Risks of Radiation — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
This guide provides a comprehensive, exam-focused breakdown of OCR GCSE Physics Topic 7.7: Risks of Radiation. It covers the crucial distinction between irradiation and contamination, the biological effects of different radiation types, and the safety protocols examiners expect you to know, ensuring you are prepared to secure every available mark.

## Overview
Welcome to your deep dive into the Risks of Radiation (OCR GCSE Physics, 7.7). This topic is a cornerstone of nuclear physics and carries significant weight in exams, accounting for a substantial portion of AO2 (application) and AO3 (evaluation) marks. It explores the hazards associated with ionising radiation and the crucial distinction between being irradiated and becoming contaminated. Understanding this topic is not just about memorising facts; it’s about applying your knowledge of alpha, beta, and gamma radiation to real-world scenarios, from medical treatments to nuclear waste management. Examiners will test your ability to evaluate risks, justify safety precautions, and interpret data related to radiation dose and half-life. This guide will equip you with the precise language and conceptual understanding needed to excel.

## Key Concepts
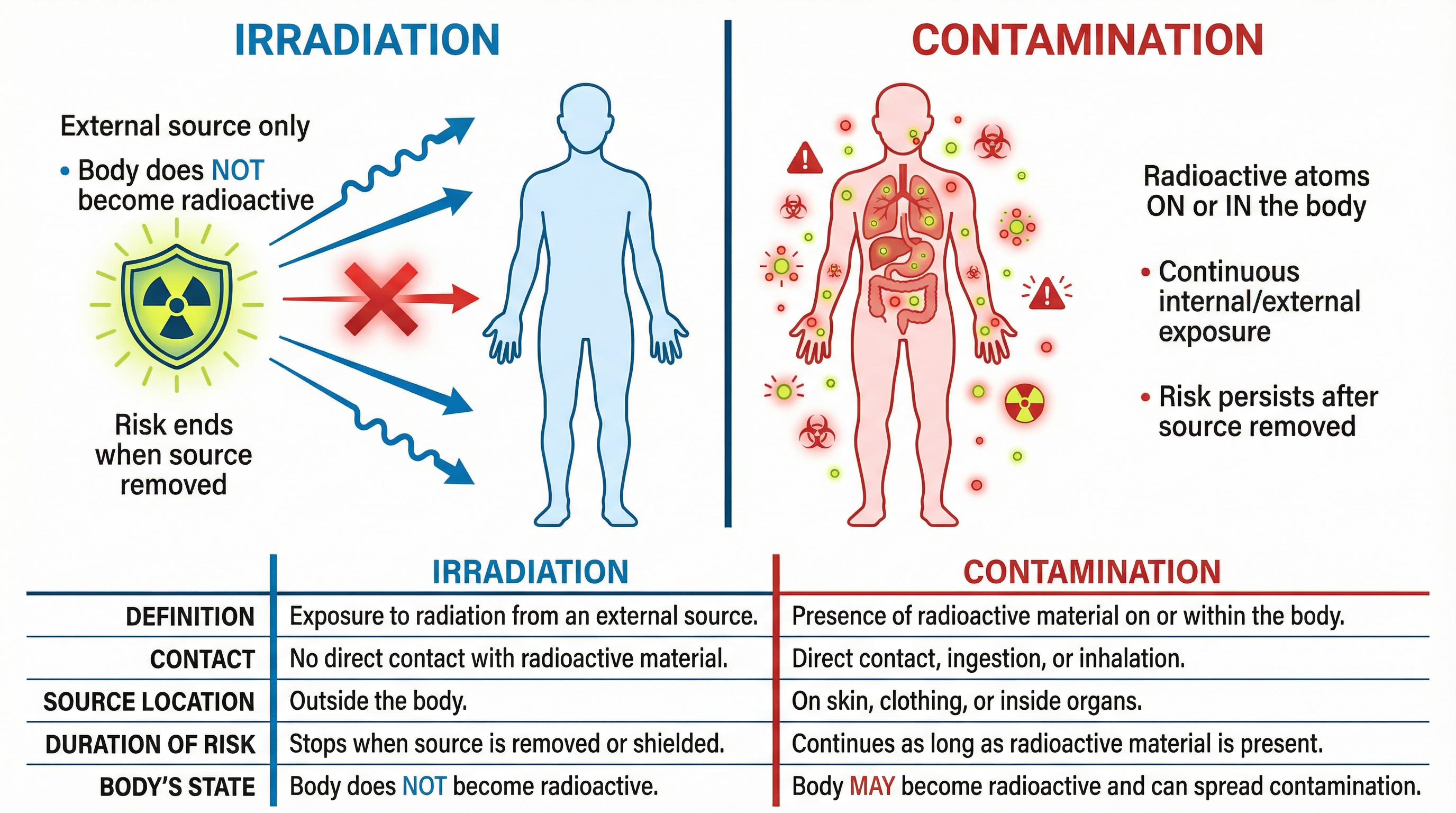
### Concept 1: Irradiation vs. Contamination
This is the single most important distinction in the topic, and a frequent source of confusion. Getting this right is essential for high marks.
