Nuclear Fission — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
This guide provides a comprehensive overview of Nuclear Fission (OCR GCSE Physics 7.8), focusing on the core principles of chain reactions, reactor components, and balancing nuclear equations. It is designed to build confidence and help students secure maximum marks in their exams."
## Overview
Nuclear fission is the process of splitting a large atomic nucleus into smaller ones, releasing a vast amount of energy. For your OCR GCSE Physics exam, understanding this topic is crucial, as it not only appears in dedicated questions but also links to concepts of energy, radioactivity, and atomic structure. This guide will break down the mechanics of induced fission, explain the controlled chaos of a chain reaction, and demystify the workings of a nuclear reactor. We will cover both Foundation and Higher Tier content, ensuring you are prepared for any question, from simple definitions to complex 6-mark explanations and equation balancing.
## Key Concepts
### 1. Induced Fission
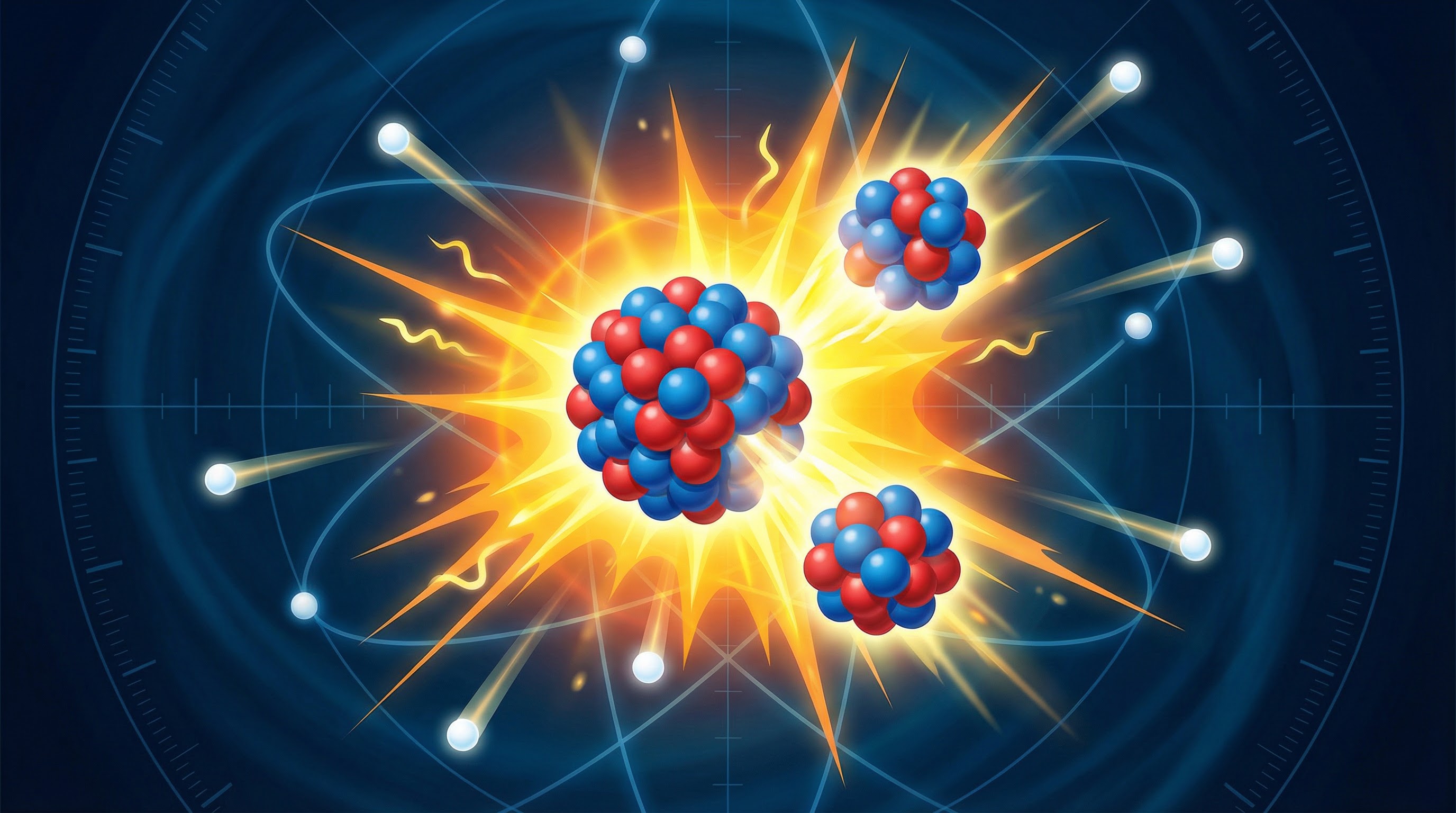
Unlike radioactive decay, which can be spontaneous, the type of fission used in nuclear reactors is **induced**. This means it is started deliberately. The process begins when a slow-moving **neutron** is absorbed by a large, unstable nucleus, typically **Uranium-235**. The nucleus becomes highly unstable (now Uranium-236) and immediately splits.
