The effect of heat on food — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
This guide provides a comprehensive, exam-focused breakdown of how heat affects food, a core topic in the OCR GCSE Food Preparation and Nutrition specification. It covers the precise scientific principles of gelatinisation, coagulation, caramelisation, and dextrinisation, equipping candidates with the detailed knowledge required to secure top marks."

## Overview
This study guide delves into the critical chemical and functional changes that occur when heat is applied to food, a fundamental area of the OCR J309 specification. Examiners expect candidates to move beyond simple descriptions of cooking and demonstrate a precise understanding of the underlying molecular transformations. This includes using accurate scientific terminology, quantifying critical temperatures, and explaining the functional impact of these changes on the final food product. This guide will equip you with the detailed knowledge of protein denaturation, starch gelatinisation, caramelisation, and dextrinisation needed to answer exam questions with confidence and precision. Marks are awarded for linking physical changes, such as texture and viscosity, to the molecular mechanisms that cause them.

## Key Processes & Developments
### Starch Gelatinisation
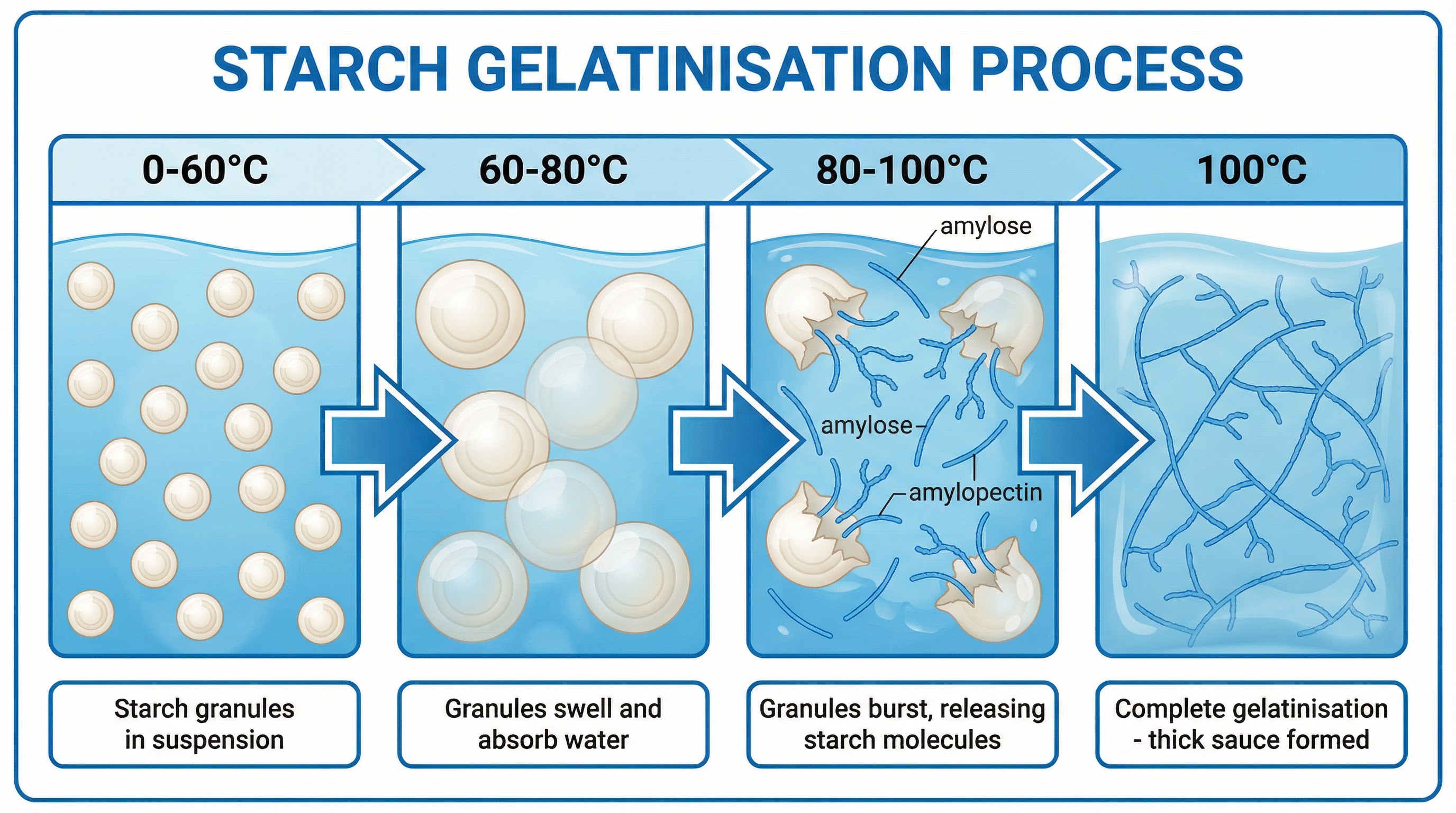
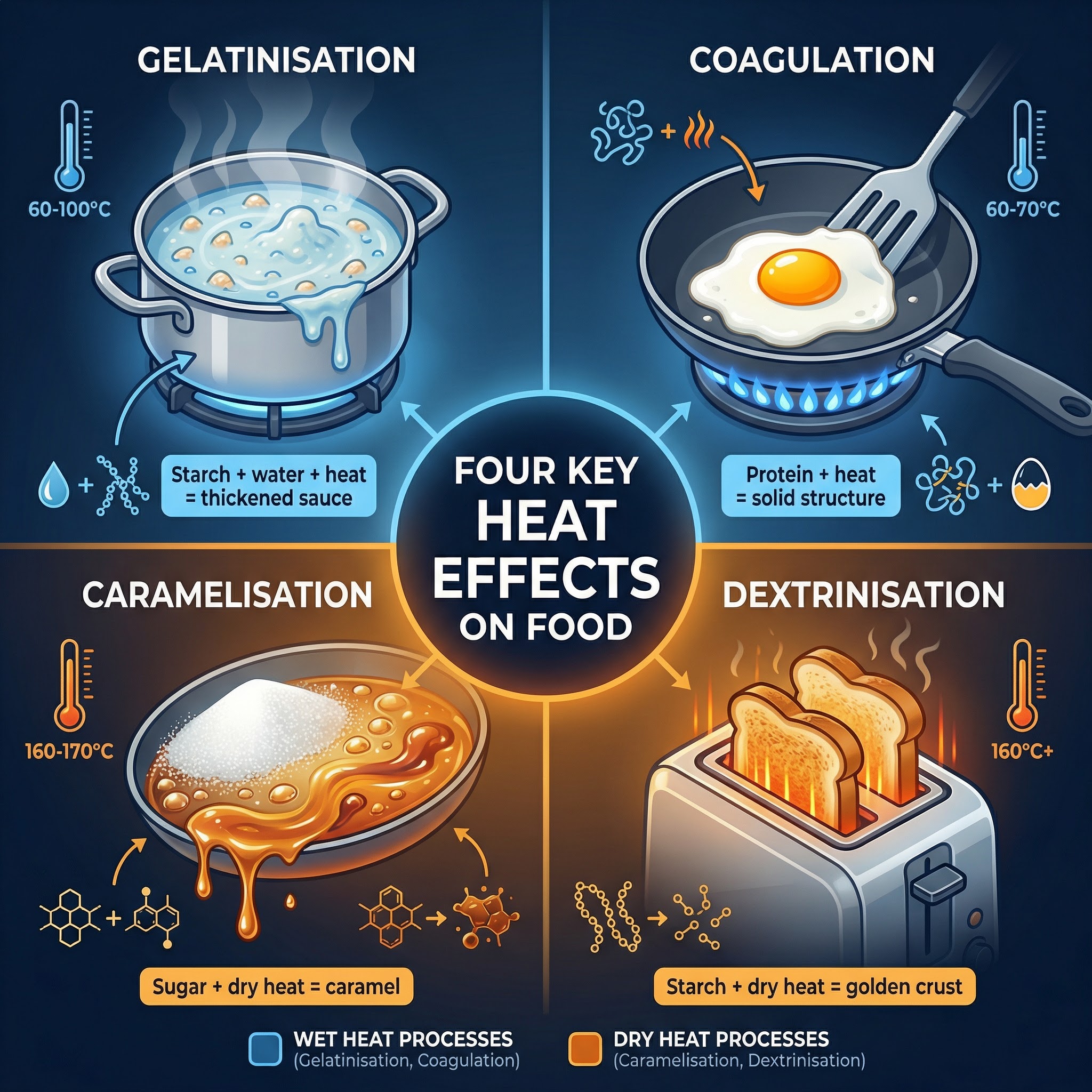
**What happened**: When starch is heated in the presence of a liquid, the granules absorb water and swell. At 60°C, they begin to swell and soften. At 80°C, the granules burst, releasing long-chain polysaccharide molecules (amylose and amylopectin) into the surrounding liquid. By 100°C, these molecules have formed a complex network that traps water, causing the liquid to thicken and form a gel.
