Energy Transfer — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
This guide provides a comprehensive, exam-focused breakdown of Energy Transfer (OCR GCSE Physics 2.3). It covers the essential stores and pathways model, key calculations for efficiency and power, and the critical distinction between specific heat capacity and thermal conductivity, ensuring candidates are prepared to maximise their marks.
## Overview
Welcome to the definitive guide for OCR GCSE Physics Topic 2.3: Energy Transfer. This topic is a cornerstone of your physics education, forming the basis for understanding everything from how a light bulb works to the mechanics of a power station. In the exam, you will be assessed on your ability to apply the strict ‘stores and pathways’ model, perform precise calculations, and evaluate energy-saving measures. Examiners are looking for candidates who can articulate the Law of Conservation of Energy and avoid common pitfalls like saying energy is ‘lost’. This guide will equip you with the language, techniques, and deep understanding required to tackle AO1 (recall), AO2 (application), and AO3 (evaluation) questions with confidence. Expect to see everything from simple definitions to complex 6-mark questions on experimental design.

## Key Concepts
### Concept 1: The Law of Conservation of Energy
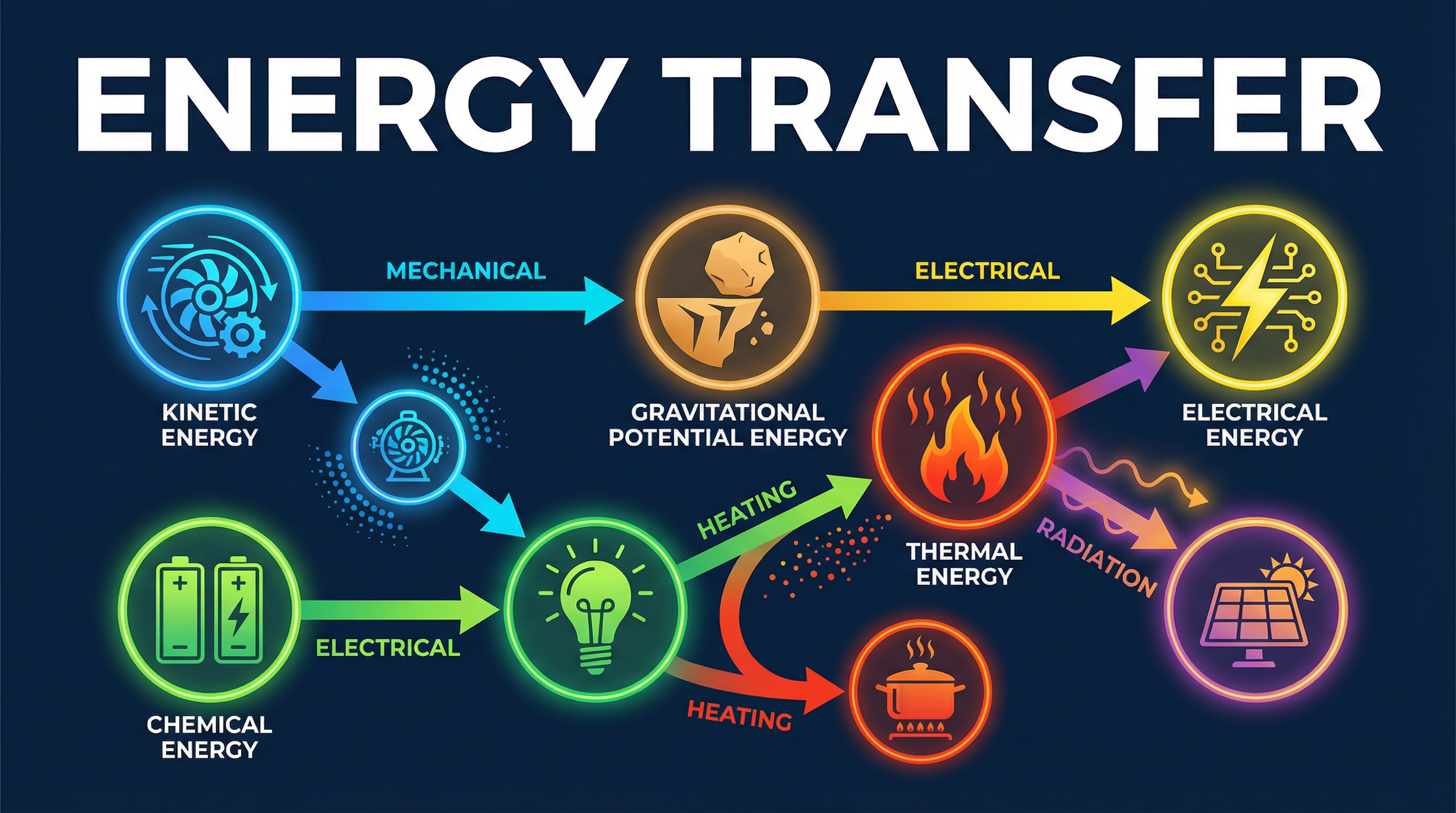