**Irradiation** is the process where an object or body is exposed to ionising radiation from an external source. The key takeaway is that the object does **not** come into contact with the radioactive source itself and does **not** become radioactive. The risk of harm only exists while the object is exposed to the radiation.
* **Analogy**: Standing in the sun. You are being irradiated by UV rays, but you do not start emitting sunlight yourself. When you go indoors, the irradiation stops.
* **Example**: A patient having a cancerous tumour treated with a beam of gamma rays from a machine (radiotherapy). The patient is irradiated, but they can leave the hospital immediately without posing a risk to others.
**Contamination** occurs when a radioactive source gets onto or into an object or body. The radioactive atoms are physically present, meaning the object itself becomes a source of radiation. The risk is continuous and persists until the contaminant is removed or decays.
* **Analogy**: Spilling glowing paint on your clothes. The paint is now on you, continuously emitting light, and you could transfer it to other things you touch.
* **Example**: Radioactive dust from the Chernobyl disaster landing on farmland. The soil and the plants that grow in it become contaminated and can be dangerous if ingested.
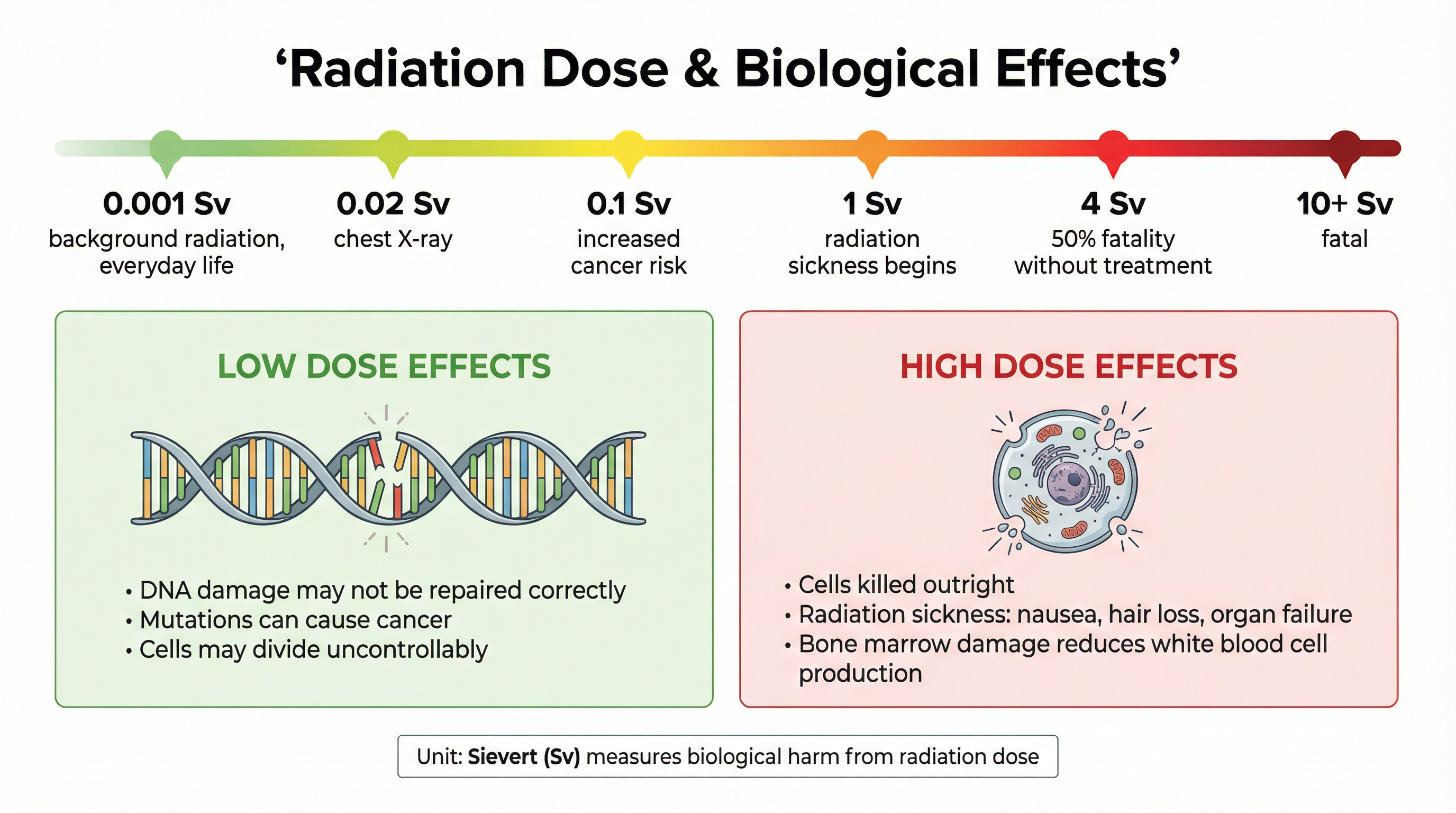
### Concept 2: Biological Effects and Dose
The danger of ionising radiation lies in its ability to knock electrons out of atoms and molecules in living cells, particularly DNA. This can have two main outcomes depending on the dose.
