo radioactive decay to become more stable. This is a random process, meaning you cannot predict when a specific nucleus will decay. They emit one or more types of ionising radiation:
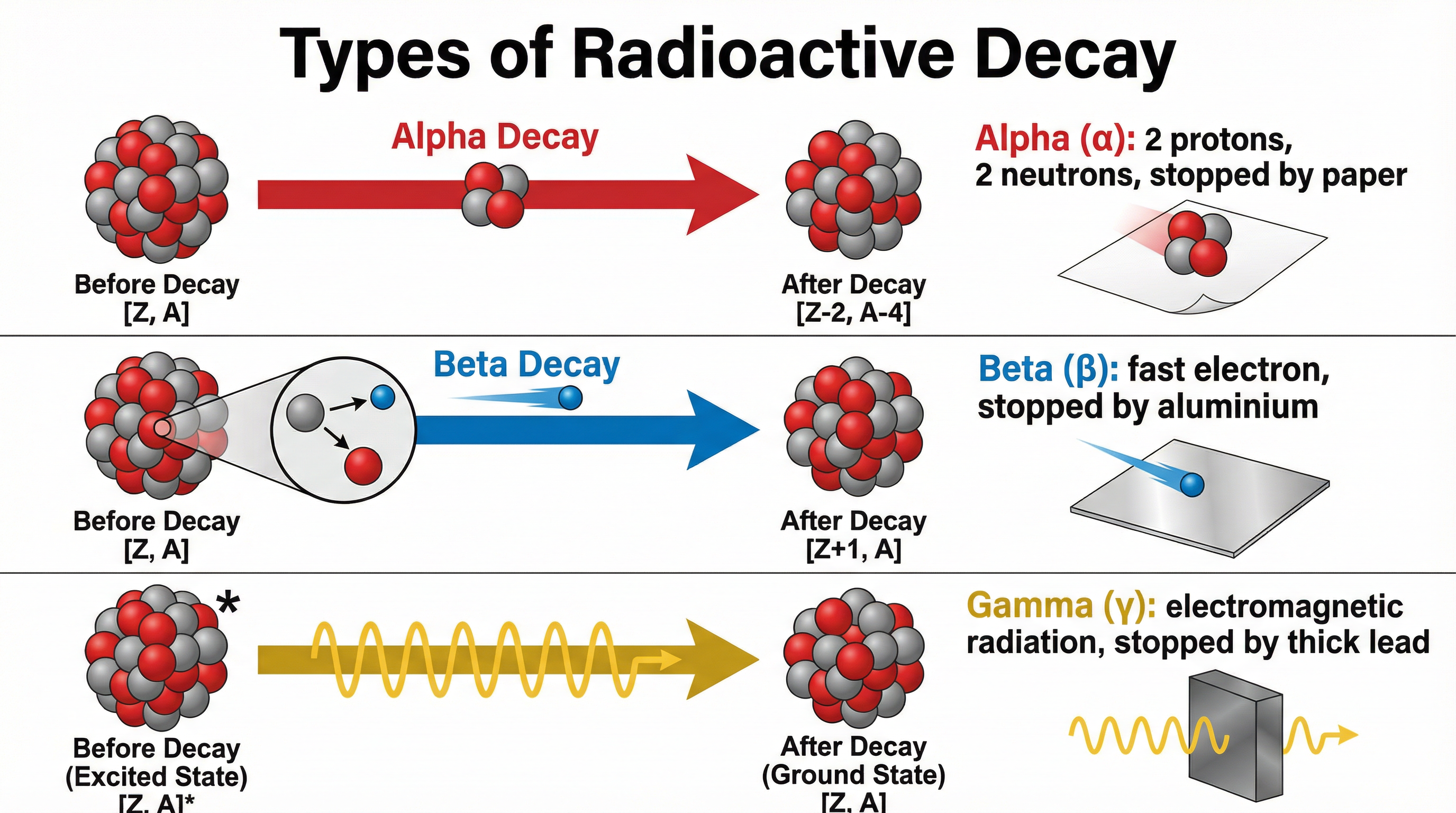
- Alpha (α): An alpha particle consists of two protons and two neutrons (a Helium nucleus). When emitted, the nucleus's mass number decreases by 4 and its atomic number decreases by 2. Alpha is highly ionising but has low penetration (stopped by paper or a few cm of air).
