Fission and Fusion — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
Unlock top marks in GCSE Physics with this guide to Nuclear Fission and Fusion (OCR P6.4). Discover the secrets of chain reactions and stellar energy, and learn exactly what examiners want to see in your answers.

## Overview
Nuclear Fission and Fusion are two of the most powerful processes in the universe, releasing vast amounts of energy from the atomic nucleus. This topic, P6.4 in the OCR specification, is crucial for understanding how nuclear power stations generate electricity and how stars like our Sun produce light and heat. For your exam, a deep understanding of the mechanics of induced fission, chain reactions, and the extreme conditions required for fusion is essential. Examiners frequently test this area with high-mark extended response questions, particularly focusing on the components of a nuclear reactor and the physics of overcoming electrostatic repulsion in fusion. This guide will break down these complex ideas into clear, mark-scoring points, ensuring you can tackle any question with confidence.

## Key Concepts
### Concept 1: Induced Nuclear Fission
Nuclear Fission is the **splitting of a large, unstable atomic nucleus** into two smaller nuclei. In a nuclear reactor, this process doesn't happen spontaneously; it must be **induced**. This is a key detail that earns marks. The process begins when a slow-moving **neutron** is absorbed by a large nucleus, most commonly **Uranium-235**. This absorption makes the nucleus highly unstable, causing it to split almost instantly.
This splitting releases:
1. **Two smaller daughter nuclei** (e.g., Barium and Krypton).
2. **Two or three fast-moving neutrons**.
3. **Gamma (γ) radiation**.
4. A tremendous amount of **energy**.
The energy release is a direct consequence of Einstein's famous equation, E=mc². The total mass of the products (daughter nuclei + neutrons) is slightly less than the mass of the original Uranium-235 nucleus and the initial neutron. This 'missing' mass, known as the **mass defect**, is converted directly into energy. **Crucially, you must state that mass is converted into energy, not that 'energy is created', which violates the principle of conservation of energy and will lose you marks.**
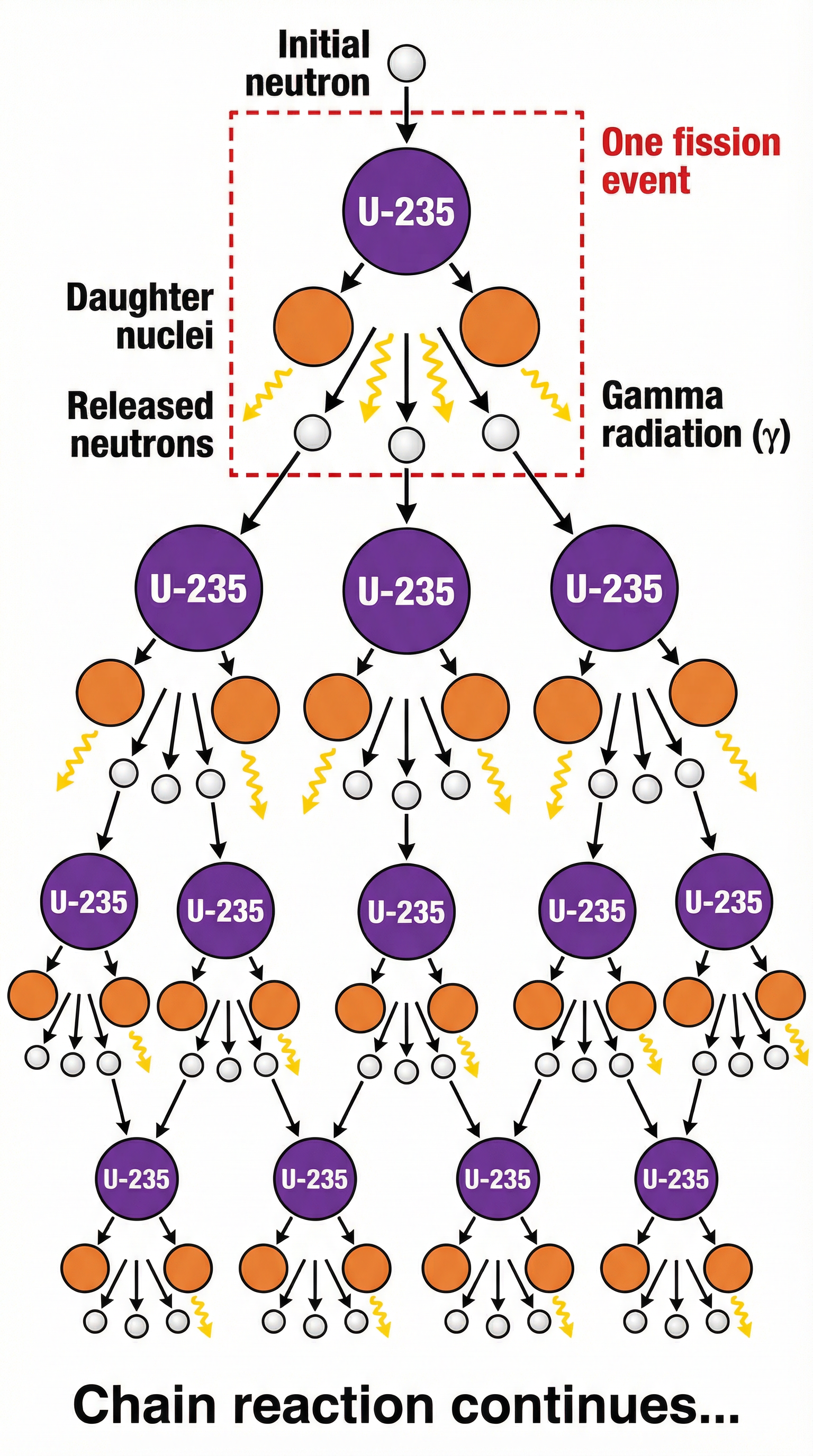
### Concept 2: The Chain Reaction
The release of 2-3 neutrons from each fission event is what makes a **chain reaction** possible. Each of these newly released neutrons can go on to be absorbed by another Uranium-235 nucleus, inducing another fission event, which in turn releases more neutrons, and so on. This creates a self-sustaining and rapidly escalating reaction.
