
Overview
Background radiation is the constant, low-level ionising radiation that is present in our environment at all times. For your OCR GCSE Physics exam, understanding this topic is crucial as it appears frequently and tests both your knowledge of specific sources (AO1) and your ability to handle data (AO2). This guide will equip you with a precise understanding of where this radiation comes from, how we measure it accurately, and the key terminology that examiners expect to see. We will explore the ubiquitous nature of radiation, linking it to the random process of radioactive decay and the critical concept of radiation dose, measured in Sieverts. Typical exam questions involve identifying sources, comparing their relative contributions, and performing a vital calculation to correct for background radiation in experimental measurements. Mastering this topic provides a strong foundation for understanding radiation safety and its applications.
Key Concepts
Concept 1: Sources of Background Radiation
Background radiation originates from a variety of sources that can be categorised as either natural or man-made (artificial). It is essential that candidates can recall specific examples from each category and their relative contributions to the average UK dose.
Natural Sources: These make up the vast majority (around 85%) of the background radiation we receive.
- Radon Gas (~51%): This is the single largest contributor. Radon is a radioactive gas produced by the decay of uranium in rocks and soil, particularly granite. Being a gas, it can seep into buildings and be inhaled, where its alpha particles can damage lung tissue. This is why its contribution is so significant and why homes in granite-rich areas like Cornwall are tested for radon levels.
- Gamma Rays from Ground & Buildings (~14%): Many building materials, such as bricks and concrete, are made from natural substances that contain trace amounts of radioactive isotopes. These emit gamma radiation, contributing to our overall dose.
- Food and Drink (~11.5%): All living things, including the plants and animals we eat, contain naturally occurring radioactive isotopes like Carbon-14 and Potassium-40. This internal radiation source is an unavoidable part of life.