This is the single most important principle in this topic: **Energy can be transferred usefully, stored or dissipated, but it cannot be created or destroyed.** In any energy transfer, the total energy in a closed system remains constant. For example, when a ball is dropped, the energy is transferred from its gravitational potential energy (GPE) store to its kinetic energy (KE) store. The total energy (GPE + KE) remains the same, assuming no air resistance. Examiners will award marks for explicitly stating this law in relevant questions.
### Concept 2: Energy Stores
OCR requires you to know eight specific energy stores. Think of these as accounts where energy is held. To earn marks, you must name the specific store involved in a transfer.
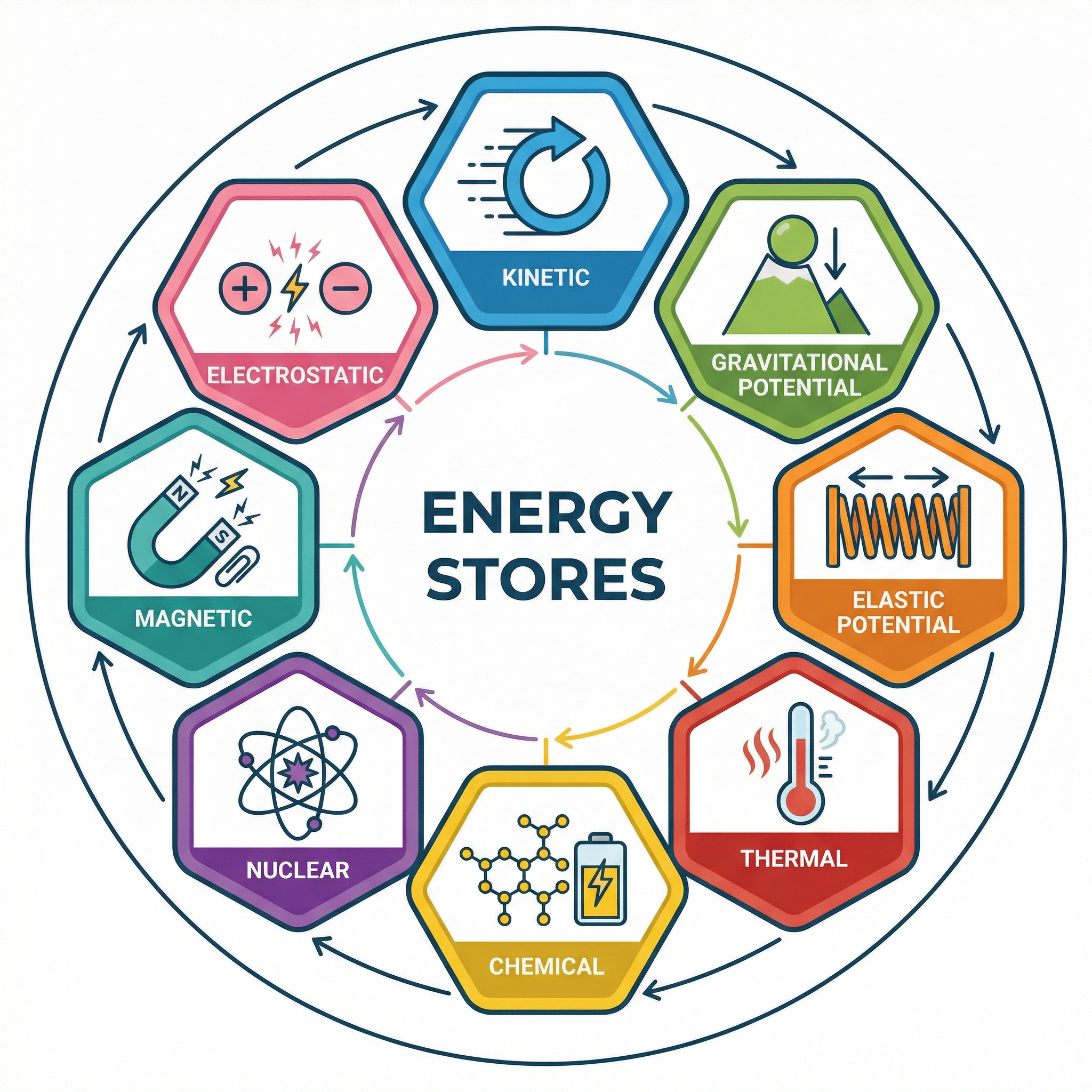
1. **Kinetic**: The energy of a moving object. The faster it moves or the more massive it is, the more energy is in its kinetic store.
2. **Gravitational Potential**: The energy an object has due to its position in a gravitational field. The higher it is, the more energy is in its GPE store.
3. **Elastic Potential**: The energy stored in a stretched or compressed object, like a spring or elastic band.
4. **Thermal**: The total kinetic and potential energy of the particles in an object. Hotter objects have more energy in their thermal store.
5. **Chemical**: The energy stored in the bonds between atoms, released during chemical reactions. Found in food, fuel, and batteries.
6. **Nuclear**: The energy stored in the nucleus of an atom, released during nuclear reactions (fission or fusion).
