
Overview
Welcome to the definitive guide for OCR GCSE Physics Topic 7.1: Atomic Structure. This topic traces the incredible scientific journey that unveiled the secrets of the atom, from a simple solid sphere to the complex nuclear model we use today. Understanding this story is crucial, as examiners frequently award marks for linking experimental evidence to the development of scientific theories. This guide will equip you with the knowledge of subatomic particles (protons, neutrons, and electrons), the concept of isotopes, and the all-important alpha scattering experiment. We will also cover the mathematical skills needed to compare the scale of the atom and its nucleus, a common Higher Tier question. By mastering these concepts, you will be able to confidently tackle a wide range of questions, from short definitions to extended 6-mark responses, and understand how this topic provides the foundation for nuclear physics (Topic 7.2).

Key Concepts
The Evolving Atomic Model
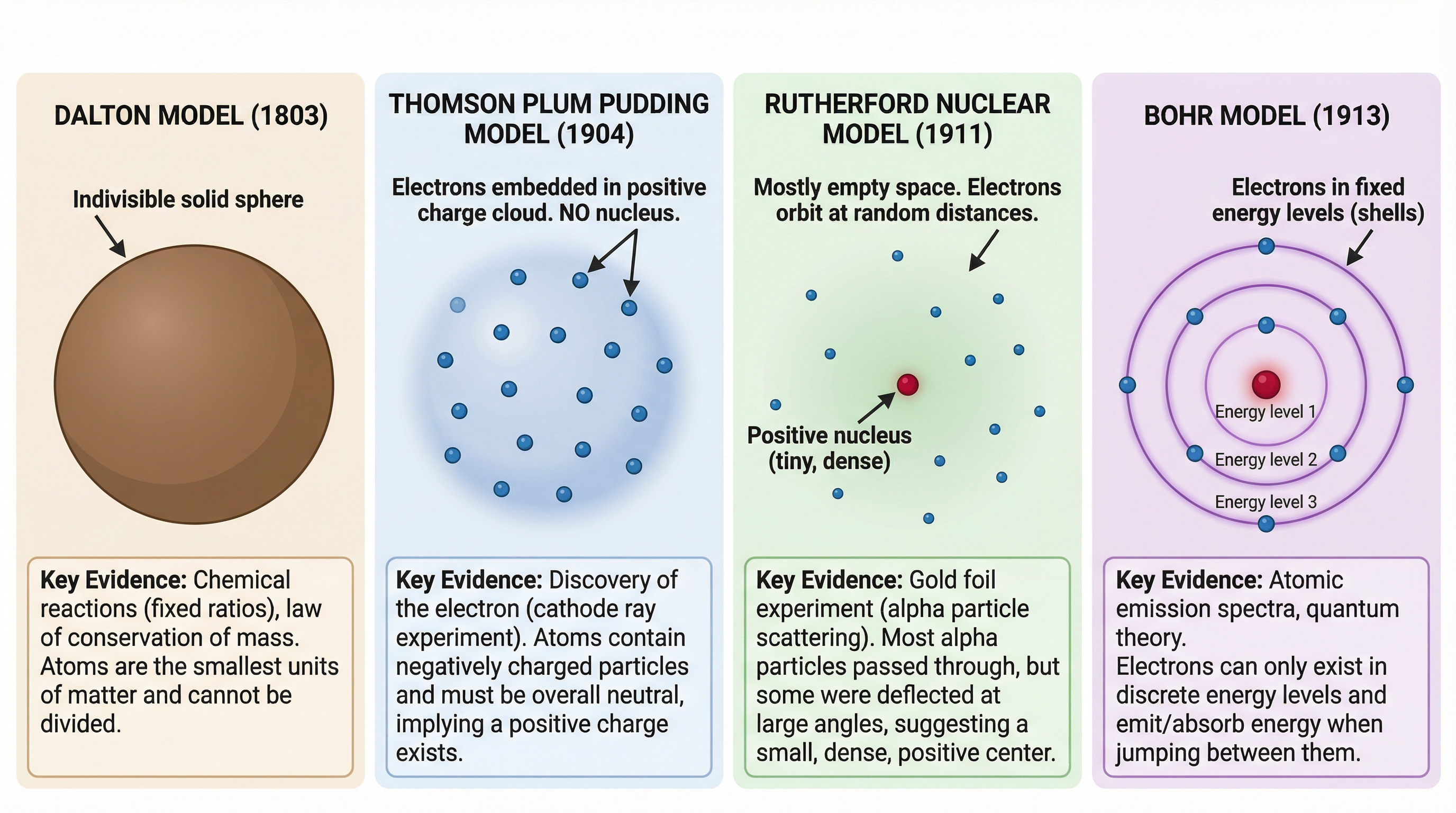
Our understanding of the atom has changed dramatically over time. It is not just a collection of facts; it is a story of discovery. For the exam, you must know the chronological order and the key evidence for each model.
