Brownian Motion — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
This guide provides a deep dive into Brownian motion for OCR GCSE Physics (6.9), explaining the crucial evidence it provides for the kinetic particle model. Master the concepts of random particle motion and unbalanced collisions to secure top marks in your exam.

## Overview
Brownian motion is one of the most compelling pieces of evidence for the existence of atoms and molecules, a cornerstone of the kinetic particle theory. For your OCR GCSE Physics exam, this topic (specification point 6.9) requires you to understand and explain the random, unpredictable movement of particles suspended in a fluid (a liquid or a gas). It's not just about observing the motion; it's about explaining *why* it happens. You'll need to articulate how tiny, invisible, fast-moving particles of the fluid collide with larger, visible suspended particles, causing them to move. Examiners will test your ability to distinguish between the observed particle (like a smoke particle) and the unobserved cause (the air molecules). A typical exam question might ask you to "Explain the motion of smoke particles in a smoke cell experiment," which requires a detailed, step-by-step account of the underlying physics. This topic forms a critical link between the macroscopic world we can see and the microscopic world of particles, connecting directly to concepts of energy, forces, and the states of matter.

## Key Concepts
### Concept 1: The Observation - Random, Unpredictable Motion
The core observation of Brownian motion is that small particles (like pollen in water or smoke in air) move about in a haphazard way. This isn't a smooth, predictable path; it's a jerky, zigzag journey. For the exam, it is crucial to use precise language. Credit is awarded for terms like **random**, **unpredictable**, or **zigzag motion**. Avoid vague descriptions like "jiggling" or "wobbling." This motion never stops, as long as the temperature is above absolute zero, because the fluid particles are always in motion.
