Cracking — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
This guide breaks down the crucial industrial process of cracking for your OCR GCSE Chemistry exam. Discover how we turn less useful long-chain hydrocarbons into high-demand fuels and plastics, and master the equations and practical tests that will earn you top marks."

## Overview
Cracking is one of the most important chemical processes in the modern world, and a key topic in OCR GCSE Chemistry (8.4). It addresses a fundamental economic problem: the fractions obtained from the fractional distillation of crude oil don't match market demand. We get a surplus of long-chain hydrocarbons (like bitumen and heavy oils) but a shortage of more valuable short-chain hydrocarbons (like petrol and the alkenes needed for plastics). Cracking solves this by breaking down the large, less useful molecules into smaller, more valuable ones. In your exam, you'll be tested on the conditions, the products, the equations, and the chemical test for its products. Expect questions that ask you to 'Explain' why cracking is necessary, 'Describe' the process, and 'Balance' the chemical equations. Mastering this topic is essential for understanding the link between the oil industry and the production of everyday materials.

## Key Concepts
### Concept 1: Thermal Decomposition
Cracking is fundamentally a process of **thermal decomposition**. This is a key phrase that examiners look for. 'Thermal' means we use heat, and 'decomposition' means we are breaking a compound down into simpler substances. In this case, we are breaking down large hydrocarbon molecules. It is a **chemical change**, not a physical one. This is a critical distinction. In fractional distillation (a physical process), intermolecular forces between molecules are overcome. In cracking (a chemical process), strong **covalent bonds** *within* the molecules themselves are broken. This requires a significant amount of energy, which is why high temperatures are needed.
**Example**: A C₁₅H₃₂ molecule is heated to a high temperature. The energy causes the C-C bonds to vibrate so violently that they snap, forming smaller, more stable molecules like a C₈H₁₈ molecule and a C₇H₁₄ molecule.
### Concept 2: Conditions for Cracking
To earn full marks, you must state the two key conditions for catalytic cracking precisely:
1. **High Temperature**: Typically in the range of 400-700°C.
2. **A Catalyst**: This speeds up the reaction, allowing it to happen at a lower temperature than it otherwise would and saving energy. For your exam, you should name a specific catalyst. The most common examples are **aluminium oxide (Al₂O₃)** and **silica (SiO₂) **. In a lab setting, a **porous pot** can be used as the catalyst.
It's vital to be specific. Simply writing 'heat' or 'a catalyst' may not be enough for the marks.
### Concept 3: Products - Alkanes and Alkenes
Cracking breaks a long-chain alkane into a shorter-chain alkane and at least one **alkene**.
* **Alkanes** (like octane, C₈H₁₈) are desirable as they are the main components of petrol.
* **Alkenes** (like ethene, C₂H₄) are even more valuable. They are the essential feedstock for the polymer industry, used to make plastics like poly(ethene) and poly(propene). Alkenes are 'unsaturated' because they contain a C=C double bond. This double bond is the site of their reactivity.