7. **Magnetic**: The energy stored when repelling poles are pushed together or attracting poles are pulled apart.
8. **Electrostatic**: The energy stored when repelling charges are moved closer or attracting charges are pulled apart.
### Concept 3: Energy Transfer Pathways
Energy moves from one store to another via one of four pathways. You must state the pathway to get full credit.
1. **Mechanically**: A force does work, causing energy transfer. (e.g., pushing a box, a moving car's engine turning the wheels).
2. **Electrically**: A charge does work, transferring energy. (e.g., current flowing from a battery to a bulb).
3. **By Heating**: Energy is transferred from a hotter object to a colder one. (e.g., a radiator heating a room).
4. **By Radiation**: Energy is transferred by waves, such as light or infrared. (e.g., the Sun heating the Earth).
**Example**: When a kettle boils water, energy is transferred **electrically** from the mains supply to the thermal store of the kettle's heating element. Then, energy is transferred **by heating** from the element to the thermal store of the water.
### Concept 4: Energy Dissipation and Efficiency
In any real-world energy transfer, some energy is always dissipated. This means it is transferred to a store that is not useful, typically the thermal store of the surroundings. **Crucially, this energy is NOT ‘lost’ or ‘used up’**. It is simply less useful. Using the term ‘dissipated’ will gain you credit.