**Example**: When Robert Brown first observed pollen grains in water in 1827, he saw them darting about. He initially thought they might be alive, but later confirmed the same motion with inanimate dust particles. This showed the motion was a property of the physics, not the biology.
### Concept 2: The Cause - The Kinetic Particle Model and Unequal Bombardment
The explanation for Brownian motion lies in the **kinetic particle model**. This model states that all matter is composed of a vast number of tiny particles (atoms, molecules, ions) that are in constant, random motion. The energy of this motion is related to temperature; the hotter the substance, the faster its particles move.
Now, imagine a single, relatively large smoke particle suspended in the air. It is surrounded by millions of tiny, invisible air molecules. These air molecules are moving incredibly fast (hundreds of metres per second) and are constantly colliding with the smoke particle from all directions.
At any given instant, by pure chance, more air molecules might collide with the left side of the smoke particle than the right side. This creates an **unbalanced force** (or **unequal bombardment**), causing a net force on the smoke particle and pushing it to the right. A fraction of a second later, more molecules might hit it from the bottom, pushing it upwards. Because the number of collisions on each side is constantly and randomly changing, the resulting net force changes direction from moment to moment, leading to the observed zigzag path.
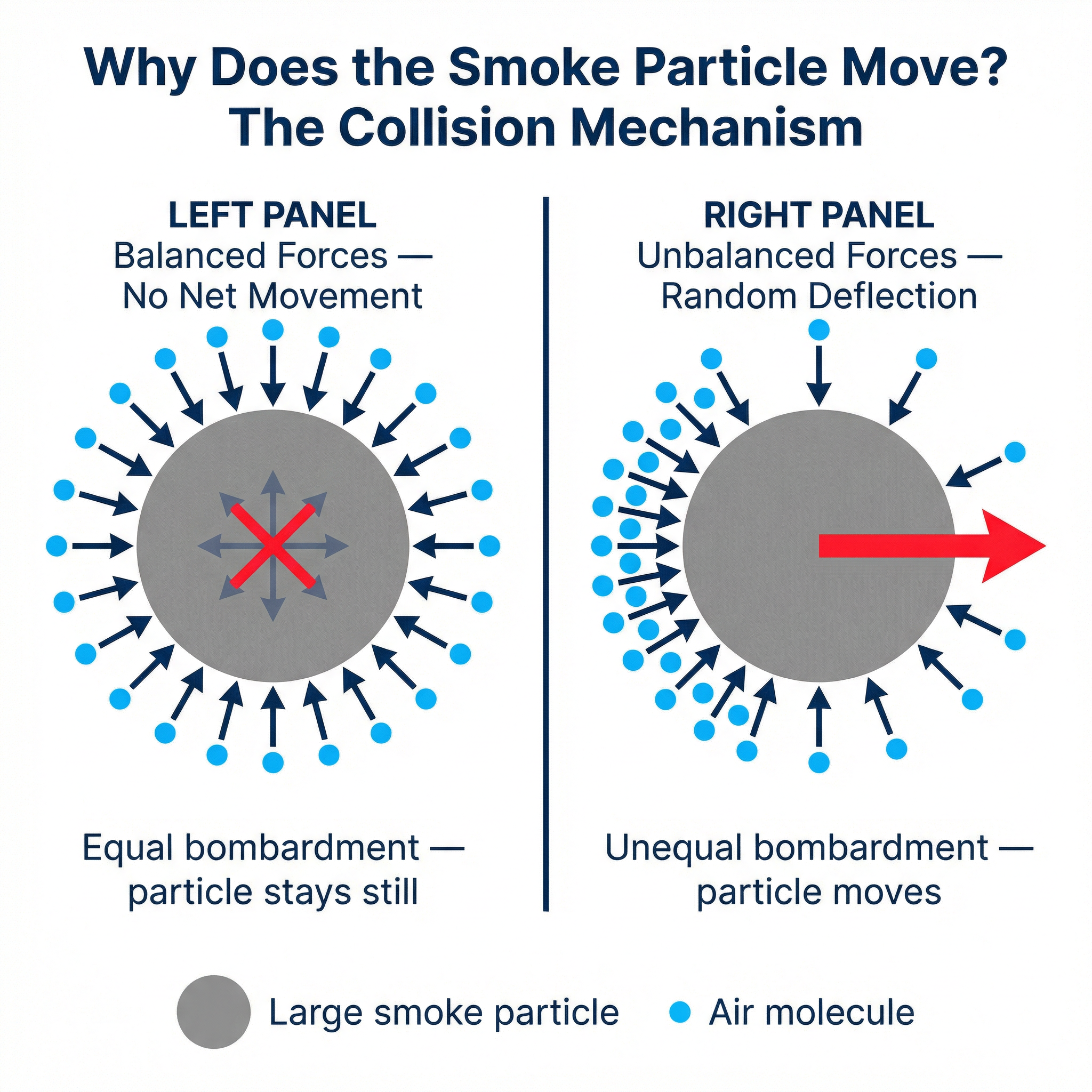
**Key Distinction for Marks**: Examiners will penalise candidates who confuse the particles. You **observe** the large smoke particle moving. You **infer** the existence and motion of the tiny, invisible air molecules. A common mistake is to say the smoke particles are colliding with each other; this is incorrect and will lose you marks. The motion is caused by the *fluid* molecules hitting the *suspended* particle.
### Concept 3: The Smoke Cell Experiment
This is the classic required practical context for demonstrating Brownian motion in a gas.
- **Apparatus**: A light source, a converging lens, a small transparent container called a 'smoke cell', and a microscope.
- **Method**: A small amount of smoke is trapped in the cell. A bright light is shone through the side of the cell and focused by the lens. The observer looks down through the microscope.
- **Observation**: You cannot see the air molecules. You see the individual smoke particles as bright specks of light against a dark background (because they are scattering the light from the lamp into the microscope). These bright specks are observed to be moving continuously and randomly.