- Cosmic Rays (~10%): The Earth is constantly bombarded by high-energy particles from space, originating from the Sun and distant supernovae. The atmosphere provides significant shielding, which is why the dose from cosmic rays increases with altitude. This is a measurable factor for airline pilots and cabin crew.
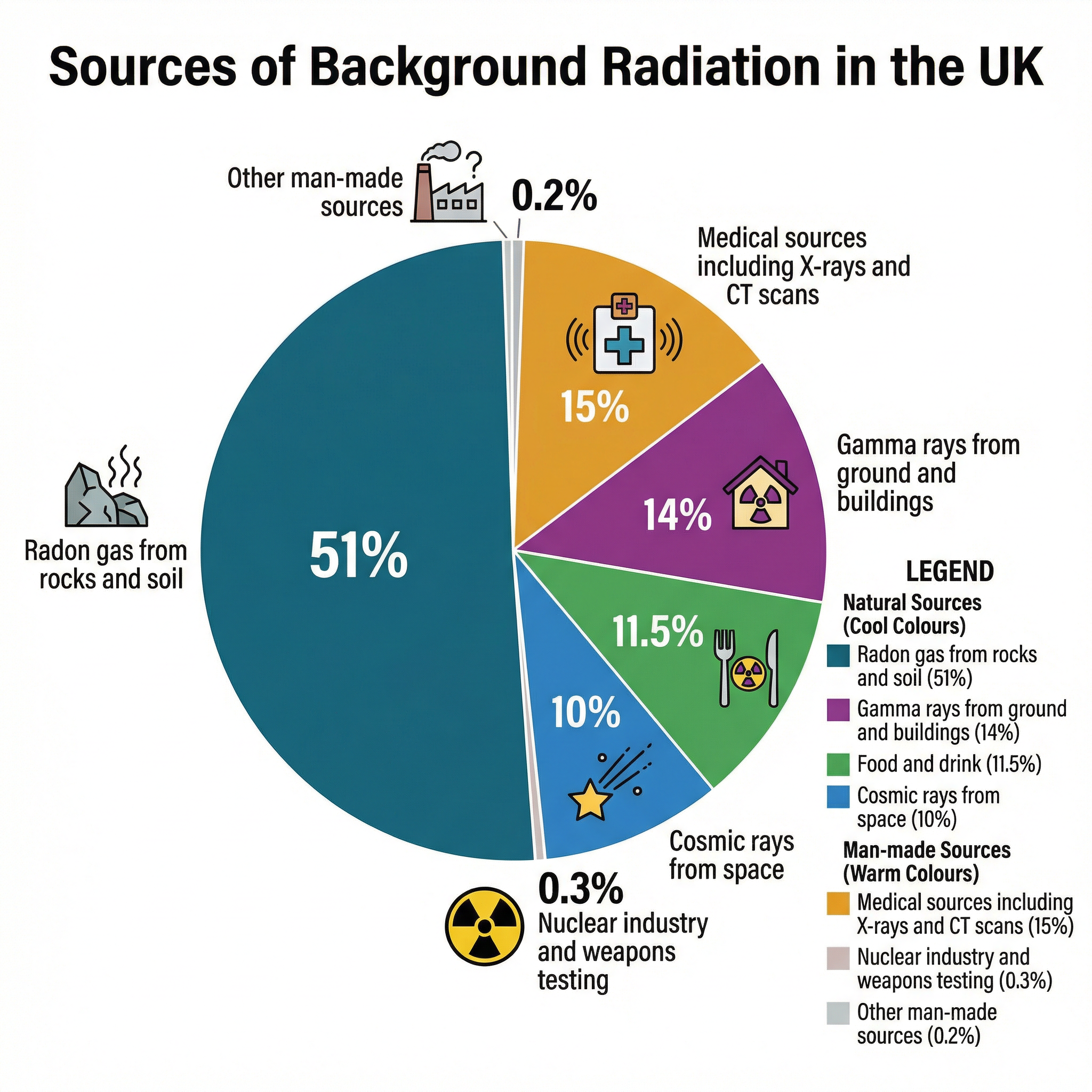
Man-Made (Artificial) Sources: These account for the remaining ~15% of the average dose.
- Medical Sources (~15%): This is the largest man-made source. It includes diagnostic procedures like X-rays, CT scans, and therapies using radioactive tracers (nuclear medicine). While the individual doses can be significant, the medical benefits are considered to outweigh the risks.
- Nuclear Industry & Weapons Testing (~0.3%): This is a common misconception among candidates. The contribution from nuclear power stations and the fallout from historical weapons testing is negligible, accounting for a tiny fraction of the total background dose.
- Other Sources (~0.2%): This includes a variety of minor sources, such as radioactive elements in smoke detectors or the luminous dials on old watches.
Concept 2: Correcting for Background Radiation
When measuring the radiation from a specific source using a Geiger-Müller (GM) tube, the detector measures both the radiation from the source and the background radiation simultaneously. To find the true activity of the source alone, candidates must perform a simple but critical calculation.
Example: A student measures the radiation from a cobalt-60 source and records a count rate of 450 counts per minute. Before placing the source, they measured the background count rate in the lab and found it to be 30 counts per minute. The actual count rate from the cobalt-60 is therefore not 450, but 420 counts per minute.

Concept 3: Activity and Dose
It is vital not to confuse the concepts of activity and dose. Examiners will penalise candidates who use these terms incorrectly.
- Activity: This measures how much radiation a source gives off. It is defined as the number of unstable nuclei that decay per second in a source. The unit of activity is the Becquerel (Bq). 1 Bq = 1 decay per second.