**Example**: Imagine throwing a small, slow-moving tennis ball into a large, wobbly tower of blocks. The impact is enough to make the whole tower collapse into smaller piles. The tennis ball is the neutron, and the wobbly tower is the U-235 nucleus.
### 2. The Products of Fission
When the U-235 nucleus splits, it forms two smaller nuclei, known as **daughter nuclei**. These are not always the same; there are many possible combinations, but they are always of roughly equal size. For example, Barium-141 and Krypton-92 are common products. Crucially, the split also releases:
- **2 or 3 neutrons**: These are key to the chain reaction.
- **Energy**: Released as gamma rays and the kinetic energy of the fission products.
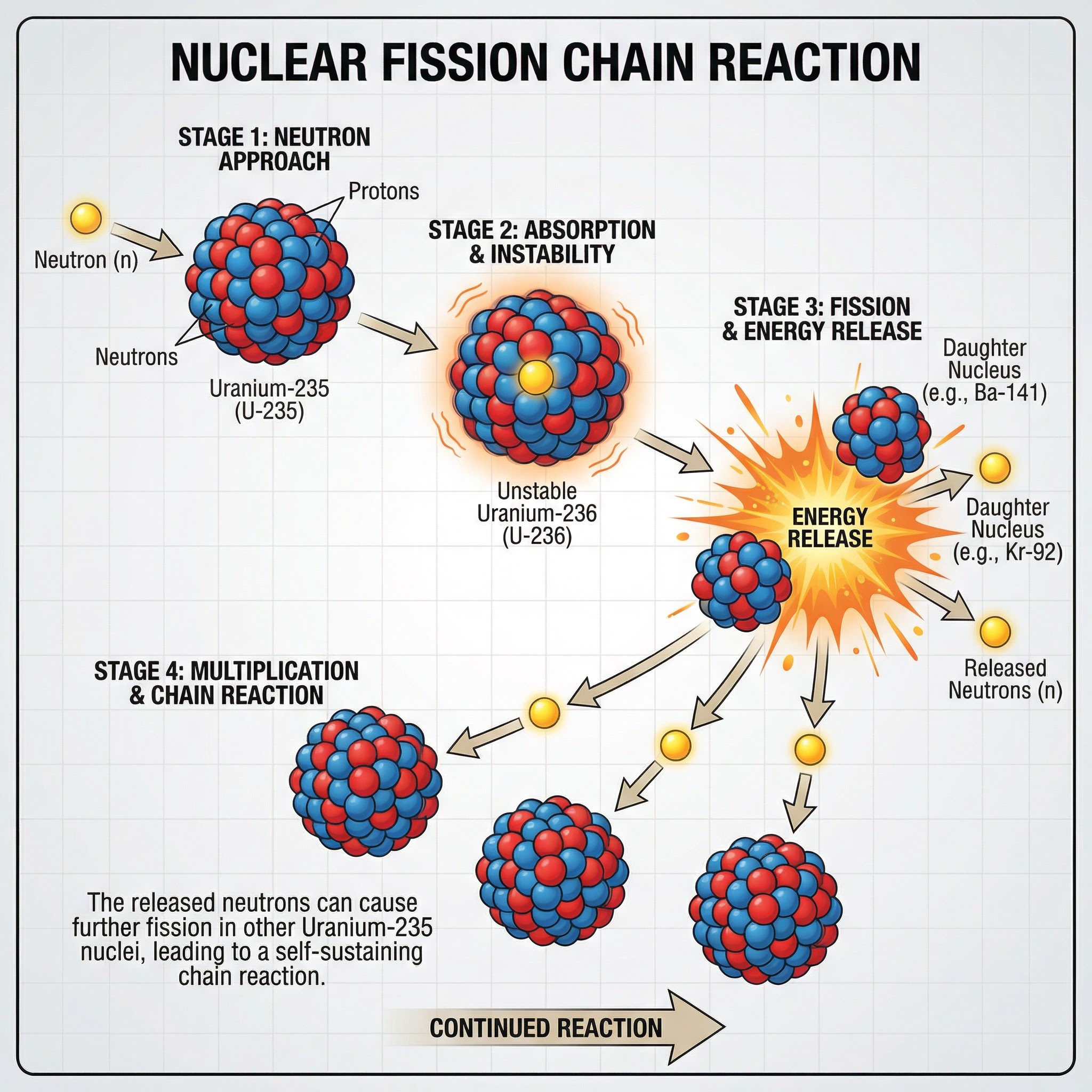
### 3. The Chain Reaction
A chain reaction is a self-sustaining process where the neutrons released from one fission event go on to trigger further fission events. If one fission releases 3 neutrons, those 3 neutrons can cause 3 more fissions, releasing 9 neutrons, which can then cause 9 more fissions, and so on. In a nuclear weapon, this reaction is uncontrolled, leading to an explosive release of energy. In a nuclear reactor, it must be precisely controlled.