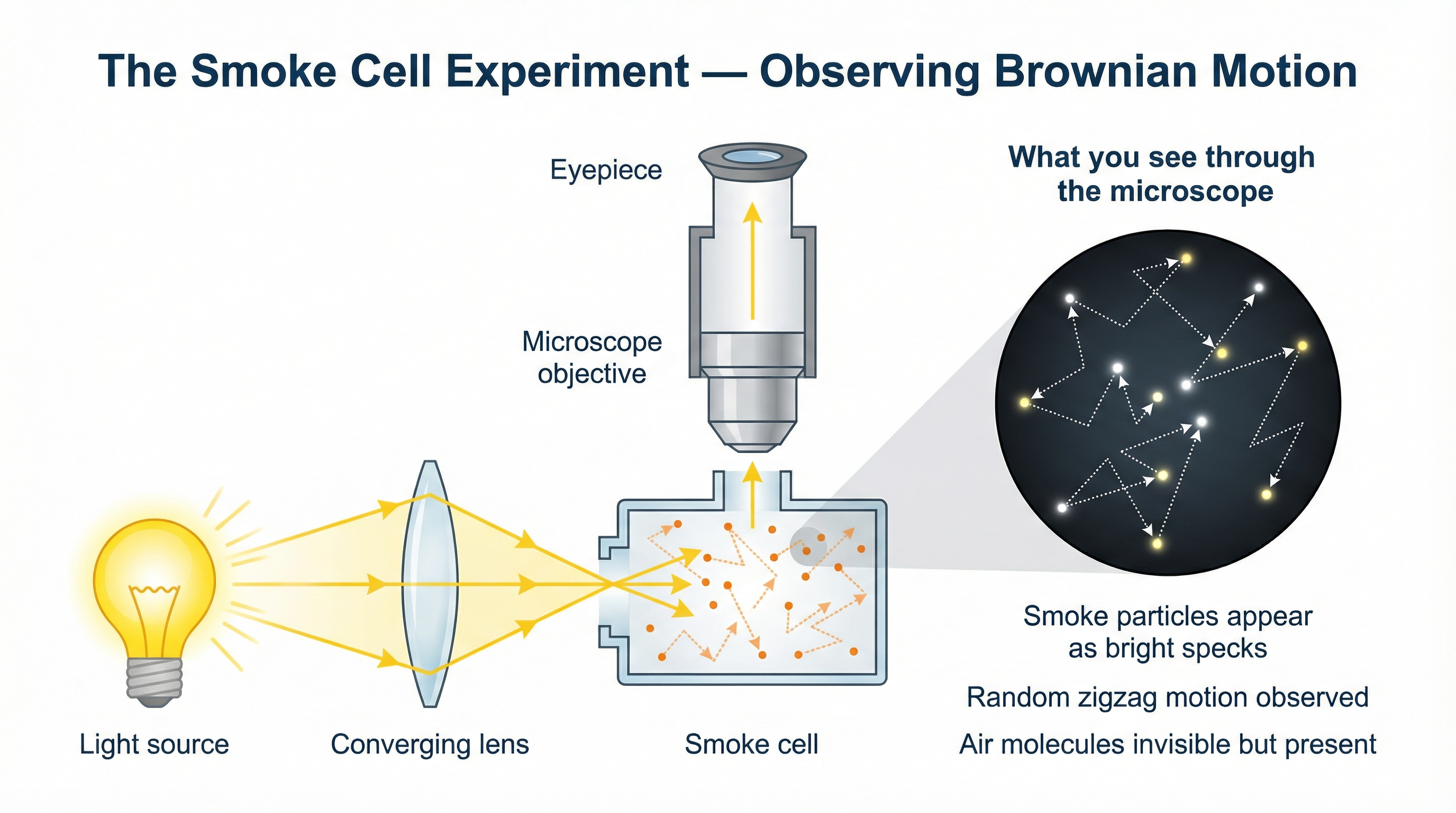
This experiment provides direct, visual evidence that invisible particles (air molecules) must exist and be in a state of constant, high-speed, random motion.
## Mathematical/Scientific Relationships
There are no specific mathematical formulas you need to memorise or calculate for Brownian motion at GCSE level. The relationships are purely conceptual:
- **Temperature and Kinetic Energy**: The average kinetic energy of the fluid particles is directly proportional to the absolute temperature (measured in Kelvin). If you increase the temperature of the air in the smoke cell, the air molecules will move faster and have more kinetic energy.
- **Effect on Brownian Motion**: Faster-moving air molecules lead to more frequent and more energetic collisions with the smoke particles, resulting in the smoke particles moving more vigorously. Their random walk becomes faster and covers more area. An exam question might ask you to describe and explain the effect of increasing the temperature on the motion of the smoke particles.
## Practical Applications
While Brownian motion itself isn't an engineering tool, understanding it is fundamental to many fields:
- **Atmospheric Science**: It helps model the diffusion and dispersal of pollutants and aerosols in the atmosphere.
- **Cell Biology**: The movement of organelles and molecules within the cytoplasm of a living cell is governed by the principles of Brownian motion and diffusion.
- **Finance**: The 'random walk' hypothesis in financial markets, which models stock price movements, is mathematically analogous to Brownian motion, suggesting that price changes are largely unpredictable.