States of Matter and Changes of State — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
Master the fundamental principles of States of Matter for your OCR GCSE Chemistry exam. This guide breaks down the particle model, changes of state, and energy transfers, providing you with the essential knowledge to secure top marks.
## Overview
Welcome to the essential guide for OCR GCSE Chemistry Topic 1.3: States of Matter and Changes of State. This topic forms the bedrock of your understanding of the physical world, explaining how and why substances exist as solids, liquids, or gases, and the energy changes involved in transitioning between them. While the concepts may seem familiar, the exam demands a precise application of the particle model to explain macroscopic properties. Examiners will test your ability to describe the arrangement, movement, and energy of particles in each state, and critically, to explain what happens during changes of state like melting and boiling. A strong grasp of this topic is vital as it provides the foundation for understanding chemical bonding, energy changes in reactions (exothermic and endothermic), and rates of reaction. Expect to see a mix of short-answer questions asking for definitions and descriptions, as well as longer, 6-mark 'Level of Response' questions that require you to compare the properties of all three states in detail.

## Key Concepts
### Concept 1: The Particle Model
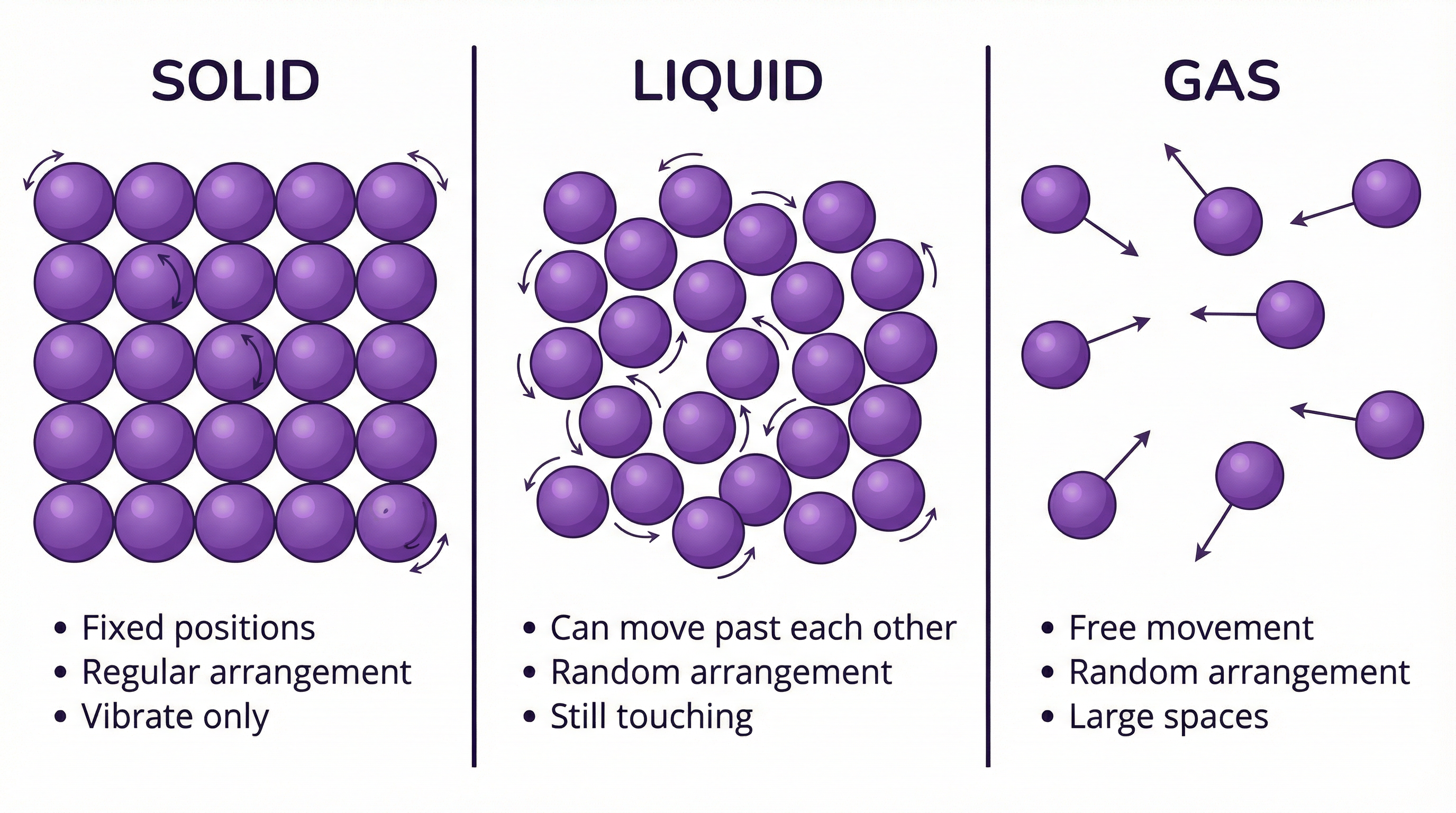
The particle model is a cornerstone of chemistry, used to explain the properties of substances in different states. It assumes that all matter is made up of tiny, constantly moving particles. The state of a substance is determined by the energy of these particles and the forces acting between them.