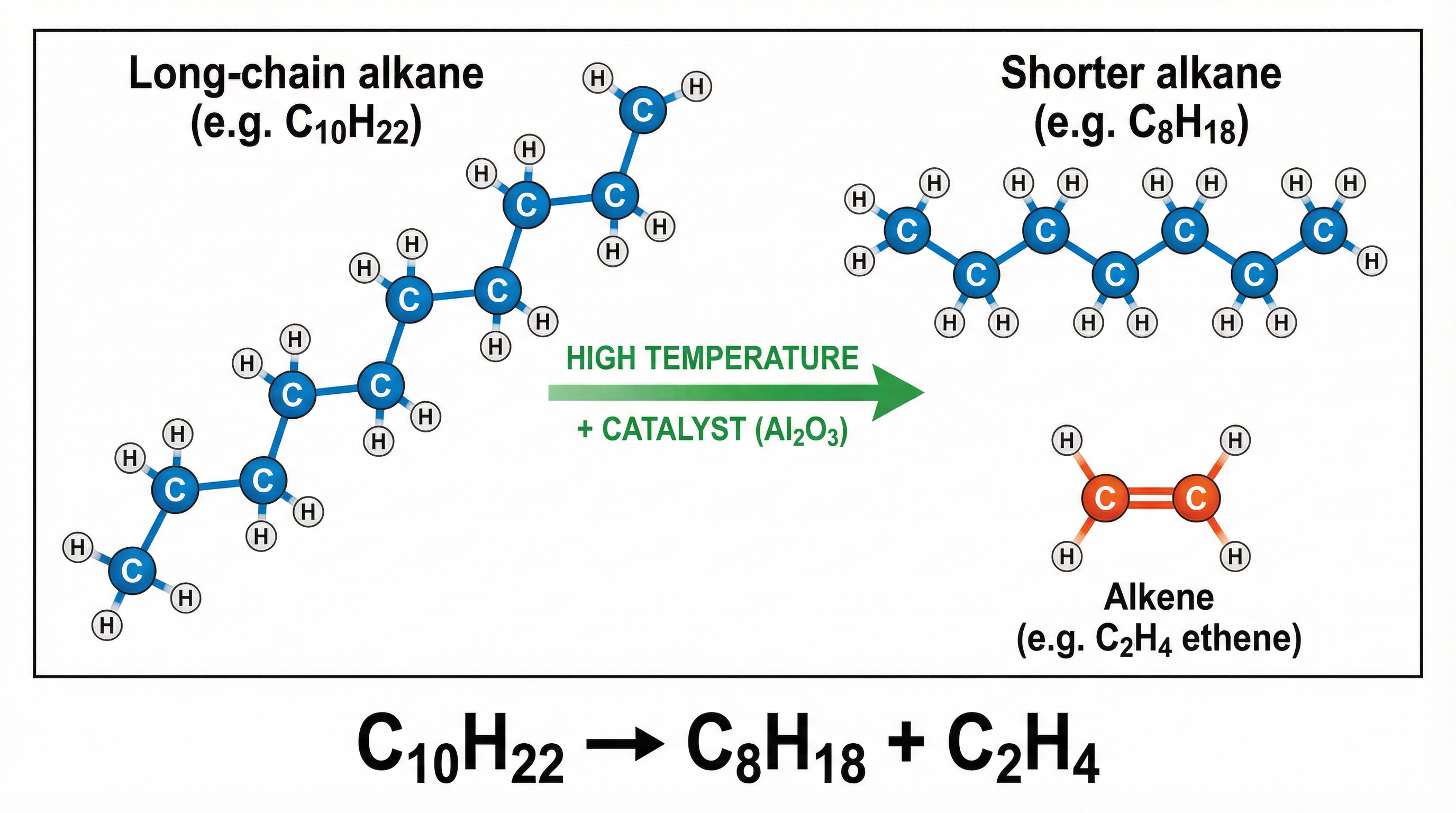
## Mathematical/Scientific Relationships
The core scientific relationship in cracking is the **conservation of mass**, which is applied when balancing symbol equations. The number of carbon and hydrogen atoms must be identical on both the reactant and product sides of the equation.
**General Equation:**
Long-chain Alkane → Shorter-chain Alkane + Alkene(s)
**Example Equation:**
Decane cracking to form octane and ethene:
C₁₀H₂₂ → C₈H₁₈ + C₂H₄
* **Reactant side**: 10 Carbon atoms, 22 Hydrogen atoms
* **Product side**: (8 + 2) = 10 Carbon atoms, (18 + 4) = 22 Hydrogen atoms
The equation is balanced.
