Overview

Specific Heat Capacity is a fundamental concept in the GCSE Physics Energy topic. It explains the relationship between thermal energy, mass, and temperature change. Understanding this topic is crucial because it bridges theoretical physics with real-world applications, such as why water is an excellent coolant for car engines and why coastal areas experience milder climates than inland regions.
Examiners frequently test this topic through calculation questions requiring the rearrangement of the ΔE = mcΔθ equation, as well as extended response questions based on the Required Practical. To succeed, candidates must be comfortable with unit conversions (especially grams to kilograms) and interpreting experimental data.

Key Concepts
Concept 1: Defining Specific Heat Capacity
Specific Heat Capacity (c) is defined as the amount of energy required to raise the temperature of one kilogram of a substance by one degree Celsius.
Different materials have different specific heat capacities. Materials with a high specific heat capacity (like water at 4200 J/kg°C) require a large amount of energy to change their temperature. They heat up slowly and cool down slowly. Conversely, materials with a low specific heat capacity (like copper at 385 J/kg°C) require very little energy to change their temperature, meaning they heat up and cool down rapidly.
Example: If you heat 1 kg of water and 1 kg of copper with the same amount of energy, the copper's temperature will increase much more than the water's temperature because copper has a much lower specific heat capacity.
Concept 2: The Specific Heat Capacity Equation
The mathematical relationship is expressed as:
ΔE = m \times c \times Δθ
Where:
- ΔE = Change in thermal energy (Joules, J)
- m = Mass (kilograms, kg)
- c = Specific heat capacity (Joules per kilogram per degree Celsius, J/kg°C)
- Δθ = Temperature change (degrees Celsius, °C)

Candidates must be prepared to rearrange this equation to solve for any of the four variables. A common pitfall is forgetting to calculate the change in temperature (Δθ) by subtracting the initial temperature from the final temperature.
Concept 3: The Required Practical
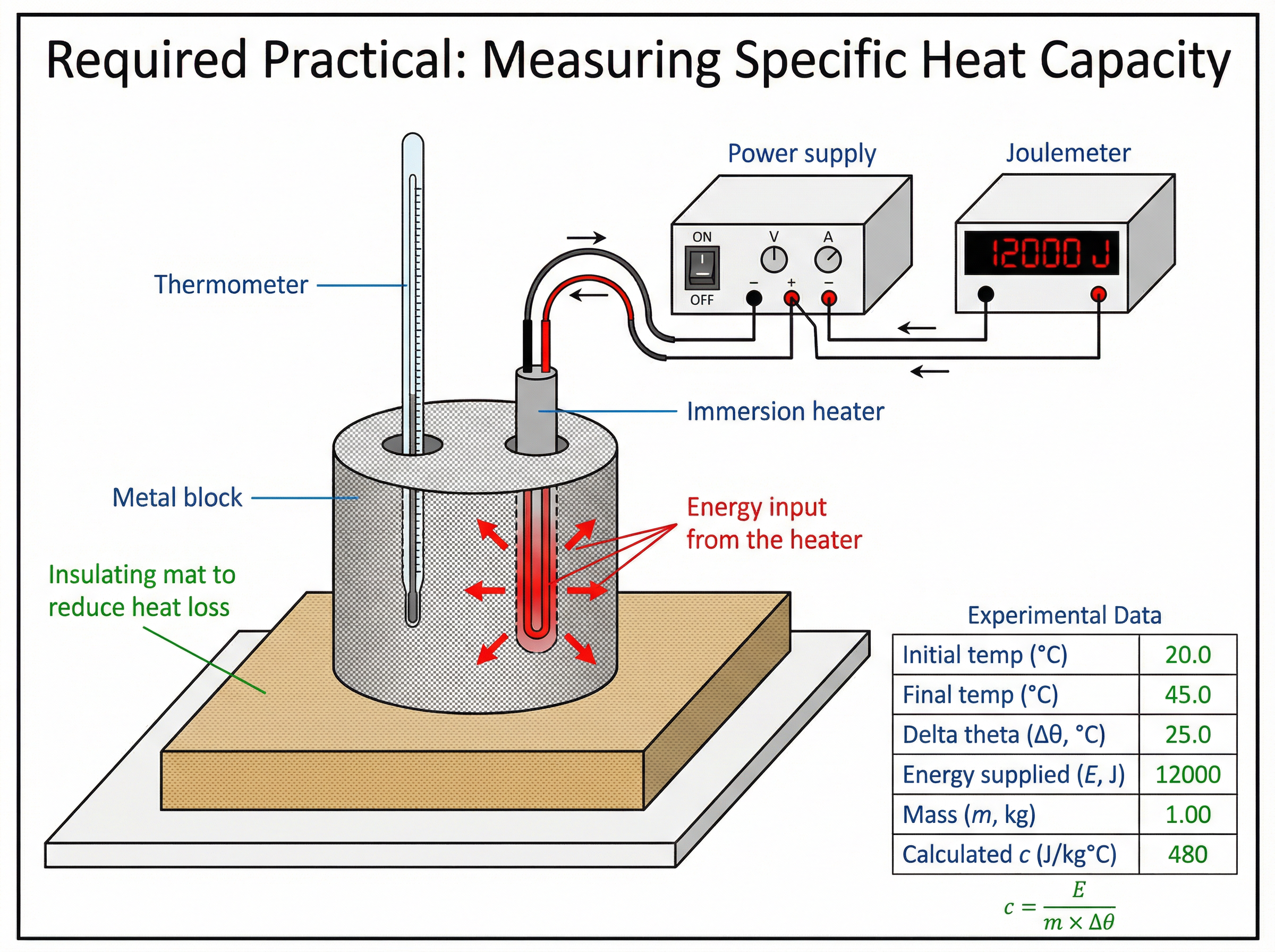
The required practical involves determining the specific heat capacity of a material (usually a metal block).
Method summary:
- Measure and record the mass of the metal block.
- Place an immersion heater into the central hole of the block.
- Place a thermometer into the smaller hole.
- Record the initial temperature of the block.
- Connect the heater to a power supply and a joulemeter.
- Turn on the power supply and leave it for a set time (e.g., 10 minutes).
- Record the final temperature and the total energy supplied (from the joulemeter).
- Calculate the temperature change (Δθ) and use the equation c = \frac{ΔE}{m \times Δθ} to find the specific heat capacity.
Key Examiner Focus: Examiners often ask why the calculated value of c is higher than the true value. The answer is heat loss to the surroundings. Because some energy from the heater warms the air rather than the block, the recorded ΔE is larger than the energy actually absorbed by the block, leading to an artificially high calculated c. Wrapping the block in insulation reduces this error.
Mathematical/Scientific Relationships
- ΔE = m c Δθ (Given on the physics equation sheet)
- m = \frac{ΔE}{c Δθ} (Must be able to rearrange)
- c = \frac{ΔE}{m Δθ} (Must be able to rearrange)
- Δθ = \frac{ΔE}{m c} (Must be able to rearrange)
- P = \frac{E}{t} (Power = Energy / time). This is often linked in higher-tier questions where energy (ΔE) is calculated first from Power \times time.
Practical Applications
- Central Heating Systems: Water is used in radiators because its high specific heat capacity allows it to carry a large amount of thermal energy around the house.
- Coolants: Water is used as a coolant in car engines and industrial plants because it can absorb a lot of heat without boiling.
- Climate: Coastal regions have milder winters and cooler summers than inland areas because the ocean acts as a massive thermal reservoir, absorbing heat in summer and releasing it in winter.