### 4. Controlling Fission: The Nuclear Reactor
To use fission for electricity generation, the chain reaction must be maintained at a steady rate, where, on average, only **one** neutron from each fission event goes on to cause another. This is achieved using key components:
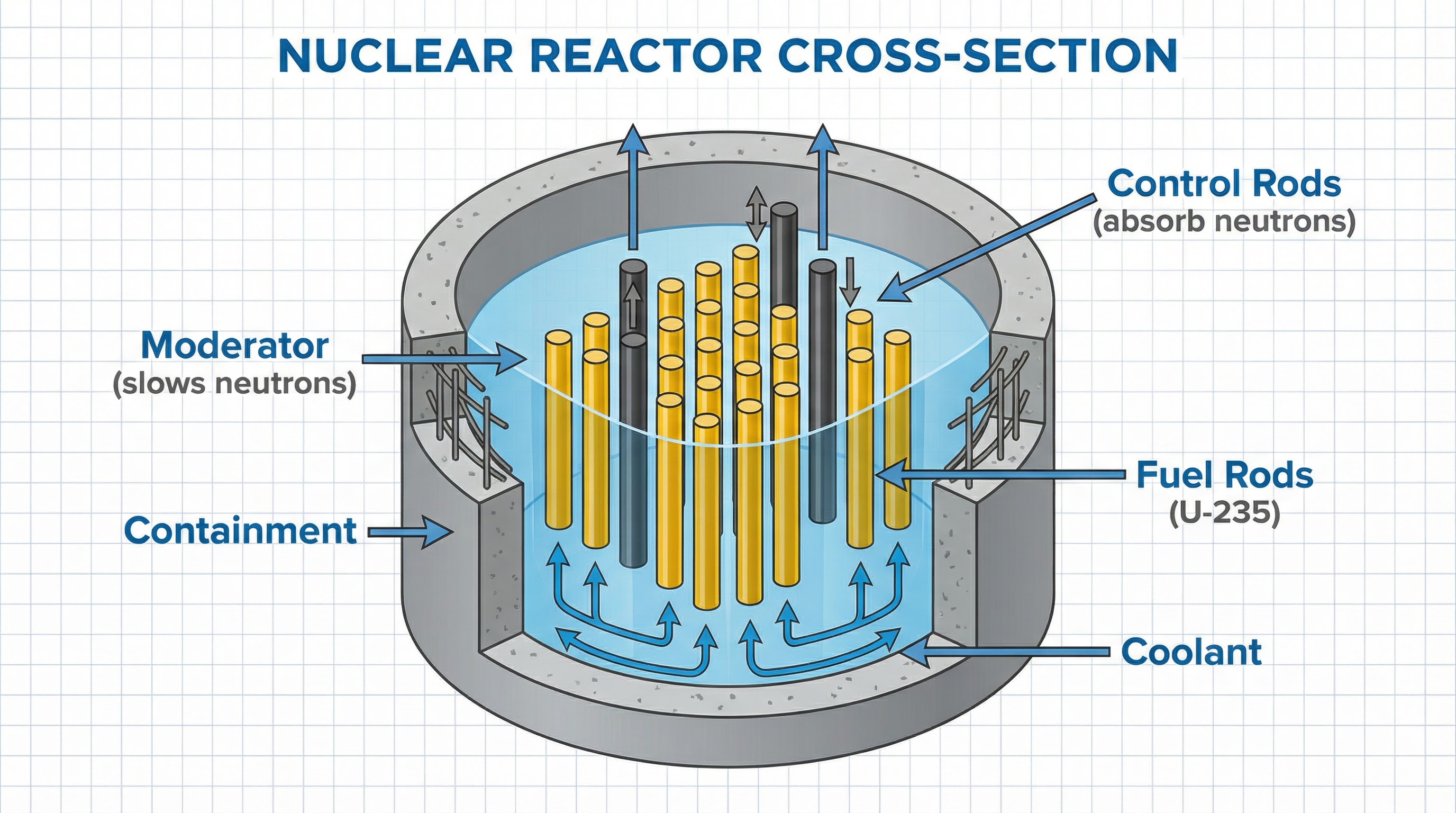
- **Fuel Rods**: Contain the fissile material, usually enriched Uranium-235.
- **Moderator**: This material (often water or graphite) surrounds the fuel rods. Its job is to **slow down** the fast-moving neutrons released during fission. Slow neutrons are much more likely to be absorbed by U-235 and cause further fission. Fast neutrons tend to just bounce off.
- **Control Rods**: Made of a neutron-absorbing material like boron or cadmium. These can be raised or lowered into the reactor core. Lowering the rods absorbs more neutrons, slowing the reaction down. Raising them absorbs fewer neutrons, speeding the reaction up. This is how the rate of fission is precisely controlled.
- **Coolant**: Usually water, this flows through the reactor to absorb the immense heat generated. The hot coolant is then used to boil water, create steam, and turn a turbine to generate electricity.