- Beta (β): A beta particle is a high-speed electron. It is emitted when a neutron in the nucleus turns into a proton. This means the mass number stays the same, but the atomic number increases by 1. Beta is moderately ionising and moderately penetrating (stopped by a few mm of aluminium).
- Gamma (γ): Gamma radiation is a high-energy electromagnetic wave, not a particle. It is emitted from a nucleus when it needs to lose excess energy after a previous alpha or beta decay. It does not change the mass or atomic number. Gamma is weakly ionising but highly penetrating (requiring thick lead or concrete to absorb it).
Concept 5: Half-Life
The half-life of a radioactive isotope is the time it takes for the number of radioactive nuclei in a sample to halve, or for the activity (count rate) of the sample to fall to half its initial level. This is a constant value for a given isotope and is unaffected by external conditions.
Example: If a sample has an activity of 800 Bq and a half-life of 10 days, after 10 days its activity will be 400 Bq. After another 10 days (20 days total), its activity will be 200 Bq.
Mathematical/Scientific Relationships
Nuclear Equations
All nuclear decay equations must be balanced. The total mass number and total atomic number must be the same on both sides of the equation.
- Alpha Decay: [^{A}{Z}X
ightarrow ^{A-4}{Z-2}Y + ^{4}_{2}\alpha] - Beta Decay: [^{A}{Z}X
ightarrow ^{A}{Z+1}Y + ^{0}_{-1}\beta]
Half-Life Calculations (Higher Tier)
The fraction of a radioactive substance remaining after 'n' half-lives can be calculated as:
Fraction remaining = (1/2)^nTo find the final activity, multiply the initial activity by this fraction. A key practical skill is remembering to subtract any background radiation from measurements before performing half-life calculations.
Practical Applications
This topic is linked to the required practical activity on radiation absorption. A typical setup involves placing a radioactive source at a fixed distance from a Geiger-Müller (GM) tube, which is connected to a counter. Different materials (e.g., paper, aluminium, lead) of varying thicknesses are placed between the source and the detector. By measuring the count rate for each material, candidates can determine the penetrating power of the radiation and identify it as alpha, beta, or gamma. Safety is paramount: sources should be handled with tongs, kept in lead-lined containers, and pointed away from the body.
"
Worked Examples
3 detailed examples with solutions and examiner commentary
Practice Questions
Test your understanding — click to reveal model answers
State two differences between an atom of Uranium-235 and an atom of Uranium-238.
Hint: Think about the definition of an isotope. What is the same and what is different?
Describe the evidence from the alpha particle scattering experiment that supported the conclusion that an atom is mostly empty space.
Hint: What did MOST of the alpha particles do, and what did this imply?
A neutron in the nucleus of a carbon-14 atom decays by beta emission. Write a balanced nuclear equation for this decay. (Carbon has an atomic number of 6).
Hint: Remember what happens to the atomic and mass numbers during beta decay.
A doctor is choosing a radioactive isotope to use as a medical tracer to investigate a patient's kidneys. The isotope will be injected into the bloodstream. Evaluate the use of an alpha emitter versus a gamma emitter for this purpose, considering their properties. (6 marks)
Hint: Think about what the radiation needs to do (be detected outside the body) and what it must not do (cause harm). Link this to penetration and ionisation.
The half-life of Cobalt-60 is 5.27 years. If a source initially contains 8.0 x 10²² atoms, calculate how many radioactive atoms will remain after 21.08 years.
Hint: First, work out how many half-lives have passed.