- **Uncontrolled Chain Reaction**: If every released neutron causes another fission, the reaction grows exponentially. This is the principle behind an atomic bomb, releasing all its energy in a fraction of a second.
- **Controlled Chain Reaction**: In a nuclear power station, the goal is to maintain a steady release of energy. This is achieved by ensuring that, on average, **exactly one** neutron from each fission event goes on to cause one further fission. This creates a critical, stable state where the reaction rate is constant.
### Concept 3: The Nuclear Reactor
A nuclear reactor is a device designed to initiate and control a sustained nuclear chain reaction to generate electricity. For a 6-mark question, you must describe the function of its key components.
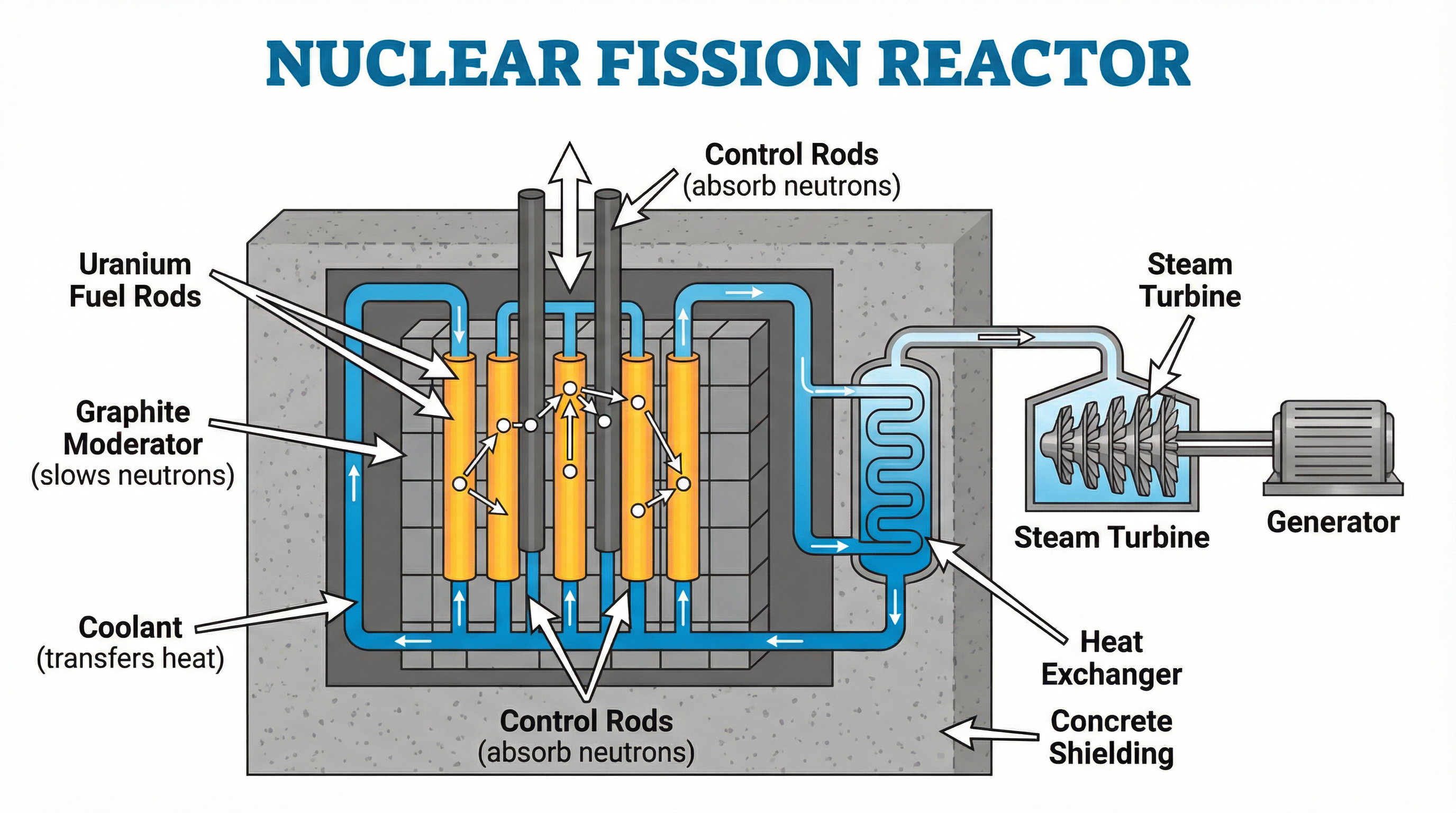
| Component | Material | Function | Examiner Tip |
| :--- | :--- | :--- | :--- |
| **Fuel Rods** | Uranium-235 (enriched) | Provides the large, unstable nuclei for fission. | State that U-235 is the specific isotope used. |
| **Moderator** | Graphite or Water | **Slows down** the fast-moving neutrons released during fission. | Neutrons must be slowed to 'thermal speeds' to be efficiently absorbed by U-235. **Do not confuse with control rods.** |
| **Control Rods** | Boron or Cadmium | **Absorb** surplus neutrons to control the rate of the chain reaction. | Raising the rods increases the rate; lowering them decreases it. Fully inserting them shuts the reactor down. |
| **Coolant** | Water or CO₂ Gas | Transfers thermal energy from the reactor core to a heat exchanger. | The heat is used to boil water, creating steam to turn a turbine. |
| **Shielding** | Thick Concrete & Steel | Absorbs hazardous gamma radiation and neutrons, preventing them from escaping. | Essential for safety. |
### Concept 4: Nuclear Fusion
Nuclear Fusion is the process of **joining two light atomic nuclei** to form a single, heavier nucleus. This process releases even more energy per kilogram of fuel than fission. This is the reaction that powers the Sun and all other stars.
In the Sun, hydrogen isotopes (like Deuterium and Tritium) fuse under immense pressure and temperature to form a Helium nucleus. A small amount of mass is converted into a vast quantity of energy.