- **Solids**: Particles are packed closely together in a fixed, regular arrangement called a lattice. They possess the least amount of kinetic energy and can only vibrate about their fixed positions. The forces of attraction between particles are very strong, which is why solids have a definite shape and volume.
- **Liquids**: Particles are still in close contact but are arranged randomly. They have more kinetic energy than solids, enough to overcome the rigid lattice structure and move past one another. The forces of attraction are weaker than in solids but still strong enough to keep the particles together, giving liquids a definite volume but an indefinite shape (they take the shape of their container).
- **Gases**: Particles have the highest kinetic energy and are far apart from each other, with negligible forces of attraction between them. They move rapidly and randomly in all directions, which is why gases have no definite shape or volume and will fill any container they are in. Because of the large spaces between particles, gases can be easily compressed.
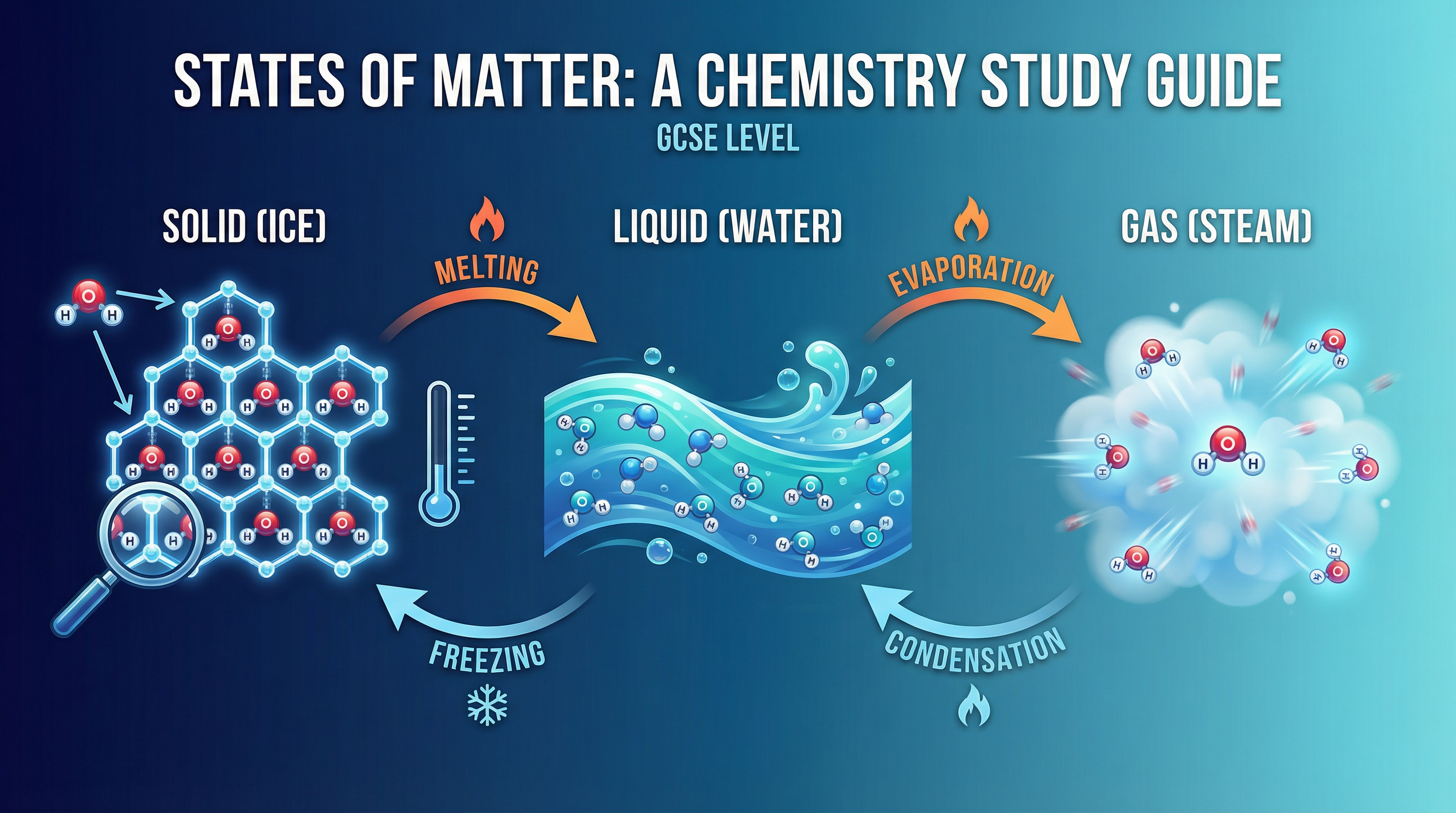
### Concept 2: Changes of State & Energy
Changes of state are physical changes because no new substances are formed. They involve energy being transferred to or from a substance, which alters the internal energy of the particles.
- **Melting (Solid to Liquid)**: When a solid is heated, its particles gain kinetic energy and vibrate more vigorously. At the melting point, the particles have gained enough energy to overcome the forces holding them in their fixed positions. The energy supplied at this point is used to break these forces, not to increase the temperature. This is known as **latent heat of fusion**.