-
Dalton Model (1803): John Dalton proposed that atoms were tiny, indivisible solid spheres. He imagined them as microscopic billiard balls. This was the first truly scientific model, but it couldn't explain the existence of subatomic particles.
-
Thomson's Plum Pudding Model (1904): After discovering the electron in 1897, J.J. Thomson suggested the atom was a sphere of positive charge with negatively charged electrons embedded within it. Crucially, there is no nucleus in this model. Candidates often lose marks by forgetting this. The positive charge is diffuse and spread throughout the entire atom.
-
Rutherford's Nuclear Model (1911): This model was born from the groundbreaking alpha particle scattering experiment. The results showed that the atom must be mostly empty space, with a tiny, dense, positively charged nucleus at its centre where almost all the mass is concentrated. The electrons were thought to orbit this nucleus.
-
Bohr Model (1913): Niels Bohr refined Rutherford's model by proposing that electrons orbit the nucleus at specific, fixed distances in energy levels or 'shells'. This explained why atoms emit light at specific frequencies.
The Alpha Scattering Experiment
This is one of the most important experiments in the specification. You must be able to describe it and, most importantly, link the observations to the conclusions.
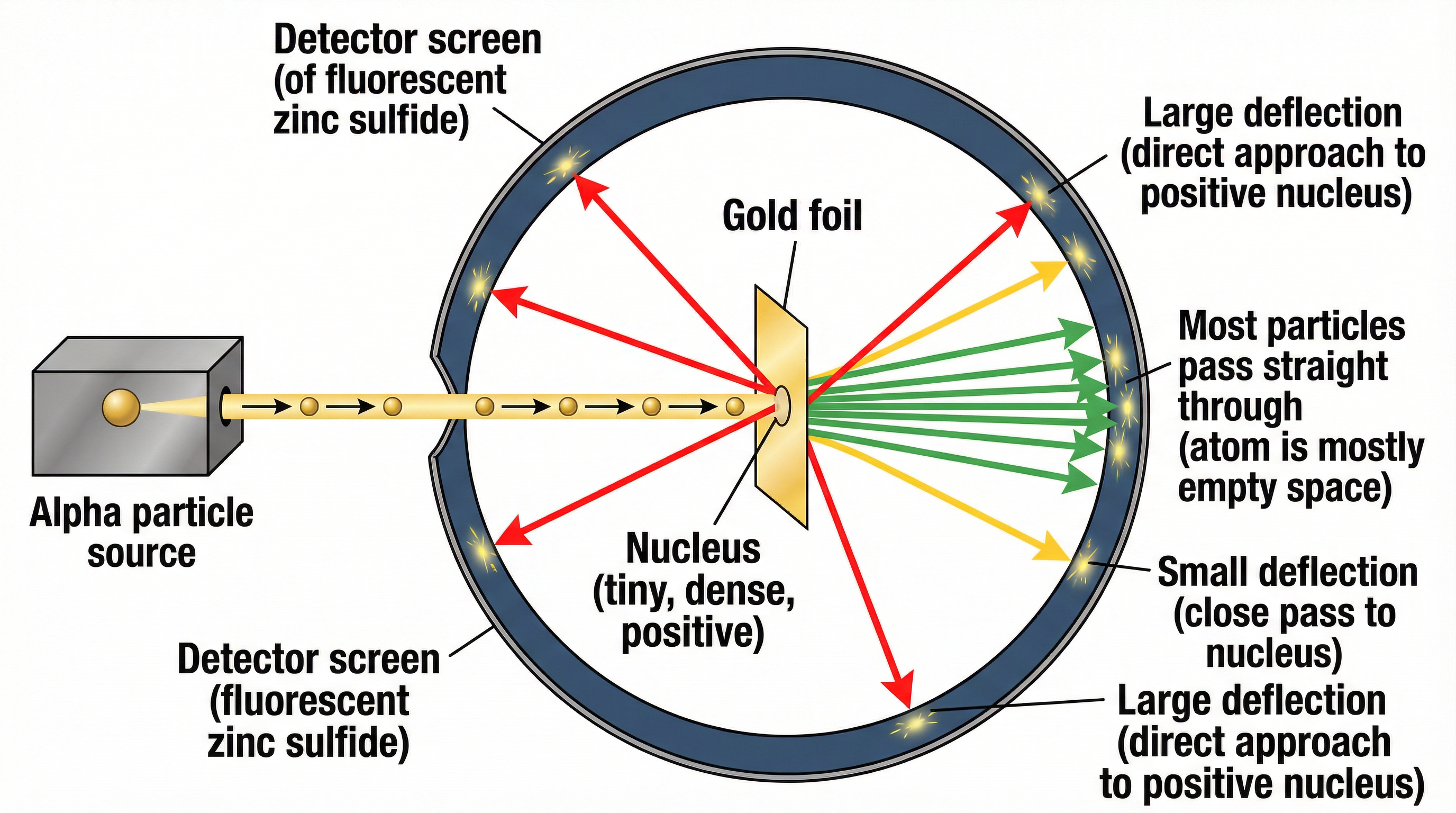
- Setup: A beam of positively charged alpha particles was fired at a very thin sheet of gold foil.