**The Challenge of Fusion on Earth**: To make two nuclei fuse, they must get close enough for the strong nuclear force to bind them. However, both nuclei are positively charged and so **electrostatically repel** each other very strongly. To overcome this repulsion, the nuclei must be heated to extreme temperatures (over 100 million °C) to give them enough kinetic energy to collide successfully. At these temperatures, the fuel exists as a **plasma** (a gas of ions). Containing this plasma is a major scientific challenge, often using powerful magnetic fields in a device called a tokamak.
## Mathematical/Scientific Relationships
### Balancing Nuclear Equations
When balancing nuclear equations, two principles must be upheld:
1. **Conservation of Mass Number (A)**: The sum of the mass numbers (top numbers) on the left side must equal the sum on the right side.
2. **Conservation of Atomic Number (Z)**: The sum of the atomic numbers (bottom numbers) on the left side must equal the sum on the right side.
**Example (Fission)**:
¹n₀ + ²³⁵U₉₂ → ¹⁴¹Ba₅₆ + ⁹²Kr₃₆ + 3(¹n₀) + Energy
- **Mass Number Check**: 1 + 235 = **236** (Left) | 141 + 92 + 3(1) = **236** (Right) ✔
- **Atomic Number Check**: 0 + 92 = **92** (Left) | 56 + 36 + 3(0) = **92** (Right) ✔
### Energy-Mass Equivalence
**E = mc²**
- **E**: Energy released (in Joules, J)
- **m**: Mass defect (the mass that was converted, in kilograms, kg)
- **c**: The speed of light in a vacuum (approximately 3 x 10⁸ m/s)
This formula is **given on the formula sheet**, but you must understand what it represents: a tiny amount of mass can be converted into a huge amount of energy.
## Practical Applications
- **Nuclear Power Stations**: Use controlled nuclear fission to generate electricity for the National Grid. They provide a reliable, low-carbon energy source but produce radioactive waste that must be managed carefully.
- **Medical Tracers**: Radioisotopes produced in reactors can be used in medicine for diagnosis and therapy.
- **Academic Research**: Fusion reactors like the Joint European Torus (JET) in the UK are used to research the feasibility of fusion as a future energy source.
- **Stellar Physics**: Understanding fusion is fundamental to astrophysics, explaining how stars are born, live, and die.