Density — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
Master OCR GCSE Physics Topic 6.2: Density. This guide breaks down the core concepts, the essential ρ = m/V formula, and the required practical for measuring regular and irregular solids. Secure top marks by mastering unit conversions and particle model explanations.
## Overview
Welcome to your deep dive into Density (OCR GCSE Physics, Topic 6.2). This topic is a cornerstone of the ‘Matter’ module and is frequently examined. Density is the measure of how ‘compact’ a substance is – how much mass is squeezed into a given volume. A solid understanding is crucial as it links directly to the particle model of matter, states of change, and pressure. Examiners assess this through calculation questions (often involving unit conversions), descriptive questions about the required practical (PAG P1), and explanation questions linking particle arrangement to density. Expect to see this topic appear in both Foundation and Higher tier papers, often carrying between 4-8 marks per question.
## Key Concepts
### Concept 1: The Density Equation
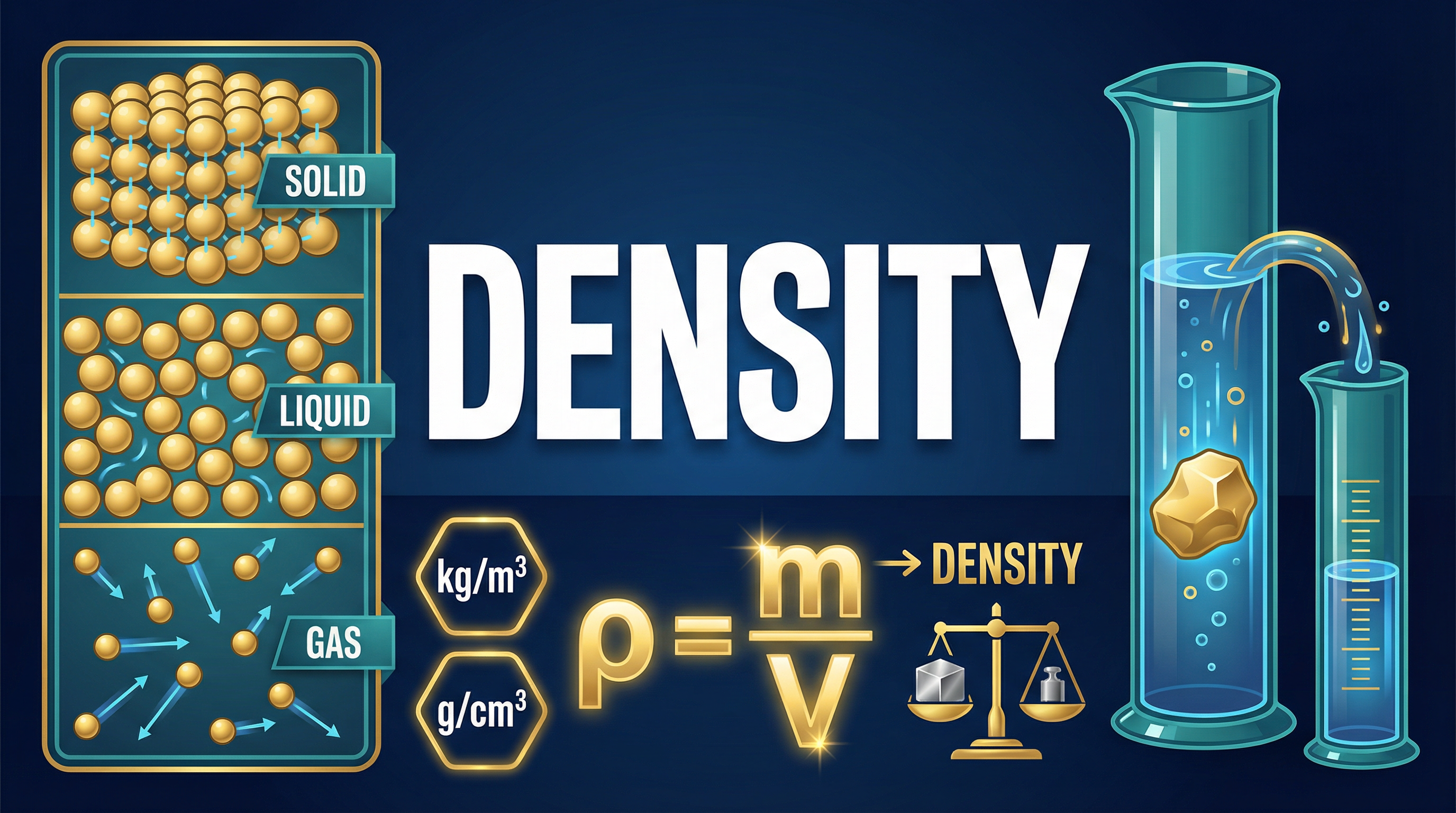
Density is a fundamental property of matter that quantifies the relationship between a substance's mass and the volume it occupies. The defining equation for density is:
**Density = Mass / Volume**
In symbolic form, this is written as:
**ρ = m / V**
Where:
- **ρ (rho)** is the symbol for density, measured in kilograms per metre cubed (kg/m³) or grams per centimetre cubed (g/cm³).
