Specific Heat Capacity — OCR GCSE Study Guide
Exam Board: OCR | Level: GCSE
This guide provides a comprehensive, exam-focused breakdown of Specific Heat Capacity (SHC) for OCR GCSE Physics (6.4). It covers the core concepts, the essential equation you must memorise, the required practical (PAG 1), and common pitfalls to help you secure every possible mark on this frequently tested topic.

## Overview
Specific Heat Capacity (SHC) is a fundamental concept in thermal physics that quantifies how much energy a substance must absorb to heat up. In your OCR GCSE exam, this topic, found in section P1 (Matter), is a rich source of marks, blending calculation, practical skills, and conceptual understanding. Examiners frequently test your ability to not only use the core equation but also to analyse the associated required practical (PAG 1), evaluating sources of error and suggesting improvements. A typical exam question might be a multi-step calculation involving a unit conversion, or a 6-mark question asking you to describe the experimental method and explain its limitations. Mastering SHC is crucial as it forms a synoptic link to topics like Conservation of Energy, Power, and States of Matter. This guide will equip you with the knowledge and exam technique to tackle any SHC question with confidence.

## Key Concepts
### Concept 1: What is Specific Heat Capacity?
In simple terms, specific heat capacity is a measure of a substance's resistance to changing temperature. Substances with a **high SHC** require a lot of energy to increase their temperature, while those with a **low SHC** heat up very quickly. The formal definition, which is worth 1-2 marks if you can state it precisely, is: **The energy required to raise the temperature of 1 kg of a substance by 1 °C.**