## Practical Applications
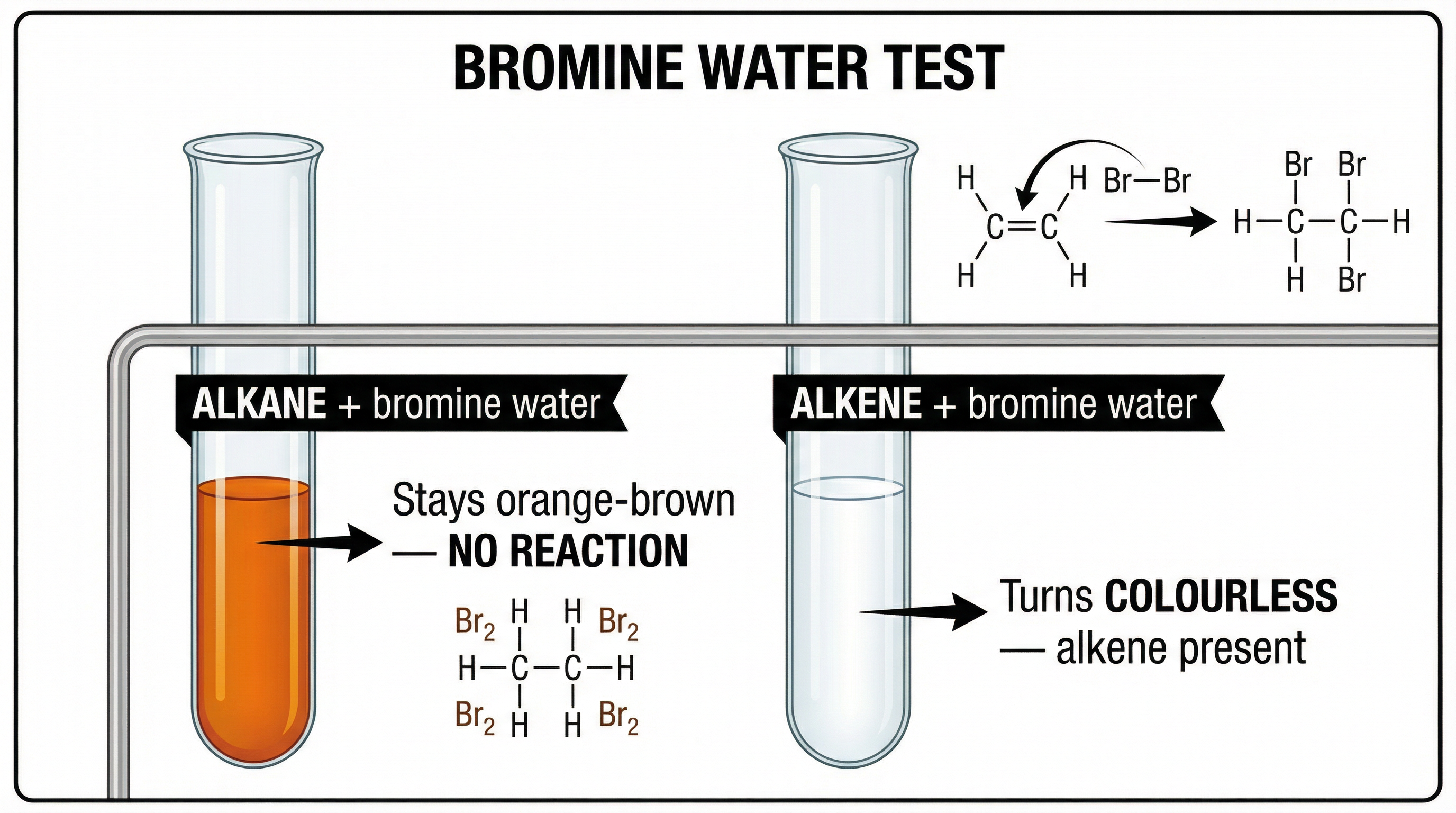
### Required Practical: The Bromine Water Test for Unsaturation
This is the standard laboratory test to distinguish between an alkane and an alkene. Alkenes are unsaturated due to their C=C double bond, and they undergo an **addition reaction** with bromine water.
* **Apparatus**: Test tubes, bromine water, samples of an alkane and an alkene.
* **Method**:
1. Add a few drops of bromine water (which is orange-brown) to a test tube containing an alkane (e.g., hexane).
2. Shake the mixture. Observe any change.
3. Repeat the process with a test tube containing an alkene (e.g., hexene).
* **Expected Results**:
* With the **alkane**, the solution remains **orange-brown**. No reaction occurs.
* With the **alkene**, the solution turns from **orange-brown to colourless**. This is a positive test for an alkene.
* **Examiner Tip**: It is crucial to use the word **colourless**, not 'clear'. Bromine water is already clear (transparent). The observation is a loss of colour.
"