## Mathematical/Scientific Relationships
**(Higher Tier Only)**
Nuclear equations must be balanced. The total mass number (top number) and atomic number (bottom number) must be the same on both sides of the equation.
**General Equation for Fission:**
¹n + ²³⁵U → [²³⁶U]* → X + Y + (2-3)¹n + Energy
Where:
- ¹n is the neutron.
- ²³⁵U is the Uranium-235 nucleus.
- [²³⁶U]* is the temporary, unstable Uranium-236 nucleus.
- X and Y are the two daughter nuclei.
**Example Equation:**
¹n + ²³⁵₉₂U → ¹⁴¹₅₆Ba + ⁹²₃₆Kr + 3¹n
**Checking the balance:**
- **Mass Number (Top):** 1 + 235 = 236 on the left. 141 + 92 + (3 x 1) = 236 on the right. It balances.
- **Atomic Number (Bottom):** 0 + 92 = 92 on the left. 56 + 36 + (3 x 0) = 92 on the right. It balances.
## Practical Applications
Nuclear fission is the primary process used in **nuclear power stations** to generate electricity. The heat from the controlled chain reaction is used to produce steam, which drives turbines connected to generators. This provides a low-carbon source of electricity, as no fossil fuels are burned in the process. However, the management of radioactive nuclear waste is a significant challenge.