- Observation 1: Most alpha particles passed straight through the foil undeflected.
- Conclusion 1: This means the atom is mostly empty space. (1 mark)
- Observation 2: A small number of alpha particles were deflected by small angles.
- Conclusion 2: This is because they were repelled by a concentrated positive charge. (1 mark)
- Observation 3: A very small number (about 1 in 8000) were deflected by large angles (>90°), essentially bouncing back.
- Conclusion 3: This indicates that the positive charge and most of the atom's mass are concentrated in a tiny, dense nucleus at the centre. (1 mark) Examiner Tip: Use the term electrostatic repulsion to explain the deflection; do not say the particles 'hit' or 'collided with' the nucleus.
Subatomic Particles and Isotopes
Atoms are built from three fundamental particles. You must know their relative mass and charge.
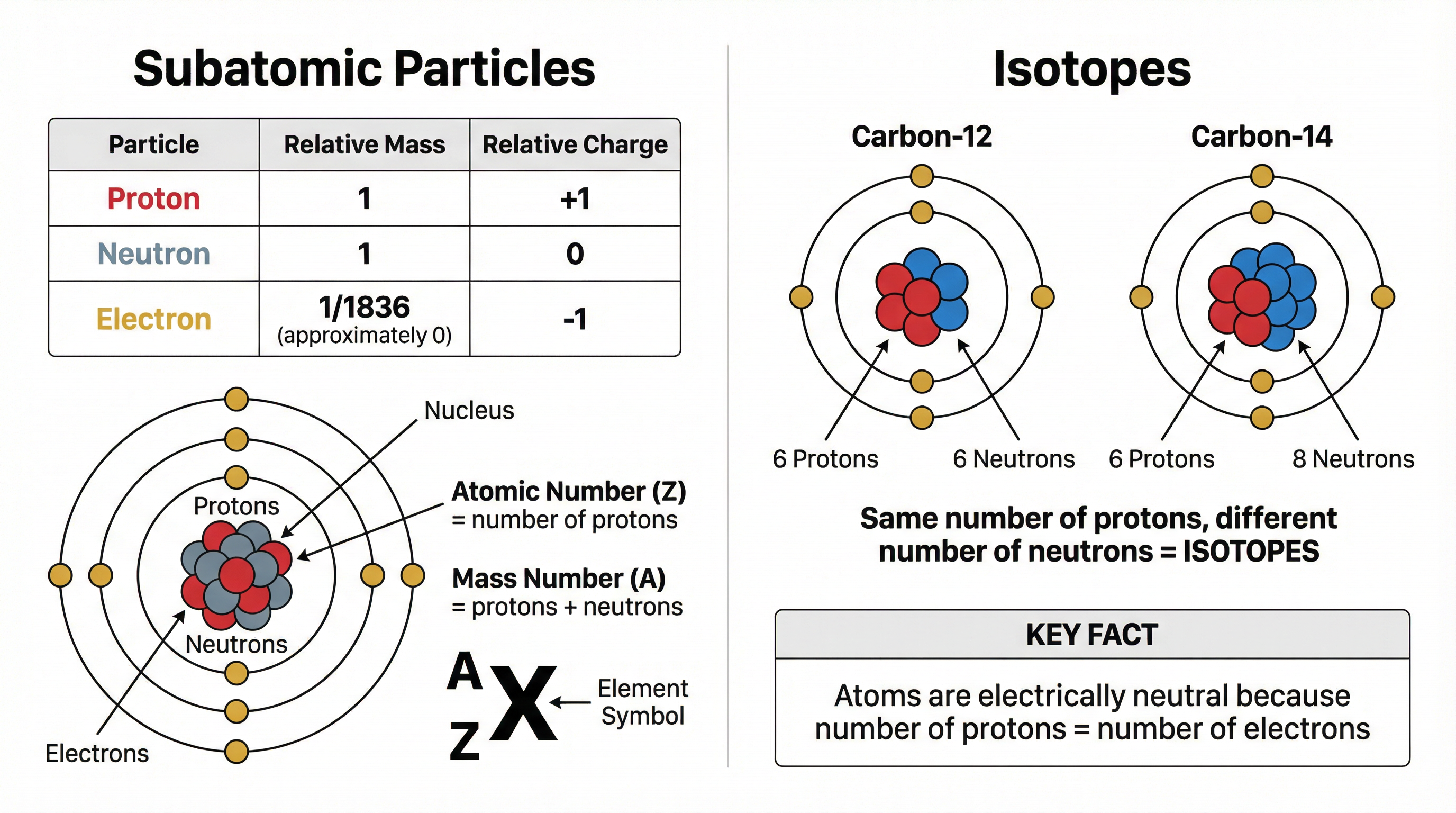
| Particle | Relative Mass | Relative Charge |
|---|
| Proton | 1 | +1 |
| Neutron | 1 | 0 |
| Electron | ~0 (1/1836) | -1 |
- Atomic Number (Z): The number of protons in the nucleus. This defines the element.
- Mass Number (A): The total number of protons and neutrons in the nucleus.
- Isotopes: Atoms of the same element with the same number of protons but a different number of neutrons. For example, Carbon-12 (6 protons, 6 neutrons) and Carbon-14 (6 protons, 8 neutrons) are isotopes of carbon. They have the same chemical properties but different masses.
Mathematical/Scientific Relationships