**Why it matters**: This process is fundamental to the creation of sauces, gravies, and custards. Candidates must be able to explain this process chronologically, using the correct temperatures, to gain full marks. Understanding gelatinisation is also key to explaining common food preparation defects, such as lumpy sauces, which occur due to insufficient agitation during heating.
**Specific Knowledge**: Starch granules, amylose, amylopectin, suspension, swelling (60°C), bursting (80°C), gelatinisation (100°C).
### Protein Denaturation and Coagulation
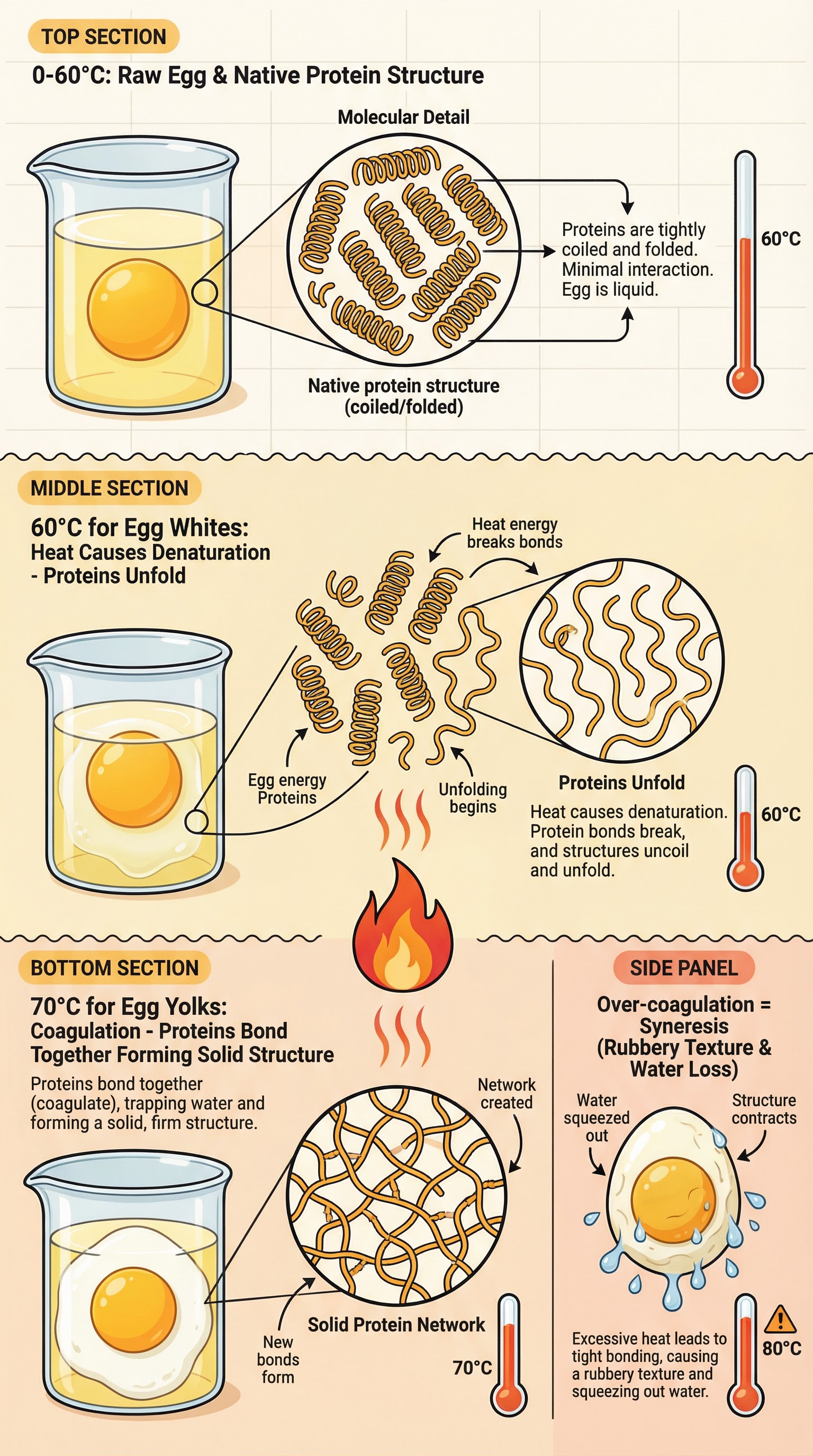
**What happened**: Heat causes protein molecules, which are normally in a tightly coiled, folded (native) state, to unravel and change shape. This irreversible process is called denaturation. As heating continues, the denatured, unfolded protein chains bond with each other, forming a solid, three-dimensional network. This is coagulation.