**Efficiency** is a measure of how much of the input energy is transferred to a useful output store.
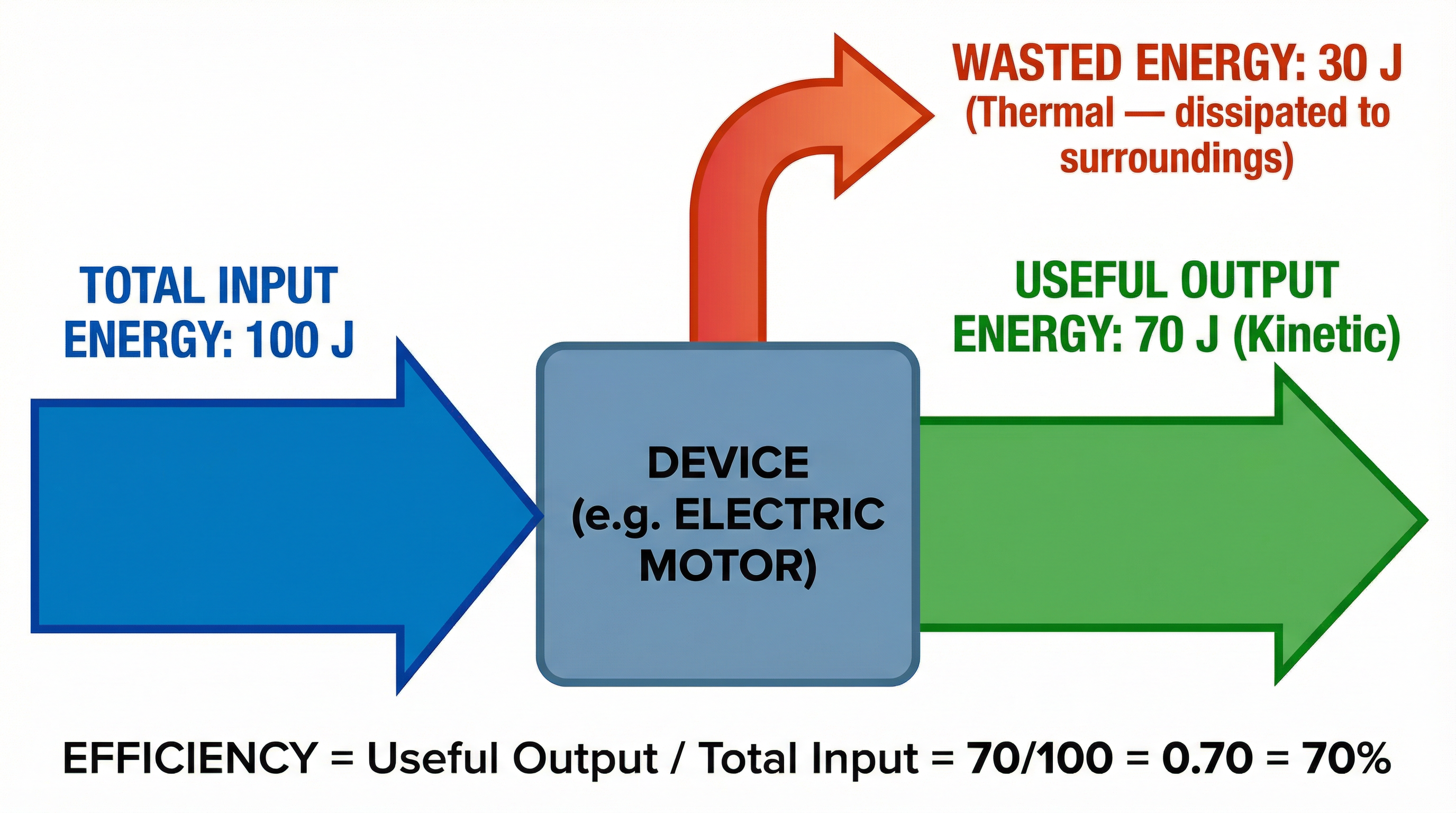
An inefficient device dissipates a lot of energy. For example, an old filament bulb dissipates most of its energy to the thermal store of the surroundings (it gets very hot) and only a small fraction to the light store. A modern LED is much more efficient, converting most of the input energy into light.