Calculating Subatomic Particles
- Number of Protons = Atomic Number (Z)
- Number of Electrons = Number of Protons (in a neutral atom)
- Number of Neutrons = Mass Number (A) – Atomic Number (Z) (Must memorise)
Example: For an atom of Lithium-7 (⁷₃Li), we have:
- Protons = 3
- Electrons = 3
- Neutrons = 7 - 3 = 4
Atomic Scale (Higher Tier)
Examiners expect you to appreciate the immense emptiness of the atom. You need to be able to compare the sizes using standard form.
- Diameter of an atom ≈ 1 x 10⁻¹⁰ m (Must memorise)
- Diameter of a nucleus ≈ 1 x 10⁻¹⁴ m **(Must memorise)**To find how many times larger the atom is than the nucleus, you calculate the ratio:
Diameter of atom / Diameter of nucleus = (1 x 10^-10) / (1 x 10^-14) = 10^4 = 10,000
The atom is about 10,000 times wider than its nucleus. This is a common calculation question.
Practical Applications
While there isn't a specific required practical for this topic, the principles are fundamental to many applications:
- Medical Imaging (PET Scans): Uses isotopes that emit positrons (a type of radiation) to create images of the body's metabolic activity.
- Carbon Dating: The isotope Carbon-14 is unstable and decays over time. By measuring the amount of Carbon-14 remaining in organic materials, we can determine their age.
- Nuclear Power: The structure of the nucleus, particularly in heavy elements like Uranium, is key to understanding nuclear fission, which releases vast amounts of energy in nuclear power stations.