**Analogy: The Shopping Bag**
Think of thermal energy as groceries and temperature as how full a shopping bag looks. A substance with a low SHC is like a small, flimsy plastic bag – a few items (energy) make it look full (high temperature) very quickly. A substance with a high SHC, like water, is like a giant, sturdy hiking backpack. You can pile in tons of groceries (energy) before it even starts to look full (its temperature rises slowly). This is why water is used in central heating systems; it can carry a huge amount of thermal energy around your house.
### Concept 2: The Required Practical (PAG 1)
This is one of the most important parts of the topic. Examiners will test your knowledge of the apparatus, method, and, most importantly, the errors involved.
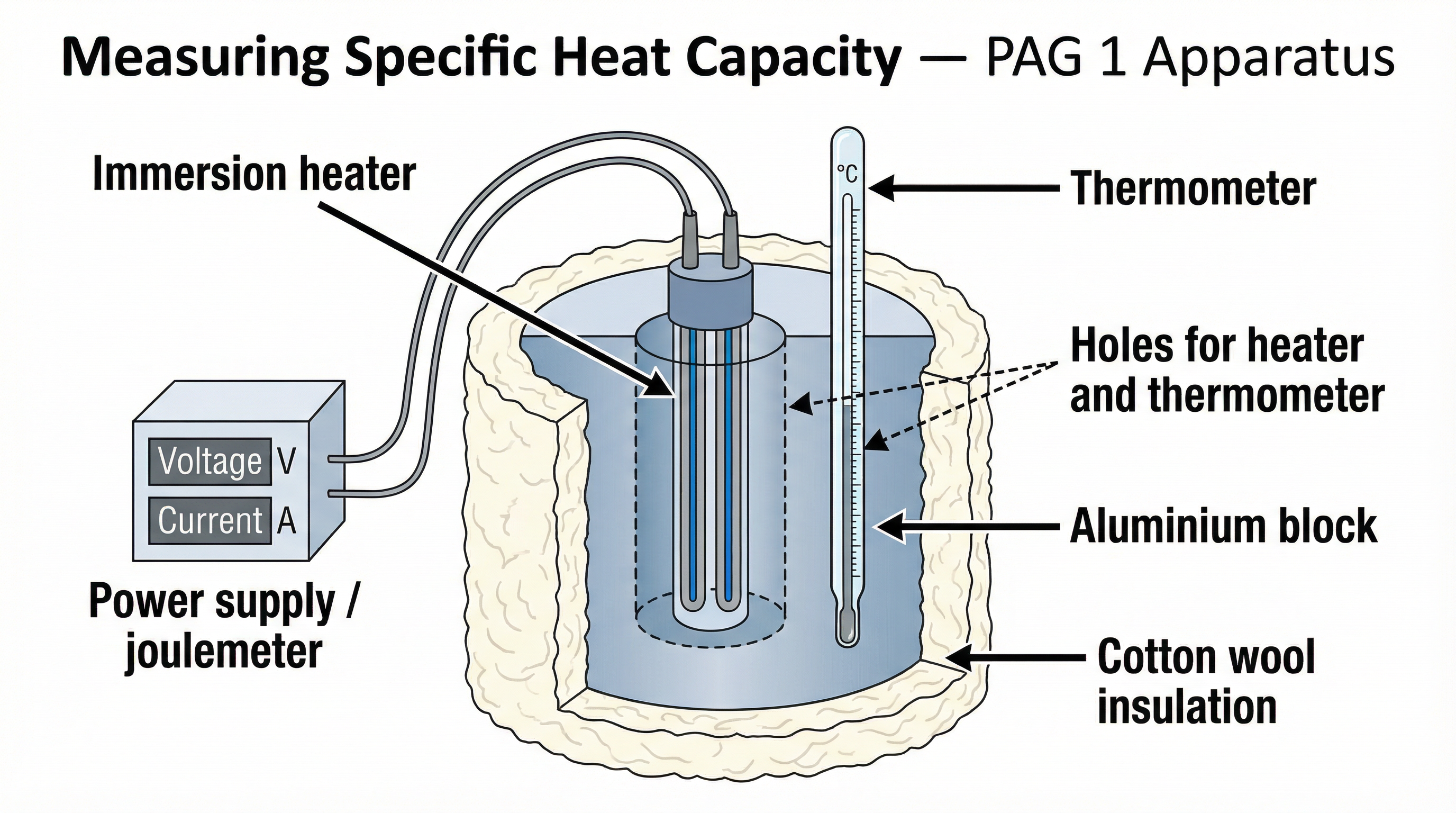
**Objective**: To measure the specific heat capacity of a material (e.g., an aluminium block).
**Method Summary**:
1. Measure the mass of the block in kilograms.
2. Place a heater in the larger hole and a thermometer in the smaller one. Add a drop of water to the thermometer hole to ensure good thermal contact.
3. Record the starting temperature of the block.
4. Connect the heater to a power supply and a joulemeter. Turn it on for a set amount of time (e.g., 10 minutes).
5. Record the total energy supplied by the joulemeter (ΔE) and the final temperature.
6. Calculate the temperature change (ΔT).
7. Use the rearranged equation `c = ΔE / (m × ΔT)` to find the specific heat capacity.
**Crucial Examiner Points**:
* **Insulation**: You MUST state that the block is insulated (e.g., wrapped in cotton wool) to **reduce the transfer of thermal energy to the surroundings**. Vague answers like 'to stop heat loss' will not be credited.
* **Systematic Error**: A key source of error is energy loss to the atmosphere. This means not all the energy from the heater goes into the block. This results in a smaller temperature change (ΔT) than expected. When you calculate `c = ΔE / (m × ΔT)`, dividing by a smaller ΔT gives a **calculated value for 'c' that is higher than the true value**. This is a 1-2 mark point that frequently appears.
## Mathematical/Scientific Relationships
The cornerstone of this topic is one vital equation. You are expected to know it by heart, as it is **not** provided on the OCR formula sheet.