Methods to reduce unwanted energy transfers (increase efficiency) include:
- **Lubrication**: Reduces friction between moving parts, minimising energy transfer to the thermal store.
- **Thermal Insulation**: Using materials with low thermal conductivity to reduce the rate of energy transfer by heating.
### Concept 5: Power
Power is the **rate at which energy is transferred** or the **rate at which work is done**. Its unit is the Watt (W), where 1 Watt is equal to 1 Joule of energy transferred per second (1 W = 1 J/s). A 100W light bulb transfers 100 joules of energy every second.
### Concept 6: Thermal Properties of Materials
It is vital not to confuse these two properties:
- **Thermal Conductivity**: A measure of how quickly energy is transferred by conduction through a material. Metals have high thermal conductivity (they are good conductors), while materials like wool, foam, and air have low thermal conductivity (they are good insulators).
- **Specific Heat Capacity (SHC)**: The amount of energy needed to raise the temperature of 1 kg of a substance by 1°C. Water has a very high SHC (4200 J/kg°C), meaning it takes a lot of energy to heat it up, but it also means it can store a lot of energy in its thermal store. This is why it's used in central heating systems.
## Mathematical/Scientific Relationships
- **Power, Energy, Time** (Must memorise):
`Power (W) = Energy (J) / Time (s)`
`P = E / t`
- **Efficiency** (Must memorise):
`Efficiency = Useful Output Energy (J) / Total Input Energy (J)`
`Efficiency = Useful Output Power (W) / Total Input Power (W)`
(Efficiency has no units; it's a ratio or percentage)
- **Kinetic Energy** (Given on formula sheet):
`Kinetic Energy (J) = 0.5 × mass (kg) × (speed (m/s))^2`
`KE = ½mv²`
- **Gravitational Potential Energy** (Given on formula sheet):
`GPE (J) = mass (kg) × gravitational field strength (N/kg) × height (m)`
`GPE = mgh` (On Earth, g ≈ 9.8 N/kg)
- **Elastic Potential Energy** (Given on formula sheet):
`Elastic Potential Energy (J) = 0.5 × spring constant (N/m) × (extension (m))^2`
`EPE = ½ke²`
- **Change in Thermal Energy** (Given on formula sheet - **Higher Tier Only**):
`Change in Thermal Energy (J) = mass (kg) × specific heat capacity (J/kg°C) × temperature change (°C)`
`ΔE = mcΔθ`
## Required Practical: Investigating Thermal Insulators
This is a common AO3 practical-based question.
**Aim**: To investigate the effectiveness of different materials as thermal insulators.
**Apparatus**: Kettle, beakers, thermometer, stopwatch, lids for beakers, various insulating materials (e.g., bubble wrap, cotton wool, foil).
**Method**:
1. Boil water in a kettle. This is a safety precaution to ensure the same starting temperature for each test.
2. Place the same volume of hot water into four identical beakers.
3. Measure the starting temperature of the water in each beaker. It should be the same.
4. Leave one beaker uninsulated. This will act as the **control**.
5. Wrap the other three beakers in different insulating materials (the **independent variable**).
6. Place lids on all beakers to reduce energy transfer from the water surface.
7. Start a stopwatch and record the temperature of the water in each beaker every 3 minutes for 15 minutes.
8. The **dependent variable** is the temperature drop over time.
**Expected Results**: The beaker that cools down the slowest is wrapped in the most effective insulator. The uninsulated beaker will cool down the fastest.
**Common Errors & How to Avoid Them**:
- **Inconsistent starting temperature**: Use water from the same freshly boiled kettle for all beakers.
- **Varying water volume**: Use a measuring cylinder to ensure the volume is identical in each beaker.
- **Reading the thermometer incorrectly**: Ensure your eye is level with the top of the liquid to avoid parallax error.
**How Examiners Test It**: You could be asked to write a method, identify variables, analyse a table of results, plot a graph of temperature against time, or evaluate the conclusion.