**Why it matters**: This process explains how liquid foods like eggs become solid when cooked, how meat changes texture, and how cheese is made. Candidates must distinguish between denaturation (the unfolding) and coagulation (the bonding and solidifying). Over-coagulation leads to syneresis, where the protein network becomes too tight and squeezes out water, resulting in a rubbery texture. This is a common defect in egg dishes and is frequently tested.
**Specific Knowledge**: Native protein, denaturation, coagulation, syneresis. Egg whites coagulate at 60°C, egg yolks at 70°C.
### Caramelisation
**What happened**: This is the process of heating sugar (a carbohydrate) in the absence of water (dry heat). At high temperatures (160°C - 170°C for sucrose), the sugar crystals melt and undergo a series of complex chemical reactions, breaking down and reforming into hundreds of new compounds that create a characteristic brown colour and rich, caramel flavour.
**Why it matters**: Caramelisation is responsible for the flavour and colour of many popular desserts and confectionery items. A common mistake is confusing it with dextrinisation. Candidates must be clear that caramelisation involves sugar and dry heat.
**Specific Knowledge**: Sucrose, dry heat, caramel, critical temperature (160°C - 170°C).
### Dextrinisation
**What happened**: When starchy foods like bread or cereals are subjected to dry heat, the long-chain starch molecules (polysaccharides) on the surface break down into smaller sugar units called dextrins. This process causes the food to turn a golden-brown colour and develop a characteristic toasty flavour and crisp texture.
**Why it matters**: Dextrinisation is responsible for the browning of toast, the crust on bread, and the crispness of baked goods. Candidates must be able to differentiate this process from caramelisation and gelatinisation, specifying that it involves starch and dry heat.
**Specific Knowledge**: Starch, polysaccharide, dextrin, dry heat, browning.
## Second-Order Concepts
### Causation
- **Heat Energy**: The primary cause of all these changes is the application of heat energy, which increases molecular vibration, breaking bonds and causing new ones to form.
- **Presence of Water**: The presence or absence of water is a critical causal factor. Gelatinisation requires water (wet heat), while caramelisation and dextrinisation require its absence (dry heat).
### Consequence
- **Textural Changes**: Heat leads to significant changes in texture, such as thickening (gelatinisation), solidification (coagulation), and crisping (dextrinisation).
- **Sensory Changes**: Heat creates new flavours and colours (caramelisation, dextrinisation, Maillard reaction), which generally improve the sensory appeal of food.
- **Nutritional Changes**: Heat can cause the loss of some heat-sensitive vitamins (like Vitamin C) but can also make some nutrients more bioavailable.
### Change & Continuity
- **Change**: The molecular structure of proteins, starches, and sugars is permanently changed by heat. These are chemical changes, not just physical ones.
- **Continuity**: The base elements (carbon, hydrogen, oxygen) remain, but they are rearranged into new molecules with different properties.
### Significance
- **Food Safety**: Heating food to specific temperatures denatures the enzymes in bacteria, killing them and making food safe to eat.
- **Food Production**: These processes are the foundation of almost all cooking and food manufacturing, from baking bread to making cheese and sauces."