**The Specific Heat Capacity Equation (Must Memorise)**
ΔE = m × c × ΔT
Where:
* **ΔE** = Change in Thermal Energy, measured in **Joules (J)**
* **m** = Mass, measured in **kilograms (kg)**
* **c** = Specific Heat Capacity, measured in **Joules per kilogram per degree Celsius (J/kg°C)**
* **ΔT** (delta T) = Change in Temperature, measured in **degrees Celsius (°C)**
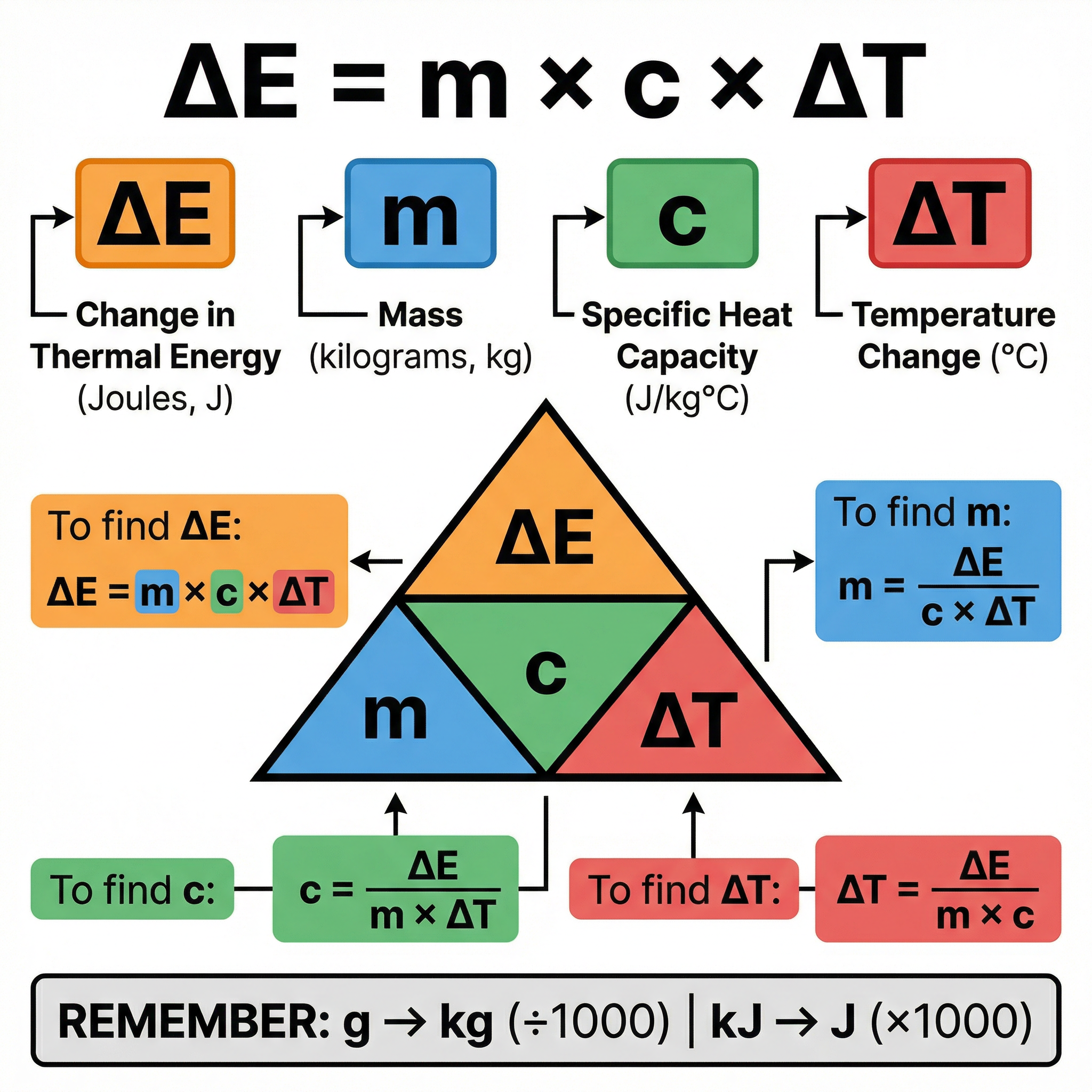
**Rearranging the Equation**
Examiners will often ask you to find 'c', 'm', or 'ΔT', so you must be confident in rearranging the formula:
* To find **c**: `c = ΔE / (m × ΔT)`
* To find **m**: `m = ΔE / (c × ΔT)`
* To find **ΔT**: `ΔT = ΔE / (m × c)`
**Unit Conversions (Common Mistakes)**
* **Mass**: If given in grams (g), you MUST convert to kilograms (kg) by dividing by 1000. (e.g., 500 g = 0.5 kg)
* **Energy**: If given in kilojoules (kJ), you MUST convert to joules (J) by multiplying by 1000. (e.g., 7.2 kJ = 7200 J)
## Practical Applications
* **Engine Coolant**: Water's very high SHC (4200 J/kg°C) makes it an excellent coolant. It can absorb a large amount of waste thermal energy from a car engine without its temperature rising to dangerous levels.
* **Saucepans**: The base of a saucepan is made from a material with a low SHC (like copper or aluminium), so it heats up very quickly to cook food. The handle, however, is made from a material with a high SHC and low thermal conductivity (like plastic), so it doesn't get hot.
* **Coastal Climates**: Coastal areas have milder climates than inland areas. The large body of water (ocean) has a high SHC, so it heats up slowly in summer and cools down slowly in winter, moderating the temperature of the nearby land.