- **Boiling (Liquid to Gas)**: As a liquid is heated, its particles gain more kinetic energy and move faster. At the boiling point, the particles have enough energy to overcome all the forces of attraction holding them together. The energy supplied is used to separate the particles completely, turning the liquid into a gas. This is known as **latent heat of vaporization**. Boiling occurs throughout the liquid.
- **Freezing (Liquid to Solid)**: When a liquid cools, particles lose kinetic energy and move more slowly. At the freezing point, the forces of attraction become strong enough to hold the particles in fixed positions, forming a solid lattice. Energy is released during this process.
- **Condensation (Gas to Liquid)**: As a gas cools, particles lose kinetic energy. The forces of attraction start to have an effect, pulling the particles closer together to form a liquid. Energy is released.
- **Sublimation**: A rare change of state where a solid turns directly into a gas without passing through the liquid phase (e.g., solid carbon dioxide, or 'dry ice').
### Concept 3: Heating Curves
A heating curve is a graph that shows how the temperature of a substance changes as it is heated over time. These graphs provide crucial evidence for the energy changes during a change of state.
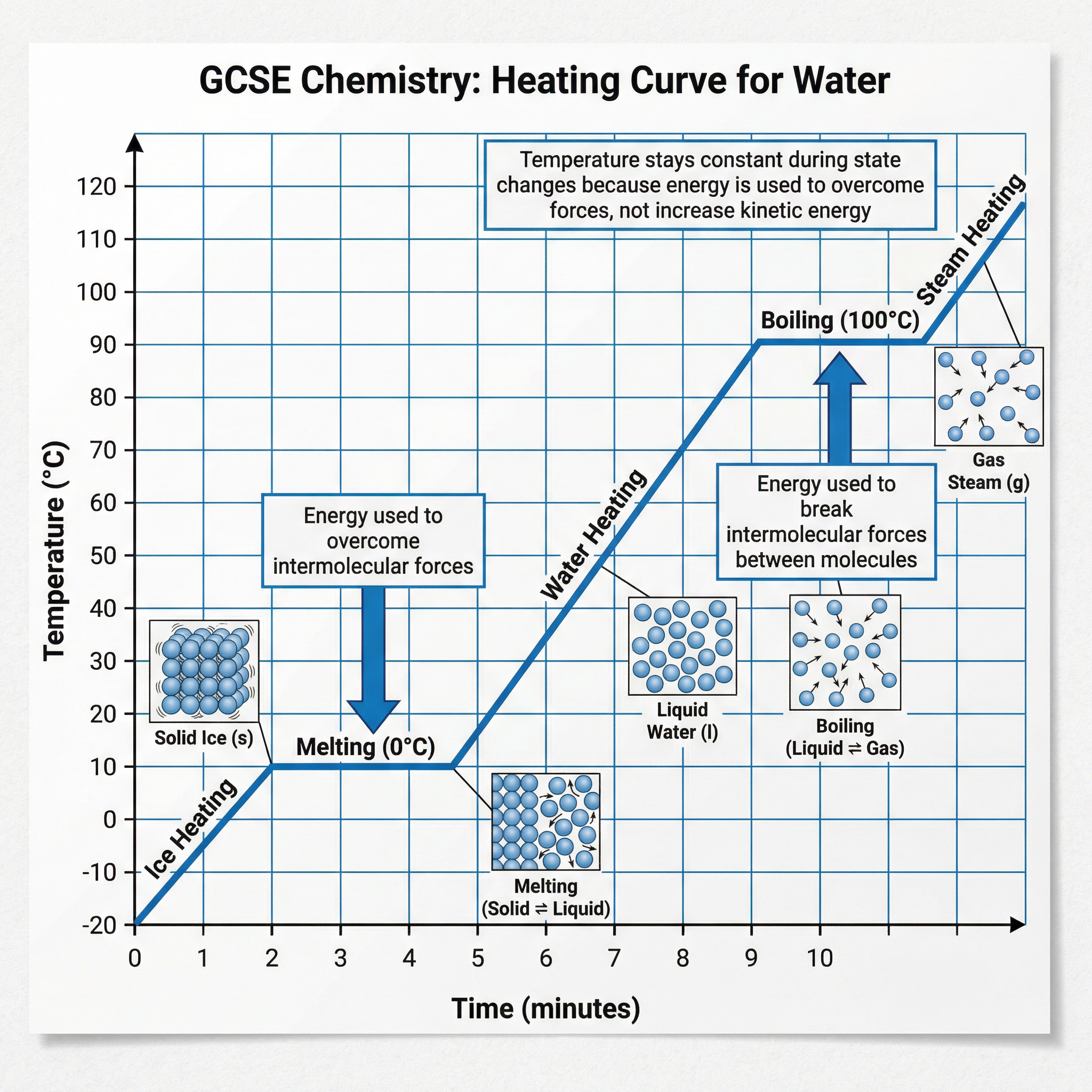
Key features of a heating curve:
1. **Sloping Sections**: The temperature of the substance is increasing. The heat energy supplied is increasing the kinetic energy of the particles.
2. **Flat Sections (Plateaus)**: The temperature remains constant, even though heat is still being supplied. This occurs during a change of state (melting or boiling). The energy is being used to overcome the intermolecular forces between particles, increasing their potential energy, not their kinetic energy. This is why the temperature doesn't rise.