* **Low Dose Effects**: At lower doses, radiation can cause mutations in DNA. If the cell's repair mechanisms fail, this can lead to the cell dividing uncontrollably, which is the cause of **cancer**. There is a time lag between exposure and the potential onset of cancer.
* **High Dose Effects**: At high doses, the radiation can kill cells outright. This leads to **radiation sickness**, with symptoms like nausea, vomiting, skin burns, and hair loss. Very high doses can damage vital organs and bone marrow, leading to death within weeks or days.
Radiation dose, which measures the potential for biological harm, is measured in **Sieverts (Sv)** or millisieverts (mSv). One Sievert is a very large dose.
### Concept 3: Risk Variation by Radiation Type
The risk posed by a radioactive source depends critically on its type (alpha, beta, or gamma) and whether it is **external** or **internal** (contamination).
| Radiation Type | External Risk (Outside Body) | Internal Risk (Inside Body - Contamination) | Justification |
| :------------- | :----------------------------------------------------------- | :------------------------------------------------------------------------ | :-------------------------------------------------------------------------------------------------------- |
| **Alpha (α)** | **Low** | **Very High** | Alpha is stopped by skin, but if inhaled/ingested, its high ionising power causes intense damage to local tissue. |
| **Beta (β)** | **Medium** | **High** | Beta can penetrate skin to damage underlying cells. It causes significant damage internally. |
| **Gamma (γ)** | **High** | **Lower** | Gamma is highly penetrating and can irradiate internal organs from outside. It is less ionising than alpha, so causes less localised damage internally. |
### Concept 4: Safety Precautions
To minimise the risk from radiation, three key principles are applied. Examiners expect you to name them and give specific examples.
1. **Time**: Minimise the time of exposure. The less time spent near a source, the lower the total dose received.
* *Example*: Radiographers leaving the room during an X-ray exposure.
2. **Distance**: Maximise the distance from the source. Radiation intensity follows an inverse square law – doubling the distance reduces the intensity to a quarter.
* *Example*: Using long-handled tongs to move radioactive sources.
3. **Shielding**: Use appropriate materials to absorb the radiation.
* *Example*: Storing sources in lead-lined containers. Radiographers wearing lead aprons.

## Mathematical/Scientific Relationships
There are no complex formulas to memorise for this specific topic, but you must understand the conceptual relationships:
* **Inverse Square Law (Conceptual)**: Intensity ∝ 1 / (distance)². You are not required to perform calculations with this formula at GCSE, but you must state that radiation intensity decreases rapidly with distance.
* **Half-Life Calculations**: While primarily covered in topic 7.6, you must be able to apply half-life concepts here. A key skill is remembering to subtract the background count rate from your measurements before calculating the half-life of a sample.
* **Formula**: Corrected Count = Total Count - Background Count
## Practical Applications
* **Medical Tracers**: A radioactive isotope with a short half-life (e.g., Technetium-99m) is injected into a patient. It is chosen to be a gamma emitter so the radiation can be detected outside the body. The half-life must be long enough for the diagnosis to take place, but short enough to minimise the radiation dose to the patient.
* **Radiotherapy**: A focused beam of high-energy gamma rays is used to kill cancerous cells. The beam is rotated around the patient to minimise damage to healthy tissue while delivering a high dose to the tumour.
* **Sterilisation**: Medical instruments are sterilised by irradiating them with a strong gamma source. This kills all microbes on the instruments without making the instruments themselves radioactive.
* **Nuclear Waste Disposal**: Waste from nuclear power stations is highly radioactive and often has a very long half-life. It must be stored securely for thousands of years. This usually involves vitrification (encasing it in glass), placing it in steel containers, and burying it deep underground in geologically stable locations.