- **m** is the mass, measured in kilograms (kg) or grams (g).
- **V** is the volume, measured in metres cubed (m³) or centimetres cubed (cm³).
Candidates must be confident in rearranging this formula to solve for mass (m = ρ x V) or volume (V = m / ρ). Credit is always given for correctly stating the formula and showing the substitution of values.
### Concept 2: The Particle Model of Matter
The density of a substance is directly explained by the arrangement and spacing of its constituent particles. Examiners expect candidates to link macroscopic properties (like high or low density) to the microscopic particle model.
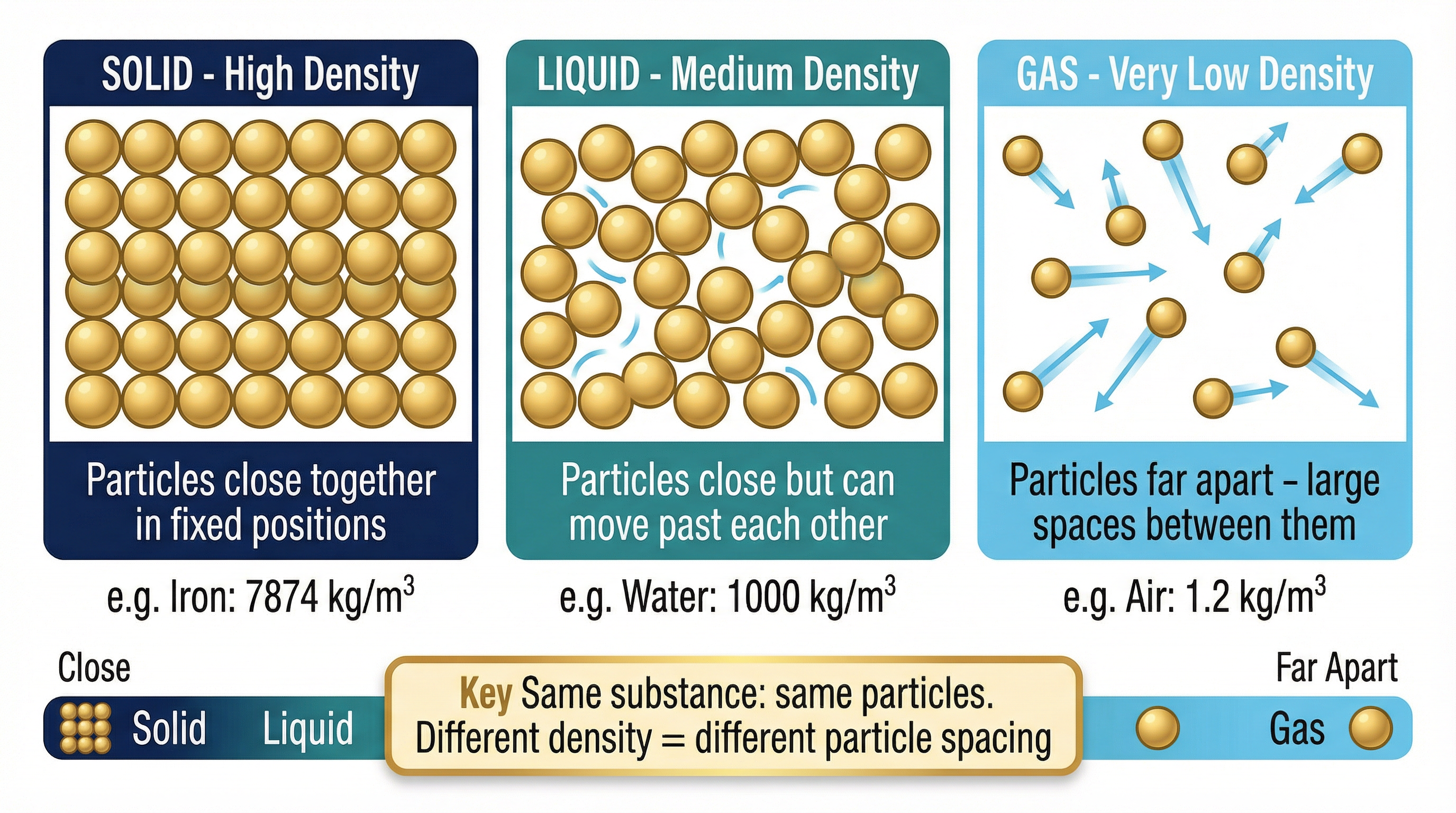
- **Solids**: Particles are tightly packed in a fixed, regular lattice. The forces between particles are very strong, holding them in place. This means a large number of particles (and therefore a large mass) are packed into a small volume, resulting in a **high density**.
- **Liquids**: Particles are still closely packed but are arranged irregularly and can move past one another. The forces are weaker than in solids. Their density is typically slightly lower than the solid form of the same substance because the particles are not as tightly packed.
- **Gases**: Particles are very far apart with no regular arrangement. The forces between particles are negligible. There are huge spaces between the particles, meaning very few particles (and therefore a very small mass) occupy a large volume. This results in a **very low density**.
## Mathematical/Scientific Relationships
### The Density Formula (Must Memorise)
**ρ = m / V**
This formula is **not** given on the OCR formula sheet, so it must be committed to memory.
### Unit Conversions
A common source of error is incorrect unit conversion. OCR will often provide values in grams and centimetres but ask for the final answer in kilograms and metres cubed.

- **From g/cm³ to kg/m³**: Multiply by 1000.
- **From kg/m³ to g/cm³**: Divide by 1000.
**Crucial Volume Conversion**: Remember that 1 metre is 100 centimetres. Therefore, to find how many cm³ are in a m³, you must calculate 100 x 100 x 100.
**1 m³ = 1,000,000 cm³**
## Practical Applications
### Required Practical (PAG P1): Determining Density
This is a core practical and is frequently tested. You need to know the method for both regular and irregular solids.
**Apparatus List**:
- A regular solid (e.g., a metal cube)
- An irregular solid (e.g., a rock or pebble)
- A 30 cm ruler or vernier calipers
- A top-pan balance (digital balance)