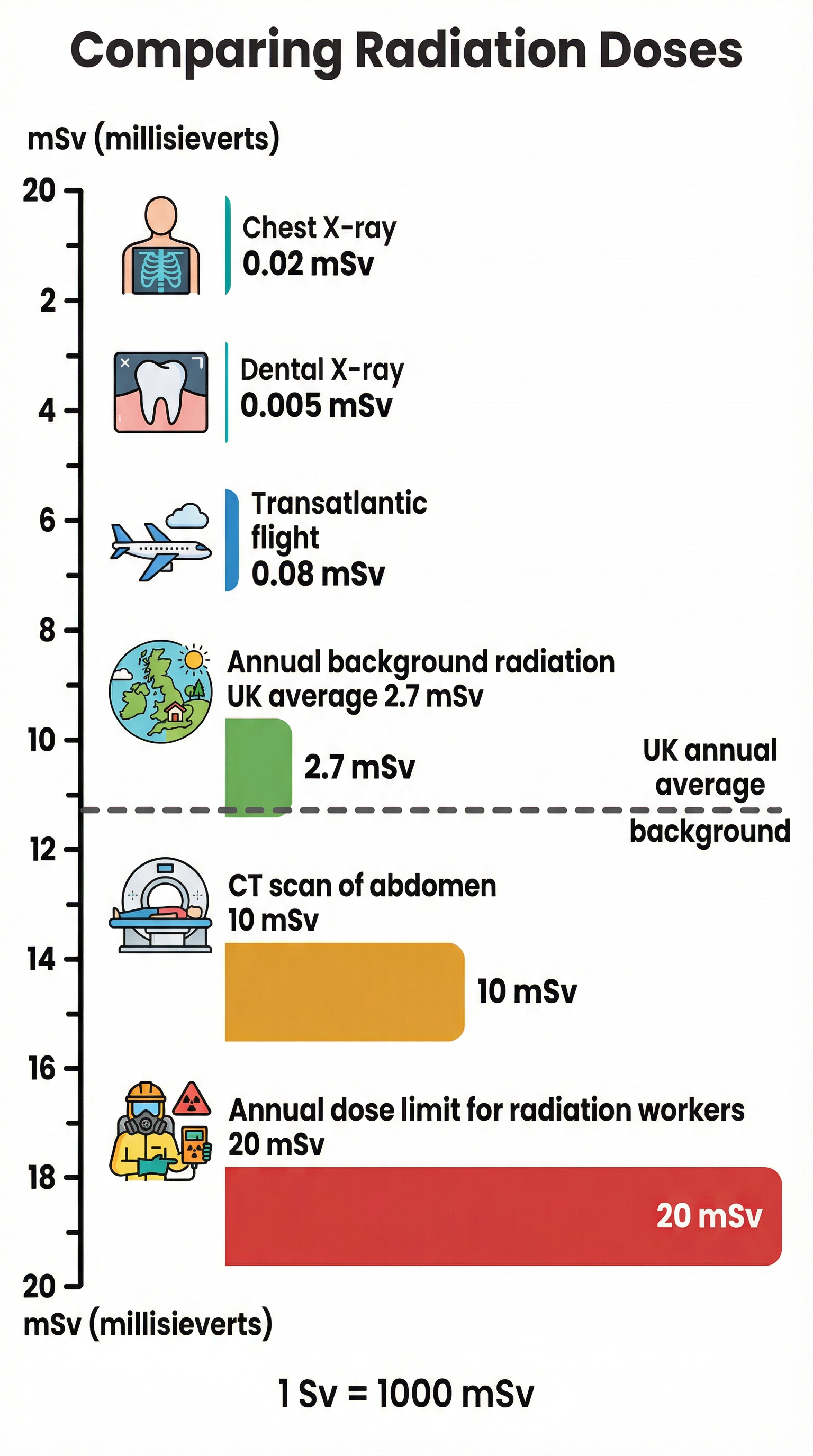
- Dose: This measures the potential harm to the body from being exposed to radiation. It takes into account the type of radiation, the energy of the radiation, and the part of the body exposed. The unit of radiation dose is the Sievert (Sv). As the Sievert is a very large unit, doses are almost always quoted in millisieverts (mSv) or microsieverts (μSv).
Mathematical/Scientific Relationships
Corrected Count Rate Formula (Must memorise)
This is the key formula for this topic and is not given on the formula sheet. Marks are awarded for showing its correct application.
Corrected Count Rate = Measured Count Rate – Background Count Rate
- Measured Count Rate: The reading on the detector (e.g., Geiger counter) with the source present. Units are typically counts per second (cps) or counts per minute (cpm).
- Background Count Rate: The reading on the detector with the source removed. This must be measured over the same time period as the main reading to be a fair test.
- Corrected Count Rate: The true activity of the source alone.
Unit Conversions (Must memorise)
Examiners frequently test unit prefixes in dose calculation questions.
- 1 Sievert (Sv) = 1,000 millisieverts (mSv)
- 1 millisievert (mSv) = 1,000 microsieverts (μSv)
To convert from Sv to mSv, you multiply by 1,000. To convert from mSv to Sv, you divide by 1,000.
Practical Applications
Understanding background radiation is fundamental to radiation protection and safety (radiological protection). Industries that involve radiation, such as medicine and nuclear power, must monitor their workers' exposure. Radiation workers wear dosimeters (like film badges) to record their accumulated dose over time, ensuring it stays below strict legal limits (e.g., 20 mSv per year in the UK). It also informs public health decisions, such as the need for radon testing in homes in certain geographical areas. The randomness of background radiation is also a perfect practical demonstration of the random nature of radioactive decay at the atomic level.
 "
"