## Mathematical/Scientific Relationships
There are no complex formulas to memorise for this topic at GCSE level. The key relationship to understand is conceptual:
**Energy Input → Increased Particle Kinetic Energy (Temperature Rise) OR Overcoming Intermolecular Forces (Change of State at Constant Temperature)**
Another key principle is the **Conservation of Mass**. During a physical change like melting or boiling, the mass of the substance does not change. This is because the number of particles remains the same; they are simply rearranged.
## Practical Applications
Understanding states of matter is crucial in many real-world applications:
- **Distillation**: Used to separate liquids with different boiling points (e.g., separating ethanol from water, or fractional distillation of crude oil). This relies on the principle that substances turn into gases at specific boiling points.
- **Refrigeration and Air Conditioning**: These technologies work by exploiting the energy changes during the evaporation and condensation of a coolant fluid. As the fluid evaporates, it absorbs heat from the surroundings (cooling the fridge), and as it condenses, it releases that heat outside.
- **Pressure Cookers**: By increasing the pressure, the boiling point of water is raised above 100°C. This allows food to cook faster at a higher temperature.
- **Required Practical**: While there isn't a specific named required practical for this topic in OCR, skills related to measuring temperature changes and plotting graphs are essential. You could be asked to interpret data from an experiment where a substance like stearic acid is melted and then allowed to cool, plotting a cooling curve to determine its melting/freezing point.