- A displacement (Eureka) can
- A measuring cylinder
- A beaker of water
- String
**Method for a Regular Solid (e.g., a Cube)**:
1. Use the balance to measure the mass of the cube. Record this in kg.
2. Use the ruler or vernier calipers to measure the length, width, and height of the cube in metres.
3. Calculate the volume using the formula: Volume = length × width × height.
4. Use the density formula (ρ = m/V) to calculate the density.
**Method for an Irregular Solid (e.g., a Rock)**:
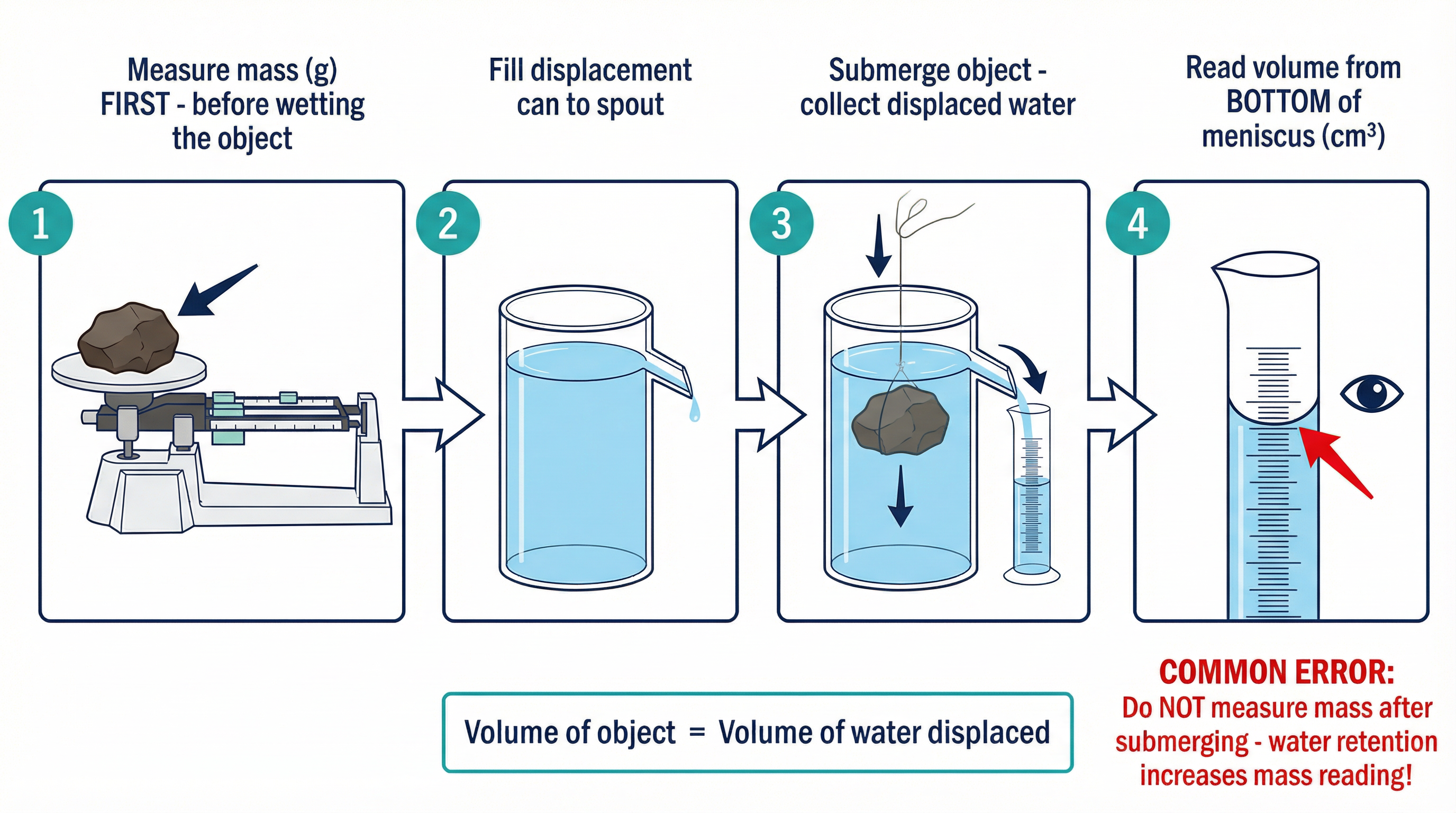
1. **Measure the mass** of the rock using the balance. **This is the most important first step.** Doing it later will result in an inaccurate reading as the rock will be wet.
2. Fill the displacement can with water until the water is just about to flow out of the spout.
3. Place an empty measuring cylinder under the spout.
4. Carefully lower the rock into the water, using a piece of string. Ensure it is fully submerged.
5. The volume of water that flows out of the spout and is collected in the measuring cylinder is equal to the volume of the rock.
6. Read the volume from the measuring cylinder. Ensure you read from the **bottom of the meniscus** at eye level to avoid parallax error.
7. Use the density formula (ρ = m/V) to calculate the density.
